Clear Sky Science · en
Quantitative analysis of small RNA pseudouridylation reveals interplay of PUS enzymes in tRNA anticodon stem-loop
Why hidden marks on RNA matter
Every cell constantly reads genetic instructions using small RNA molecules that help build proteins and process other RNAs. This study looks at a subtle chemical tweak on these RNAs, called pseudouridine, that can change how they behave without altering the underlying genetic code. By mapping exactly where these tweaks occur and which enzymes place them, the authors reveal an unexpected "conversation" among enzymes that fine‑tune a crucial part of the protein‑making machinery.
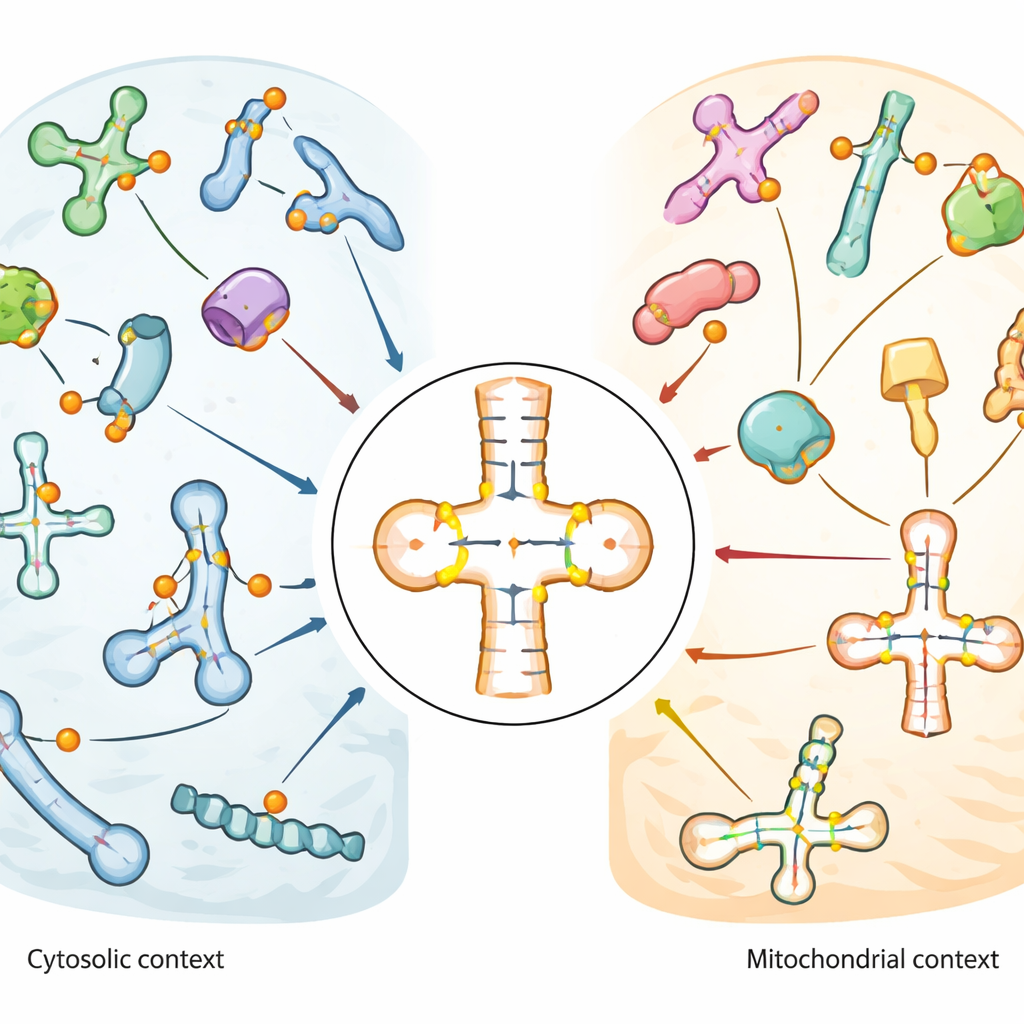
Chemical edits on the cell’s workhorse RNAs
Cells rely on many kinds of small RNAs, especially transfer RNAs (tRNAs), which act as adaptors that read three‑letter words in the genetic code and deliver the right amino acid during protein synthesis. These tRNAs, along with other small RNAs that guide RNA processing, are densely decorated with chemical modifications. Pseudouridine is one of the most common. It differs from ordinary uridine only in how its atoms are connected, but that small change strengthens base pairing and can alter RNA shape and stability. Because pseudouridine is linked to stress responses, brain function, and cancer, scientists want to know exactly where it appears and which enzymes, called pseudouridine synthases, or PUS enzymes, are responsible.
Building a precise map of pseudouridine
Traditional methods could flag many pseudouridine sites but struggled to measure how extensively each site was modified. The researchers refined a newer chemical‑sequencing method they previously developed, called PRAISE, to do this quantitatively on small RNAs. They first treated RNA to remove other blocking modifications, then used a specialized bisulfite reaction and sequencing strategy that turns the presence of pseudouridine into a characteristic deletion signal in the readout. Applying this to human HEK293T cells, they generated a high‑resolution landscape of pseudouridine across cytosolic and mitochondrial tRNAs, as well as small nuclear RNAs (snRNAs) and small nucleolar RNAs (snoRNAs). The method proved highly reproducible and matched or exceeded existing techniques, while adding precise measurements of modification levels at each position.
Distinct patterns in different cellular neighborhoods
The map revealed that pseudouridine is especially enriched in the anticodon stem‑loop of tRNAs, the region that directly pairs with messenger RNA during translation. Some positions, such as a universally present site near the tRNA’s base (known as position 55), were almost fully modified across many tRNAs. Others showed moderate or variable levels, suggesting regulatory flexibility. Mitochondrial tRNAs, which operate inside the cell’s energy‑producing organelles, displayed a markedly different pattern: key sites common in regular (cytosolic) tRNAs were missing or only weakly modified, while other positions near the anticodon were strongly and consistently marked. This contrast implies that mitochondria use a distinct set of editing rules for their own tRNAs. In snRNAs and snoRNAs, pseudouridine clustered in functional regions that help guide other RNA modifications or splicing, and some sites changed in response to heat shock or oxidative stress.
Who writes the marks: assigning enzymes to sites
To link specific enzymes to specific RNA sites, the team compared wild‑type cells with a panel of cell lines lacking individual PUS enzymes or with reduced levels of a guide enzyme called DKC1. They showed that snoRNA marks come from two routes: an RNA‑guided pathway using DKC1 and a stand‑alone enzyme PUS7 that writes a single distinctive site. In tRNAs, they systematically assigned many sites to particular enzymes. PUS1 emerged as a major writer of marks near the anticodon in both cytosolic and mitochondrial tRNAs. Members of the RluA family split their duties: RPUSD1 modified certain sites in cytosolic tRNAs, including a previously unrecognized site near the tRNA tail, while RPUSD2 targeted nearby positions in mitochondrial tRNAs, and RPUSD3 showed no detectable tRNA activity. Related enzymes PUS1L and PUS7L turned out to specialize in distinct regions or tRNA types, indicating that closely related proteins can evolve surprisingly different tasks.
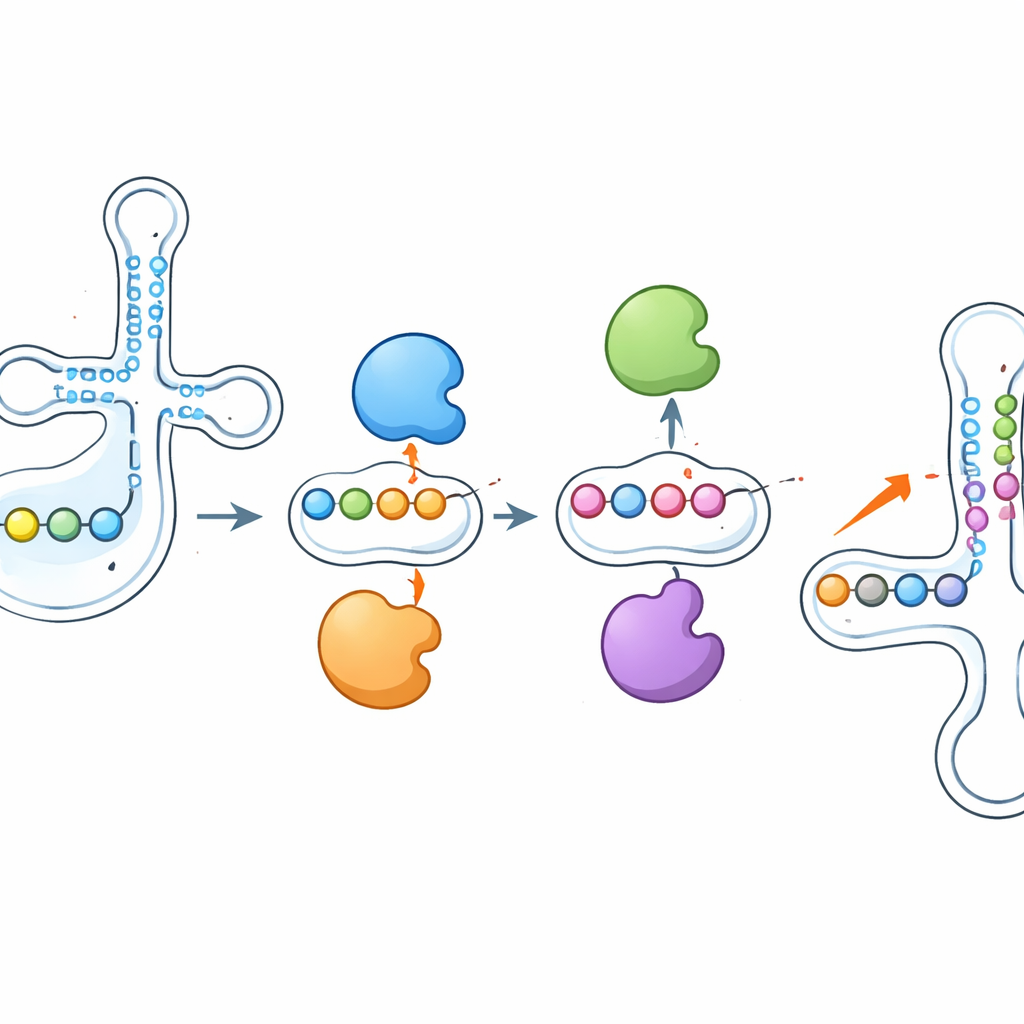
Enzymes that push and pull on each other’s work
One of the most striking findings was that these enzymes do not act in isolation. When the researchers deleted PUS1, the pseudouridine marks it normally installs near the anticodon dropped sharply—but neighboring sites, written by other enzymes like RPUSD1 or PUS7, became more heavily modified on the same tRNA. Knocking out RPUSD1 produced the mirror image at overlapping positions, and similar see‑saw patterns appeared between PUS7 and another enzyme, PUS3. These changes were not explained by shifts in enzyme or tRNA abundance, pointing to a direct interplay at the level of individual tRNA molecules. The authors suggest that enzymes may physically compete for nearby binding sites or that adding or removing a pseudouridine reshapes the local RNA structure, making neighboring positions more or less accessible to other enzymes.
What this means for health and disease
To a non‑specialist, the key message is that the chemical marks on tRNAs are not fixed decorations but part of a dynamic, interconnected system. Multiple enzymes collaborate and compete to pattern the anticodon region, which is central to reading the genetic code accurately and flexibly. Because mutations in several of these enzymes cause human disease—from blood disorders and muscle weakness to brain dysfunction and cancer—understanding how they share and adjust their targets may illuminate why losing one enzyme sometimes has milder effects than expected, or how cells compensate under stress. This study provides a detailed map of where pseudouridine appears on small RNAs and shows that the enzymes writing these marks can modulate one another’s activity, offering new entry points for exploring and eventually correcting RNA‑based defects in human health.
Citation: Liu, W., Ma, Y., Wang, L. et al. Quantitative analysis of small RNA pseudouridylation reveals interplay of PUS enzymes in tRNA anticodon stem-loop. Nat Commun 17, 2811 (2026). https://doi.org/10.1038/s41467-026-69177-7
Keywords: tRNA modifications, pseudouridine, RNA enzymes, mitochondrial function, translation regulation