Clear Sky Science · en
NRIP1 co-activates nuclear translocated FOXO3 to upregulate TFAM expression and promote radioresistance in non-small cell lung cancer
Why making lung cancer treatments work better matters
Radiotherapy is a mainstay for treating non-small cell lung cancer, the most common type of lung cancer. Yet many tumors gradually learn to survive radiation, allowing the disease to return or spread. This study asks a simple but crucial question: what is happening inside cancer cells that lets some of them shrug off radiation, and can those inner workings be turned against the tumor to make treatment more effective?
How cancer cells dodge the damage from radiation
Radiation kills cancer cells largely by generating bursts of harmful molecules called reactive oxygen species and by breaking DNA. These attacks are closely tied to the cell’s power stations, the mitochondria. The researchers noticed that radioresistant lung cancer cells not only survive better after radiation but also have more mitochondrial DNA and higher levels of a key mitochondrial protein called TFAM, which helps copy and protect mitochondrial DNA. When these resistant cells were compared with their original, non-resistant counterparts, they showed less oxidative damage, less programmed cell death, and clearly higher TFAM levels, pointing to TFAM as a central shield that helps cancer cells endure treatment.
Switches inside the cell nucleus that strengthen this shield
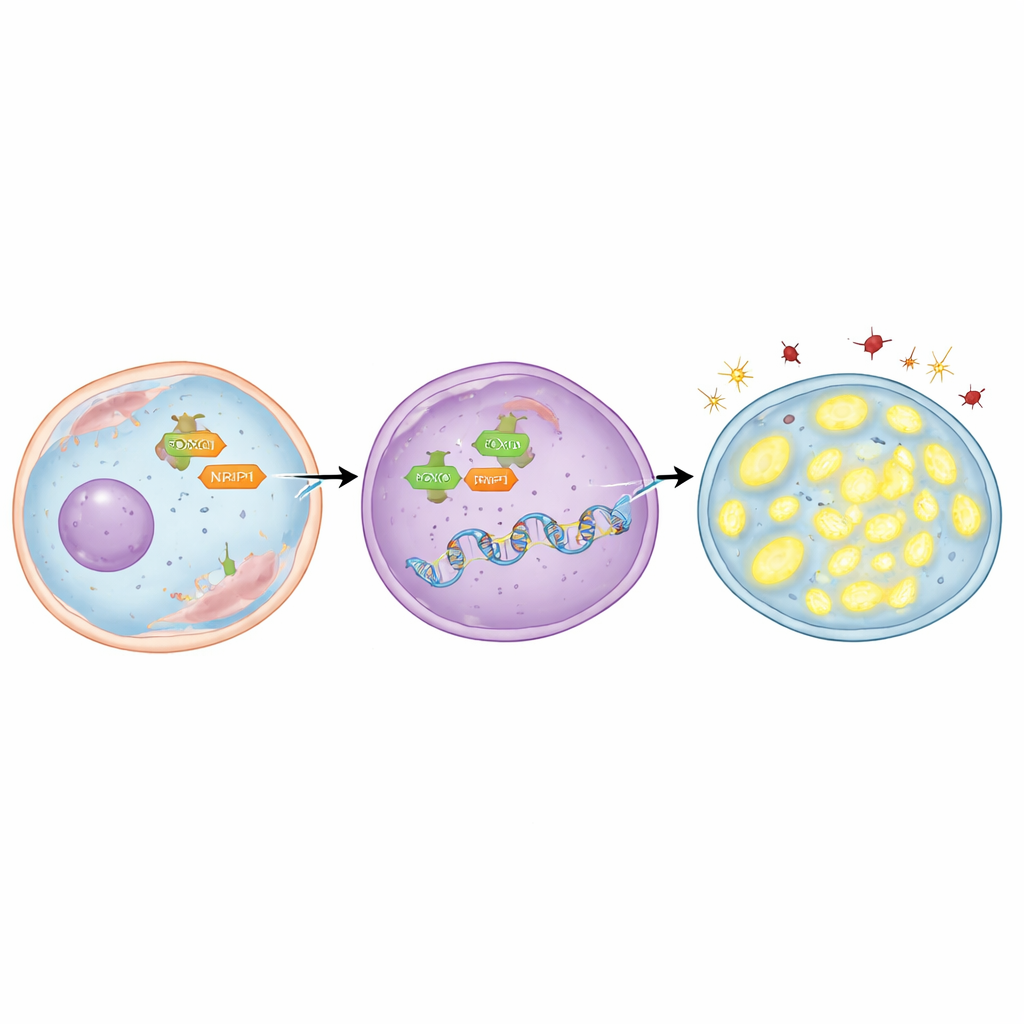
To find out who turns TFAM “on,” the team focused on a regulatory protein called FOXO3, which can move from the cell’s watery interior into the nucleus, where genetic decisions are made. They discovered that the TFAM gene contains several short DNA sequences that FOXO3 can latch onto. Using both computer modeling and laboratory techniques that map protein–DNA contacts, they showed that FOXO3 directly binds these regions. In radioresistant lung cancer cells, FOXO3 was found in higher amounts and was concentrated in the nucleus. When FOXO3 was experimentally reduced, TFAM levels dropped, damaged molecules built up, mitochondria lost DNA, and more cancer cells died after radiation. Conversely, using a compound that drives FOXO3 into the nucleus boosted TFAM, preserved mitochondria, lowered damage, and made cells harder to kill—unless TFAM itself was simultaneously blocked.
A helper protein that boosts the protective response
The story does not stop with FOXO3. The researchers also examined NRIP1, a helper protein that partners with many gene regulators in cancer. They found that NRIP1 physically interacts with FOXO3 and that this partnership is stronger in radioresistant cells. In one lung cancer cell line that naturally lacked functional NRIP1, TFAM levels were relatively low; restoring normal NRIP1 raised TFAM without changing the total amount of FOXO3. In radioresistant cells that did express NRIP1, knocking NRIP1 down reduced TFAM, increased oxidative stress, lowered mitochondrial DNA, and pushed more cells into death after radiation. Importantly, NRIP1 did not change how much FOXO3 reached the nucleus; instead, it acted as a co-activator, making FOXO3 better at turning on TFAM once it was already in place.
Evidence from tumors in living animals
To test whether these molecular events matter in a whole organism, the team implanted radioresistant lung cancer cells into mice to form tumors. When FOXO3 was stably reduced in these cells before implantation, the resulting tumors grew more slowly. Adding radiotherapy on top of FOXO3 reduction shrank tumors even further, without harming the animals’ overall health. These FOXO3-deficient tumors showed more oxidative stress, less mitochondrial DNA, stronger signs of cell death, and substantially lower TFAM levels. Together, these findings support the idea that FOXO3-driven TFAM expression, boosted by NRIP1, helps tumors maintain robust mitochondria and withstand radiation in a living system.
What this means for future lung cancer treatment
For non-specialists, the takeaway is that some lung cancers become radiation-tough by reinforcing their cellular power plants. A trio of players—FOXO3 moving into the nucleus, NRIP1 helping it work, and TFAM strengthening mitochondria—forms a protective circuit that lets tumor cells repair damage and keep going. By blocking FOXO3’s activity, disrupting its partnership with NRIP1, or directly lowering TFAM, future therapies might strip tumors of this hidden armor. Such strategies could make standard radiation more effective, potentially allowing lower doses or better tumor control with fewer treatment failures.
Citation: Zha, Y., Huang, H., Liu, Y. et al. NRIP1 co-activates nuclear translocated FOXO3 to upregulate TFAM expression and promote radioresistance in non-small cell lung cancer. Cell Death Discov. 12, 196 (2026). https://doi.org/10.1038/s41420-026-03028-8
Keywords: non-small cell lung cancer, radioresistance, mitochondria, FOXO3, TFAM