Clear Sky Science · en
Structural basis for activation and conformational plasticity of the GluA4 AMPA receptor
Why this brain channel matters
Every thought, memory, and movement in your body depends on tiny electrical signals that pass between nerve cells. Many of these signals are carried by AMPA receptors, molecular “gates” that open when they sense the chemical messenger glutamate. This study focuses on a rare version of that gate, called GluA4, which is concentrated in fast‑firing brain cells and in the cerebellum. Because changes in GluA4 have been linked to epilepsy, intellectual disability, and even brain tumors, understanding how this receptor works at the atomic level could reveal why it is so powerful—and how it might be targeted by future medicines.
The brain’s fast switches
AMPA receptors are protein complexes that sit in the membrane of nerve cells and convert a chemical signal into an electrical current by letting charged particles flow in. Each receptor is built from four related parts (subunits), and the mix of these subunits shapes how quickly the channel opens, how much current it passes, and whether calcium can enter. GluA4 is a calcium‑permeable subunit that is unusually abundant in specific cell types, such as parvalbumin‑positive inhibitory neurons and certain auditory and cerebellar neurons. Genetic studies show that damaging changes in the gene for GluA4 can cause seizures and cognitive problems, hinting that its special properties are crucial for brain function yet have remained structurally mysterious.
Capturing GluA4 in the act
To reveal how GluA4 operates, the researchers used cryo‑electron microscopy, a technique that images flash‑frozen molecules at near‑atomic resolution. They purified receptors made entirely of GluA4 and tightly linked to an auxiliary partner protein called TARPγ2, which naturally associates with GluA4 in the brain and helps fine‑tune its behavior. The team locked the receptors in an active, non‑desensitized state using glutamate together with a drug that prevents the channel from shutting off. Electrical recordings from artificial membranes confirmed that these purified complexes behaved like real AMPA receptors, producing brief and long openings with multiple levels of current.
A flexible yet intact architecture
The structural maps revealed that GluA4 receptors keep the classical Y‑shaped layout seen in other AMPA receptors: a top “head” region outside the cell that senses glutamate, and a bottom core that forms the ion channel across the membrane. The uppermost layer, called the amino‑terminal domain, turned out to be surprisingly mobile, adopting many different positions while still maintaining key contacts that hold the four subunits together. This shows that GluA4’s outer scaffold is intact but highly flexible, potentially allowing it to interact with diverse partner proteins in different brain regions without falling apart.
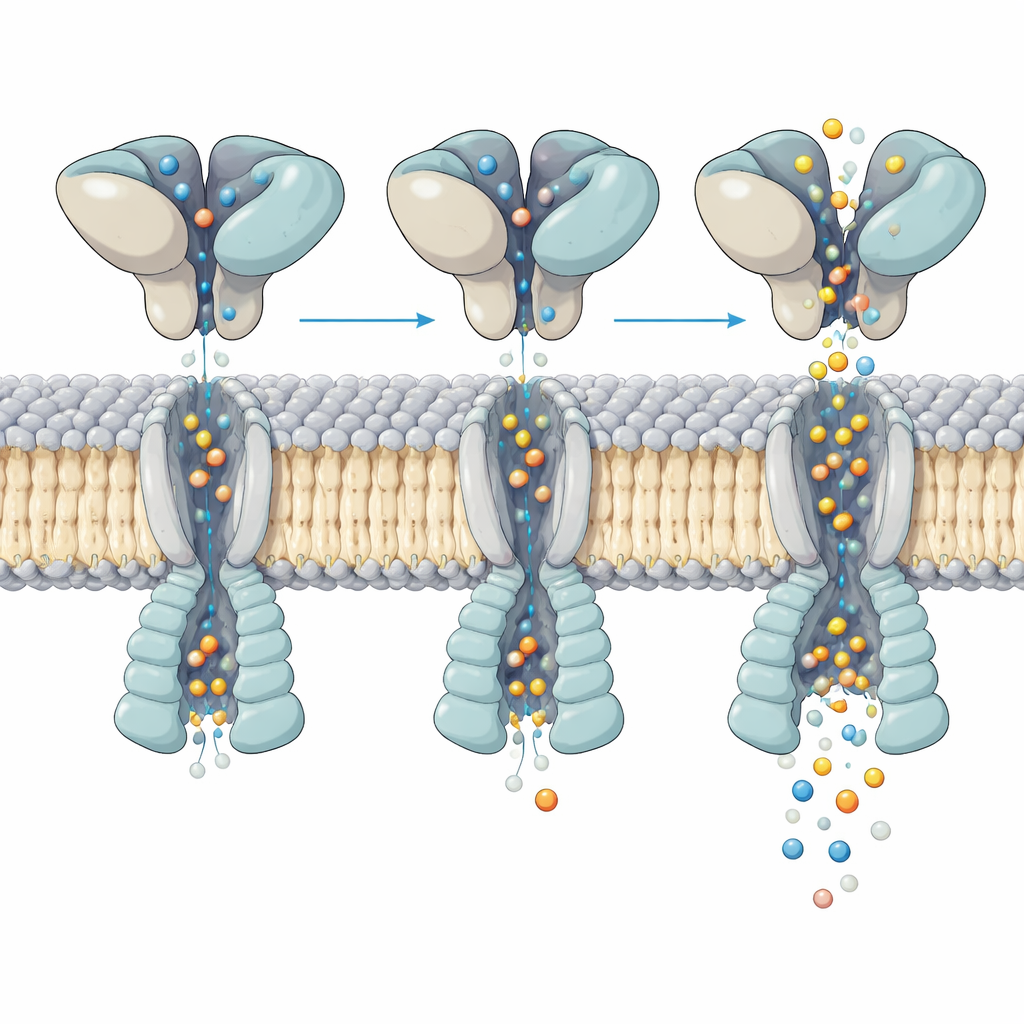
How shape‑shifting controls the flow of ions
Closer inspection of the active channel exposed how motion in the glutamate‑binding layer above is translated into opening of the pore below. All four binding pockets were filled with glutamate, yet the helices that form the gate bent in an uneven, asymmetric way across the four subunits. By analyzing hundreds of thousands of particles, the team resolved five distinct “substates” of the same activated receptor. In each substate, the upper domains rock and twist slightly relative to the membrane core, and this small rearrangement shifts where and how strongly key helices kink within the pore. These subtle differences change the pore’s width at critical points, matching the multiple conductance levels seen in single‑channel recordings, even though glutamate occupancy is the same.
Linking structure to disease‑related variants
The study identifies nearly every amino acid in a short, highly conserved stretch of the pore‑forming helix as capable of serving as a bending hinge. Several of these positions are exactly where disease‑causing mutations in human GluA4 have been found in patients with epilepsy and intellectual disability. The authors propose that these variants likely alter how easily the helix can kink, shifting the balance among sublevels of channel opening or changing calcium access to a nearby binding site. In effect, tiny changes in this hinge zone could over‑ or under‑tune the electrical signal carried by GluA4‑containing receptors, with major consequences for neural circuits that rely on extremely fast, precise firing.
What this means for brain health
By showing that GluA4 is both structurally stable and unusually shape‑flexible, this work reframes how scientists think about AMPA receptor function. Instead of viewing different current levels as simply the result of how many glutamate molecules are bound, the findings support a picture in which a fully loaded receptor can still sample multiple conformations, each with a distinct pore opening. This “conformational plasticity” provides a structural explanation for the graded responses of GluA4 channels and suggests new ways drugs or partner proteins might tune their activity. In the long run, mapping these microscopic shape changes may guide therapies for disorders where GluA4 is a key player—from certain epilepsies to circuit changes in psychiatric disease and cancer‑related brain signaling.
Citation: Hale, W.D., Wang, H., Huganir, R.L. et al. Structural basis for activation and conformational plasticity of the GluA4 AMPA receptor. Nat Commun 17, 2932 (2026). https://doi.org/10.1038/s41467-026-68953-9
Keywords: AMPA receptor, GluA4, synaptic transmission, cryo electron microscopy, ion channel gating