Clear Sky Science · en
Amino acid and viral binding by the high-affinity Cationic Amino acid Transporter 1 (CAT1) from Mus musculus
How Cells Control Vital Building Blocks and Face Viral Invaders
Our cells depend on a steady flow of certain amino acids—tiny molecules that build proteins and fuel key reactions. At the same time, some viruses have evolved to hijack the very gateways that bring these nutrients into cells. This study reveals, in atomic detail, how one such gateway in mice, called CAT1, both ferries positively charged amino acids across the cell membrane and doubles as the entry dock for a leukemia-causing virus. Understanding this dual role helps explain how cells sense nutrients, how some cancers thrive, and why specific viruses infect some species but not others.
The Protein Gatekeeper at the Cell Surface
CAT1 sits in the cell’s outer membrane and specializes in importing three positively charged amino acids: arginine, lysine, and ornithine. These molecules are vital for shaping proteins, removing toxic ammonia through the urea cycle, switching on growth pathways, and generating signaling molecules such as nitric oxide. When CAT1 does not work properly, mice die shortly after birth, and in humans the related transporter is linked to cancers of the ovary and colon. Intriguingly, the same protein also serves as the main receptor for a mouse retrovirus known as Friend murine leukemia virus, which uses CAT1 to recognize and infect rodent cells.
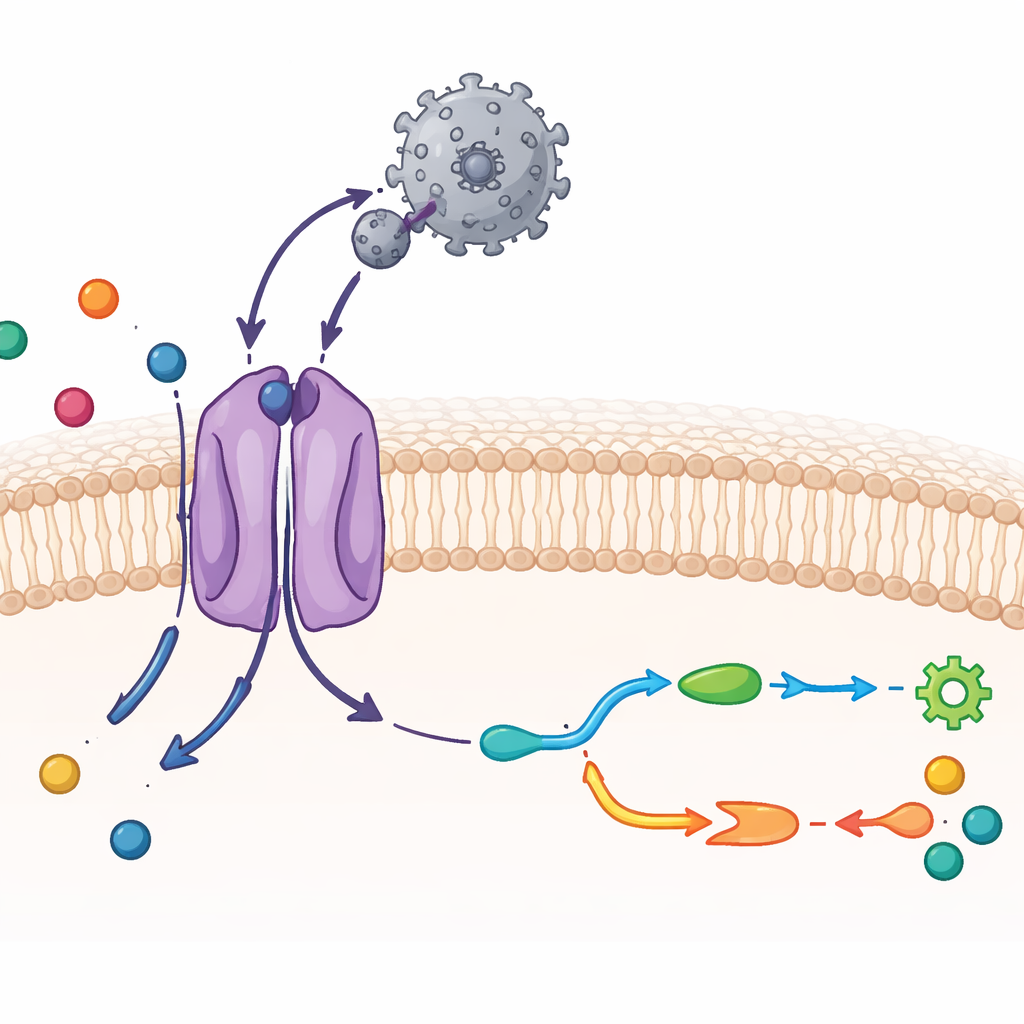
Seeing CAT1 and a Virus in Atomic Detail
The researchers used cryo–electron microscopy, a technique that images frozen molecules at near-atomic resolution, together with biochemical tests. They purified mouse CAT1 and mixed it with the virus’s outer binding region, a compact protein fragment that latches onto the transporter. The resulting structure shows a one-to-one complex: a single viral binding domain attached to a single CAT1 molecule in the membrane. Most of the transporter’s 14 membrane-spanning segments could be visualized, including two extra segments that are unique to this family. The viral fragment hugs an exposed loop on the outer side of CAT1 and forms a dense web of hydrogen bonds, accounting for its extraordinarily tight grip. Yet the viral protein itself hardly changes shape, which helps maintain this strong interaction with little energetic cost.
How CAT1 Picks and Moves Its Cargo
Inside CAT1, the team identified the precise pocket where arginine, lysine, and ornithine bind. The backbone of each amino acid is held in place by a ring of protein atoms that form multiple hydrogen bonds, while side chains are nestled into a cavity lined with carefully positioned side groups and a single critical negatively charged residue. When this acidic residue or nearby contact points were altered by mutation, CAT1 lost its ability to stabilize when amino acids were added, revealing their importance for recognition. Comparing structures with arginine, lysine, and ornithine bound showed that CAT1 grips all three in almost the same way, adjusting only slightly to accommodate differences in side-chain length. These details explain why CAT1 strongly prefers positively charged amino acids and largely excludes neutral ones.
A Moving Gate That Still Holds the Virus
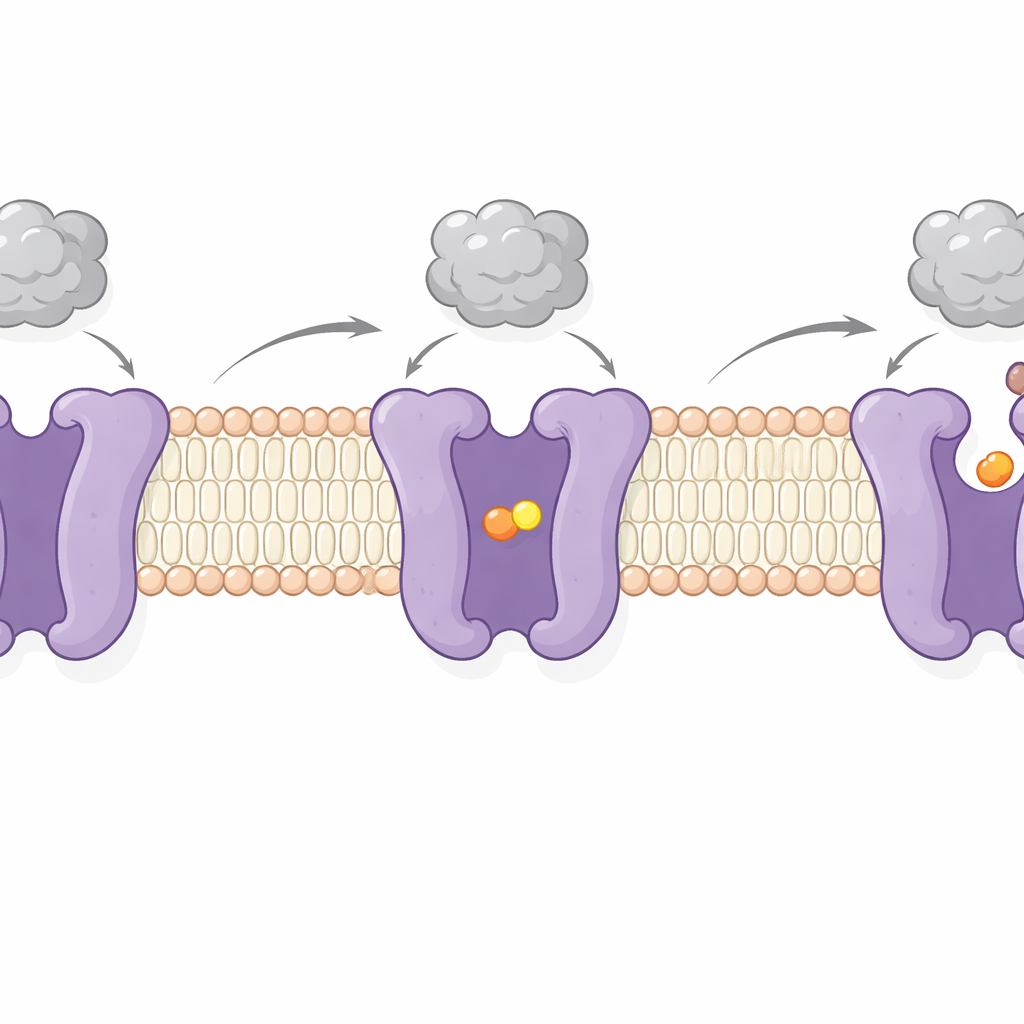
By imaging CAT1 with and without bound amino acid, the authors captured two key steps of its working cycle. In one, the cargo site is sealed off from both sides of the membrane—an “occluded” state in which the amino acid is locked inside. In the other, the site opens toward the cell interior, allowing cargo release. These changes involve swinging motions of several membrane-spanning segments and a flexible “latch” peptide on the inner surface that appears to control opening toward the cell. Notably, the virus-binding region on the outer side moves only slightly and stays firmly attached during these shifts. This supports earlier evidence that viral docking and amino acid transport are largely independent—CAT1 can keep feeding the cell even while serving as a viral receptor.
Why This Dual Role Matters
By mapping exactly how CAT1 recognizes both nutrients and a virus, this work clarifies why the Friend leukemia virus infects rodents but not humans: the virus engages a short outer loop of CAT1 that differs markedly between species. Swapping this segment from mouse into human CAT1 is enough to restore viral binding in cell tests. The structures also hint at how CAT1 helps cells sense arginine levels and relay that information to growth-control pathways, and they point to specific sites where drugs or chemical modifiers could tune transport or block viral entry. In essence, the study shows how a single membrane protein can act as both a nutrient valve and a viral handle, with different faces of the same molecule serving life-sustaining and disease-promoting roles.
Citation: Ye, M., Liang, Z., Zhou, D. et al. Amino acid and viral binding by the high-affinity Cationic Amino acid Transporter 1 (CAT1) from Mus musculus. Nat Commun 17, 2829 (2026). https://doi.org/10.1038/s41467-026-69421-0
Keywords: amino acid transport, membrane protein structure, viral receptors, arginine metabolism, host tropism