Clear Sky Science · en
Perivascular mesenchymal cells instruct ST2+ reparative macrophages to promote endovascular injury-induced neointimal hyperplasia in mice
Why fixing damaged arteries can backfire
When doctors open clogged heart arteries with tiny metal stents, they save lives by restoring blood flow. But in many patients, the treated vessel slowly narrows again as cells in the vessel wall grow back too vigorously. This study in mice asks a key question behind that problem: which cells inside the artery decide whether healing stays smooth, or tips into harmful overgrowth that can clog the vessel all over again?
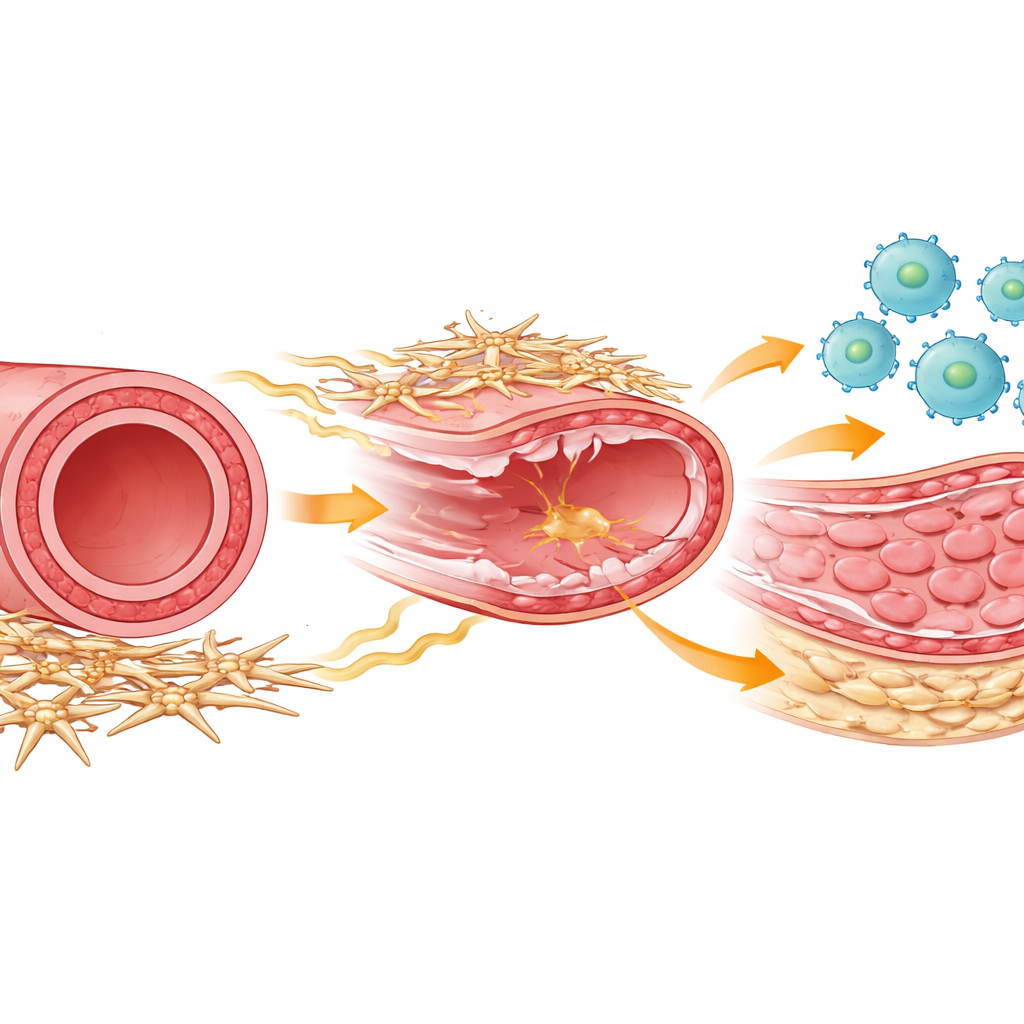
The players in artery healing
Inside every artery, several kinds of cells work together to repair damage. Immune cells called macrophages rush in after an injury to clear debris and coordinate healing. Smooth muscle cells form the muscular middle layer that can thicken and narrow the vessel if they start multiplying too much. Wrapping around the outside of the artery is a lesser-known group of support cells, often called mesenchymal or stromal cells, that help maintain the vessel’s structure and talk chemically to nearby cells. The authors used a mouse model that mimics the scraping injury caused when a stent is placed, then tracked how all these cell types behaved over time using powerful single-cell gene-reading methods and detailed cell profiling.
From first responders to repair crew
Right after the arterial injury, immune cells flooded into the vessel wall, peaking within a couple of days. At this early stage, most macrophages showed a classic “inflammatory” profile suited for fighting danger signals. But by about a week, a different macrophage personality dominated: a reparative type tuned toward rebuilding tissue and encouraging smooth muscle growth. The researchers were able to separate these groups using surface markers and timing, and showed that both inflammatory and reparative macrophages largely came from circulating monocytes recruited from the blood. When they experimentally removed macrophages during the later, reparative phase, smooth muscle cell division dropped sharply and the abnormal inner layer, or neointima, failed to form, proving that these late-arriving repair-oriented macrophages are necessary for the thickening that can re-clog arteries.
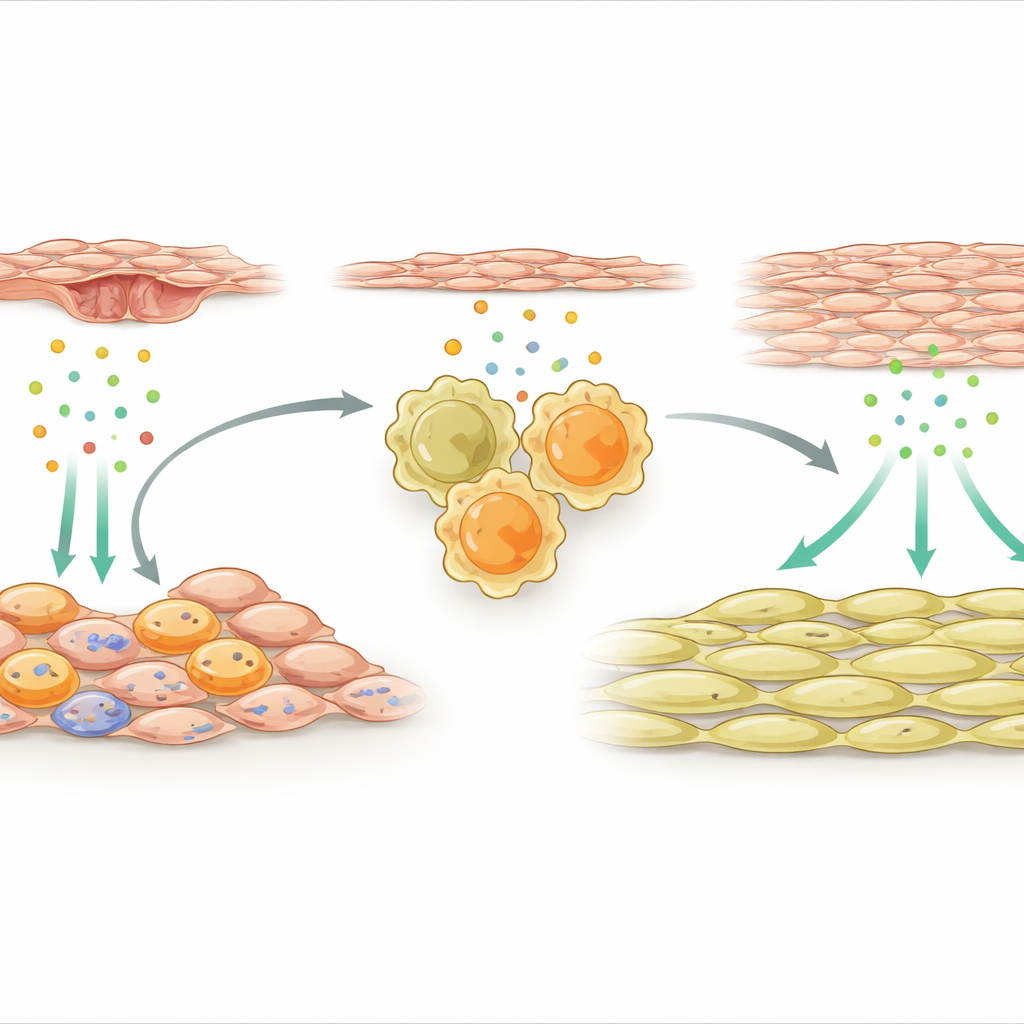
Support cells that switch on repair macrophages
The team then turned to the outer support cells that wrap the artery. After injury, a specialized subset of these mesenchymal cells became highly active and started expressing many immune-related genes. One signal stood out: IL-33, a distress molecule that is released by damaged or stressed tissues. These IL-33–producing support cells clustered near macrophages in the vessel wall. The researchers showed that factors released from injured endothelial cells, the inner lining of the artery, activated a molecular switch called NFκB in mesenchymal cells, which in turn boosted IL-33 production. In both mice and human patients with stent-related re-narrowing, levels of IL-33 were elevated, suggesting that this pathway is clinically relevant.
A chemical chain reaction to vessel thickening
IL-33 works by binding to a receptor known as ST2 on immune cells. The study found that after arterial injury, many of the reparative macrophages in the wall carried this receptor. When IL-33 from outer support cells stimulated ST2-positive macrophages, it pushed them into a strongly pro-repair mode and caused them to release another protein, osteopontin. Osteopontin then acted directly on smooth muscle cells, driving them to proliferate and pile up on the inside of the artery, creating a thickened layer that narrows the blood-flow channel. If the researchers removed IL-33 specifically from mesenchymal cells, or removed ST2 from macrophages, or blocked osteopontin with gene-silencing molecules packed into a local hydrogel, smooth muscle growth was curtailed and the artery stayed more open.
New ideas for keeping stents open
For non-specialists, the central message is that harmful re-narrowing of arteries after stenting is not just a problem of smooth muscle cells misbehaving on their own. Instead, it is orchestrated by a conversation between outer vessel support cells and a particular subset of “repair” macrophages. Injury triggers support cells to release IL-33, which activates ST2-positive macrophages, which then secrete osteopontin to overstimulate smooth muscle growth. Interrupting this IL-33–ST2–osteopontin chain locally at the injury site prevented excessive thickening in mice. That suggests a future in which stents might be paired with smart gels or coatings that fine-tune healing rather than bluntly suppressing it, helping arteries repair themselves without closing up again.
Citation: Ping, Y., Qin, Z., Huang, X. et al. Perivascular mesenchymal cells instruct ST2+ reparative macrophages to promote endovascular injury-induced neointimal hyperplasia in mice. Nat Commun 17, 3635 (2026). https://doi.org/10.1038/s41467-026-68587-x
Keywords: vascular repair, macrophages, IL-33 signaling, restenosis, smooth muscle cells