Clear Sky Science · en
Crosstalk between EZH2 and DNA methylation mediates neuroendocrine prostate cancer lineage plasticity
Why cancer cell identity matters
Prostate tumors do not always stay the same over time. Some initially rely on male hormones and respond to standard treatments, but later shift into a far more aggressive form called neuroendocrine prostate cancer. This shape‑shifting ability, known as lineage plasticity, helps tumors escape therapy and is a major cause of treatment failure. The study summarized here explores how two chemical "off switches" on DNA and its packaging work together to control this dangerous identity change, and how blocking one switch can cause the other to compensate.
Two chemical brakes on the genome
Every cell carries the same genes, but chemical tags decide which ones are on or off. The authors focus on two types of repressive tags. One is DNA methylation, in which small chemical groups are added directly onto DNA and usually silence nearby genes; this is maintained by an enzyme called DNMT1. The other is a tag called H3K27me3 that sits on DNA‑wrapping proteins and is placed there by a protein named EZH2, part of the polycomb repressive complex 2. Both systems are known to change in prostate cancer, especially when tumors become neuroendocrine, but how they influence each other and together steer cell identity had not been clearly mapped.
Patterns that mark aggressive prostate tumors
Using genetically engineered mice, patient tumor samples, and miniature tumors grown in the lab, the researchers compared typical castration‑resistant prostate adenocarcinoma with neuroendocrine prostate cancer. They found consistent shifts in DNA methylation between the two states: hundreds of genomic regions lost methylation, while others gained it. Strikingly, regions that became less methylated in neuroendocrine tumors often accumulated more of the EZH2‑linked mark H3K27me3, even though these two repressive systems are usually thought to avoid each other. Many of these regions sat near genes involved in nerve‑like features and in so‑called bivalent promoters, which keep developmental genes poised for rapid switching on or off.
What happens when one brake is released
To probe cause and effect, the team disabled EZH2 in mouse models that are prone to evolve into neuroendocrine tumors, and separately treated patient‑derived organoids with an EZH2‑blocking drug. In both settings, the global DNA methylation landscape was extensively rewired. Regions that were previously targeted by EZH2 often became more methylated, while other stretches lost methylation and turned on genes linked to neural differentiation and known neuroendocrine signatures. In other words, removing the H3K27me3‑based brake allowed the DNA methylation system to move in and keep many of the same genes silenced, while simultaneously loosening control at other sites that reinforce the neuroendocrine state.
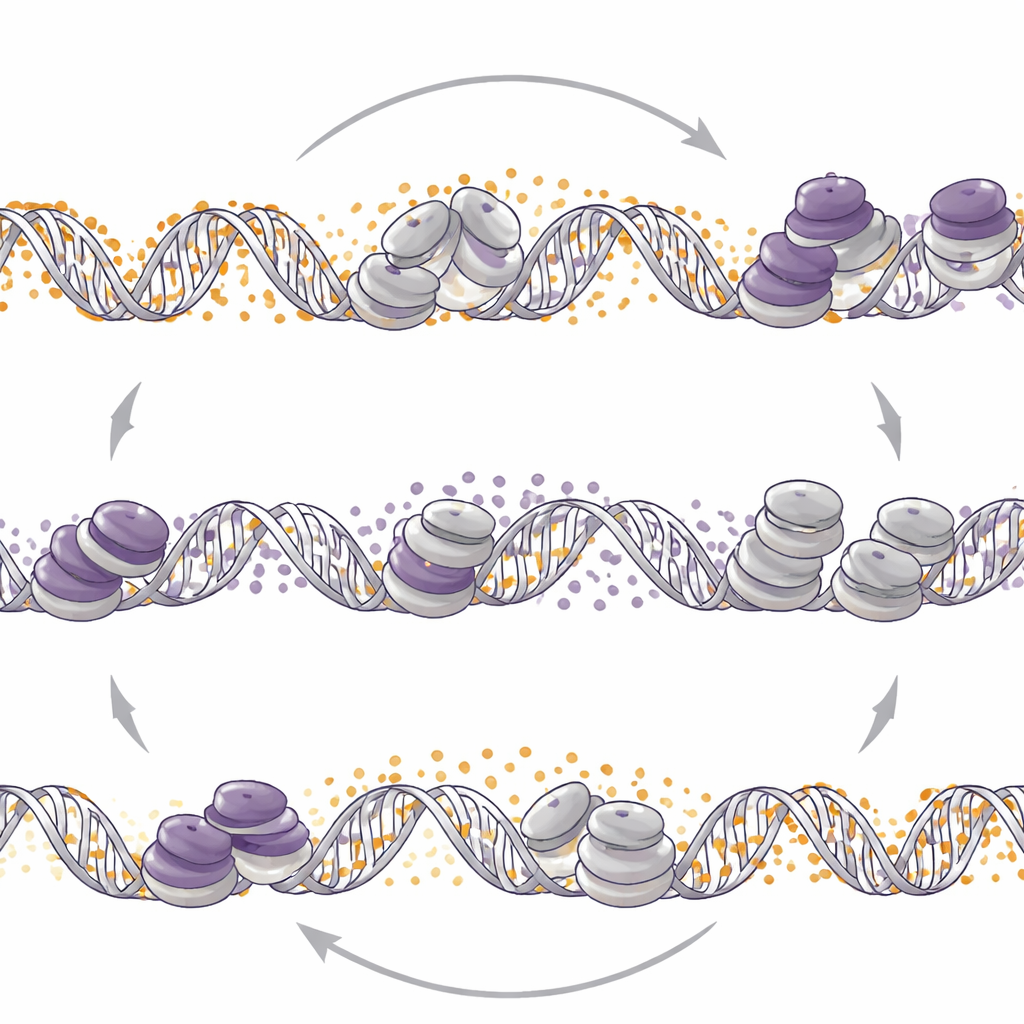
Flipping the other switch reshapes control
The scientists then asked whether the reverse was also true: does altering DNA methylation reposition EZH2 marks? When they deleted DNMT1 or used the DNA methylation‑lowering drug decitabine in neuroendocrine models, they saw broad redistribution of H3K27me3 across the genome. In these tumors, reducing DNA methylation led to more H3K27me3 at certain poised promoters, loss of an activating histone tag, and reduced activity of neuroendocrine genes. In contrast, in a prostate adenocarcinoma cell line, DNMT1 loss decreased H3K27me3 at similar poised regions and increased the activating tag, unleashing neuroendocrine‑related genes. Thus the same molecular nudge—weakening DNMT1—can either dampen or ignite neuroendocrine programs depending on the tumor context.
What this means for future treatments
Together, these experiments reveal that DNA methylation and EZH2‑driven histone marking behave like two coordinated brakes that can trade places to keep critical genes silent or poised, helping prostate tumors switch identity and resist therapy. Interfering with one system alone often triggers compensatory changes in the other, which may limit the lasting benefit of drugs that target EZH2 or DNA methylation by themselves. The work suggests that carefully designed strategies that account for this molecular tug‑of‑war—potentially including combination or timed use of both types of epigenetic drugs—could one day offer better ways to prevent or treat the most aggressive, therapy‑resistant forms of prostate cancer.
Citation: Singh, R., Venkadakrishnan, V.B., Imada, E. et al. Crosstalk between EZH2 and DNA methylation mediates neuroendocrine prostate cancer lineage plasticity. Nat Commun 17, 2992 (2026). https://doi.org/10.1038/s41467-026-69308-0
Keywords: prostate cancer, neuroendocrine tumors, epigenetics, DNA methylation, EZH2