Clear Sky Science · en
Transient protein structure guides surface diffusion pathways for electron transport in membrane supercomplexes
How Cells Keep Their Power Lines Safe
Every second, the cells in your body move billions of electrons to make the energy molecule ATP. If even a small fraction of those electrons leak away, they can form dangerous reactive chemicals that damage DNA, proteins, and fats. This study digs into how tiny “power line” proteins on the inner membrane of mitochondria guide these electrons safely and efficiently from one station to another, revealing a surprising role for flexible, seemingly disordered protein pieces and special membrane fats.
The Busy Power Grid Inside Mitochondria
Inside mitochondria, energy production runs through a chain of large protein machines embedded in a membrane. Two key stations, known as Complex III and Complex IV, hand off electrons using a small carrier protein called cytochrome c. Traditionally, scientists pictured cytochrome c drifting in three dimensions through the watery interior until it bumps into each complex. But many organisms also arrange these complexes into clusters called supercomplexes, raising the puzzle of whether building such clusters actually improves how efficiently electrons are shuttled, or merely keeps the machinery organized and reduces accidental leaks.
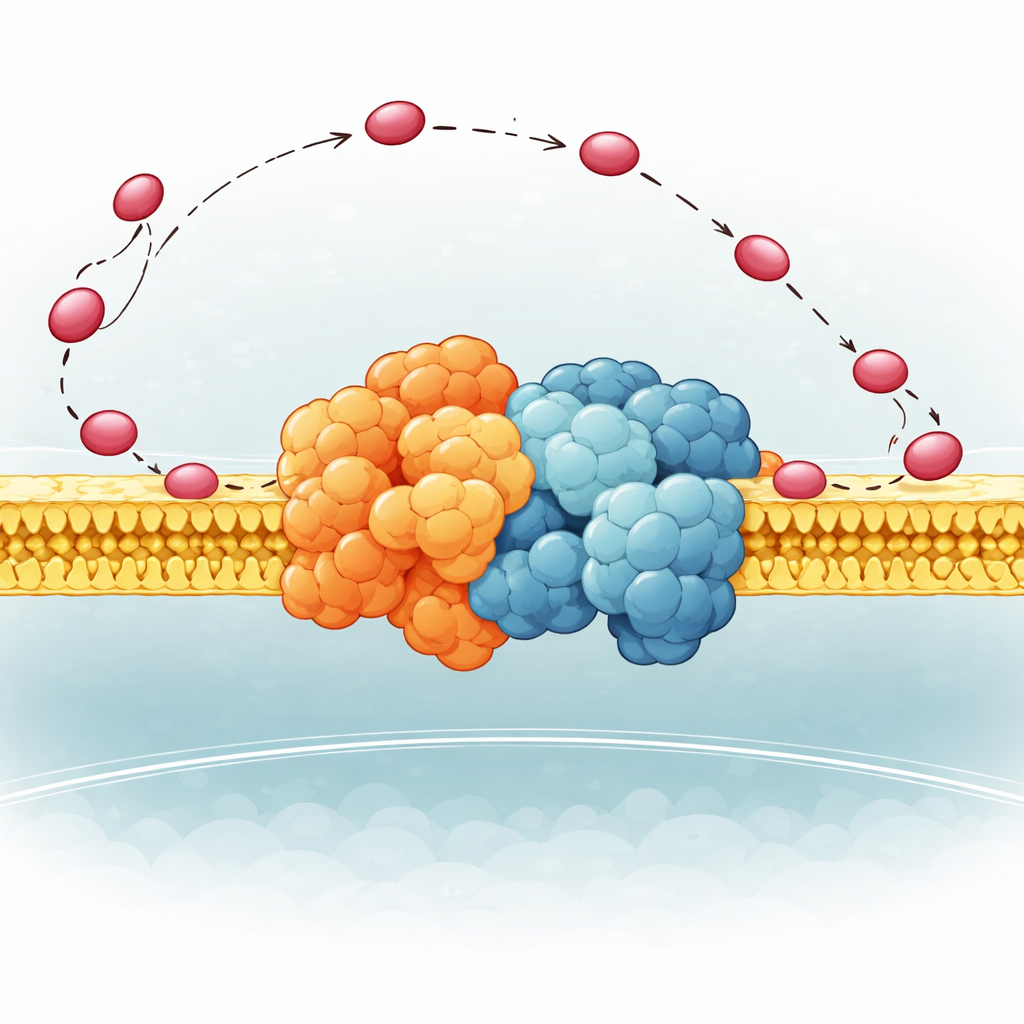
A Flexible Hinge That Steers the Carrier
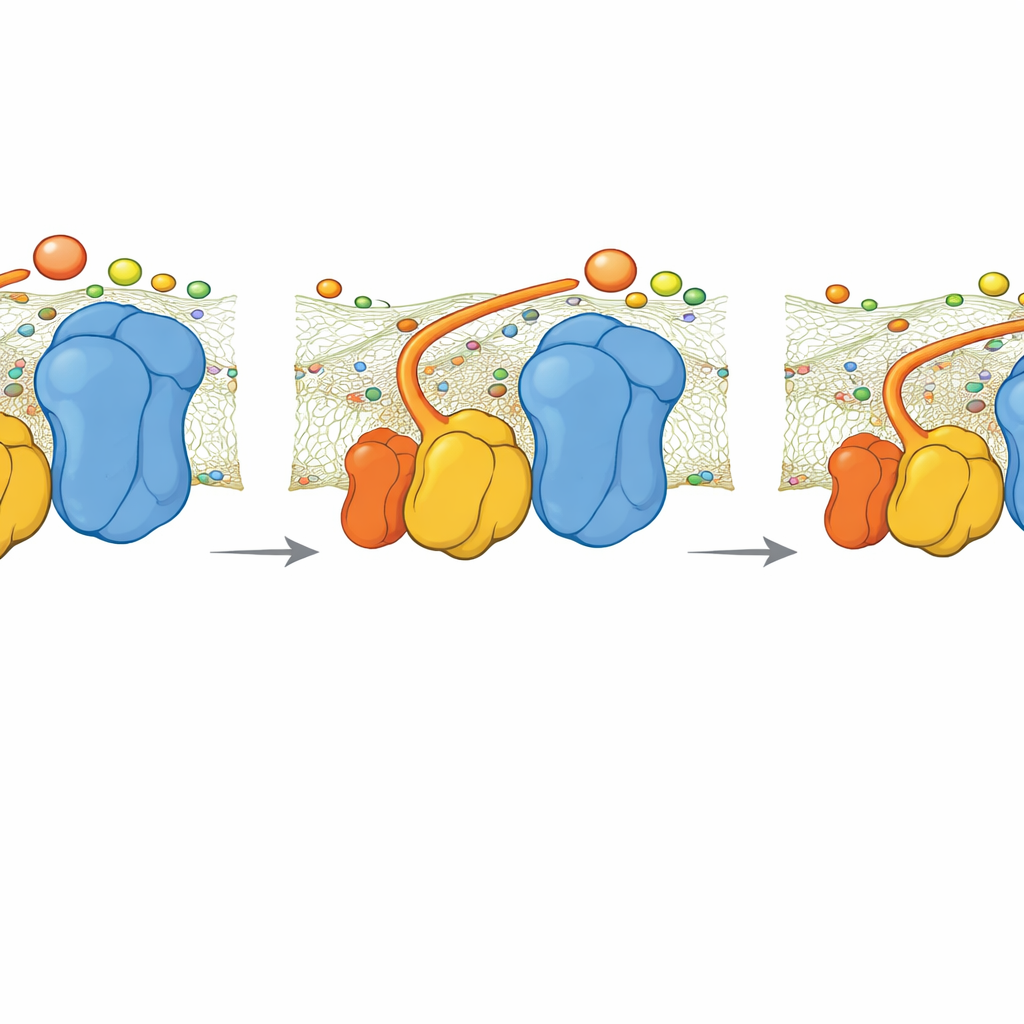
The authors focused on a small part of Complex III in yeast called QCR6, which has a long, negatively charged “hinge” segment whose structure had been too floppy to capture clearly before. Using a combination of advanced computer methods—protein folding algorithms, molecular dynamics, and cryo-electron microscopy—they generated an ensemble of realistic shapes this hinge can take while attached to a full Complex III–Complex IV supercomplex in a membrane. They found that QCR6 does not sit still: when cytochrome c comes near, parts of the hinge fold into temporary helices and grab the carrier; as cytochrome c moves from Complex III toward Complex IV, the hinge can stretch and partially unfold, following and guiding it along the membrane surface instead of letting it wander off into the surrounding fluid.
Membrane Fats as Invisible Guides
The membrane itself also plays an active role. The team replaced generic lipids in their models with cardiolipin, a negatively charged fat known to be important for healthy mitochondria and often altered in heart disease and neurodegeneration. Simulations showed that positively charged spots on QCR6 latch onto cardiolipin headgroups, effectively tethering the hinge to the membrane while it escorts cytochrome c. This coupling makes it energetically cheaper for the carrier to slide from Complex III to Complex IV along the surface, rather than hopping randomly in three dimensions. When cardiolipin was removed, the energetic cost of this journey rose sharply, more cytochrome c drifted into the bulk solution, and its position became less clearly biased toward the electron-accepting site.
From Random Wandering to Guided Surfing
By running large-scale Brownian dynamics simulations, the researchers tracked where cytochrome c tends to linger around the supercomplex. With an intact QCR6 hinge and anionic lipids present, most cytochrome c molecules remain close to the membrane and spend more time near productive contact sites on Complex III and Complex IV. As the hinge extends, the preferred cloud of cytochrome c positions shifts from the donor side to the acceptor side, effectively “surfing” along the complex surface. This guided surface diffusion improves how quickly electrons can be handed off without turning cytochrome c into a permanently attached part of the machinery; carriers can still exchange freely with the surrounding pool.
What Happens When the Hinge Is Missing
The team also studied a mutant yeast strain lacking QCR6. Cryo-EM imaging showed that the supercomplex rearranges into a more compact, slightly different shape that partly restores a continuous negative charge across the surface. Nonetheless, simulations revealed that without the QCR6 hinge the rate at which cytochrome c moves electrons from Complex III to Complex IV drops. A simple kinetic model, tying electron flow to ATP output, suggests that under high energy demand this loss of guidance could cut ATP production by roughly 30 percent. At lower demand, the difference is smaller, because slower, less organized diffusion still suffices.
Why This Matters for Health and Evolution
Put together, the work supports a new picture: instead of acting as static scaffolds, flexible protein segments and charged lipids cooperate to confine electron carriers to the membrane surface and bias their motion along safe, productive routes. In yeast, an unusually long QCR6 hinge and abundant cardiolipin generate this “refolding-guided diffusion” that limits electron leakage and boosts energy output. In other organisms, shorter hinges, different supercomplex designs, or even covalent tethers appear to serve a similar goal—keeping carriers close to where they are needed. Understanding these subtle steering mechanisms may help explain why defects in cardiolipin or supercomplex organization show up in aging, heart failure, and neurodegenerative disease, and could eventually inform strategies to fine-tune cellular energy metabolism.
Citation: Chan, C.K., Nguyen, J., Hryc, C.F. et al. Transient protein structure guides surface diffusion pathways for electron transport in membrane supercomplexes. Nat Commun 17, 2892 (2026). https://doi.org/10.1038/s41467-025-67110-y
Keywords: mitochondrial supercomplexes, cytochrome c diffusion, cardiolipin, electron transport chain, QCR6 hinge