Clear Sky Science · en
Fibroblast-mediated KRAS activation in double-negative prostate cancer
Why this prostate cancer study matters
For many men, hormone-blocking therapies can keep prostate cancer in check for years. Yet some tumors eventually return in a stealthy form that no longer responds to hormone drugs and does not trigger the usual blood tests. This study digs into how these "double-negative" prostate cancers stay alive and spread, revealing a hidden conversation between cancer cells and their surrounding support cells—and pointing to a new weak spot that future drugs might exploit.
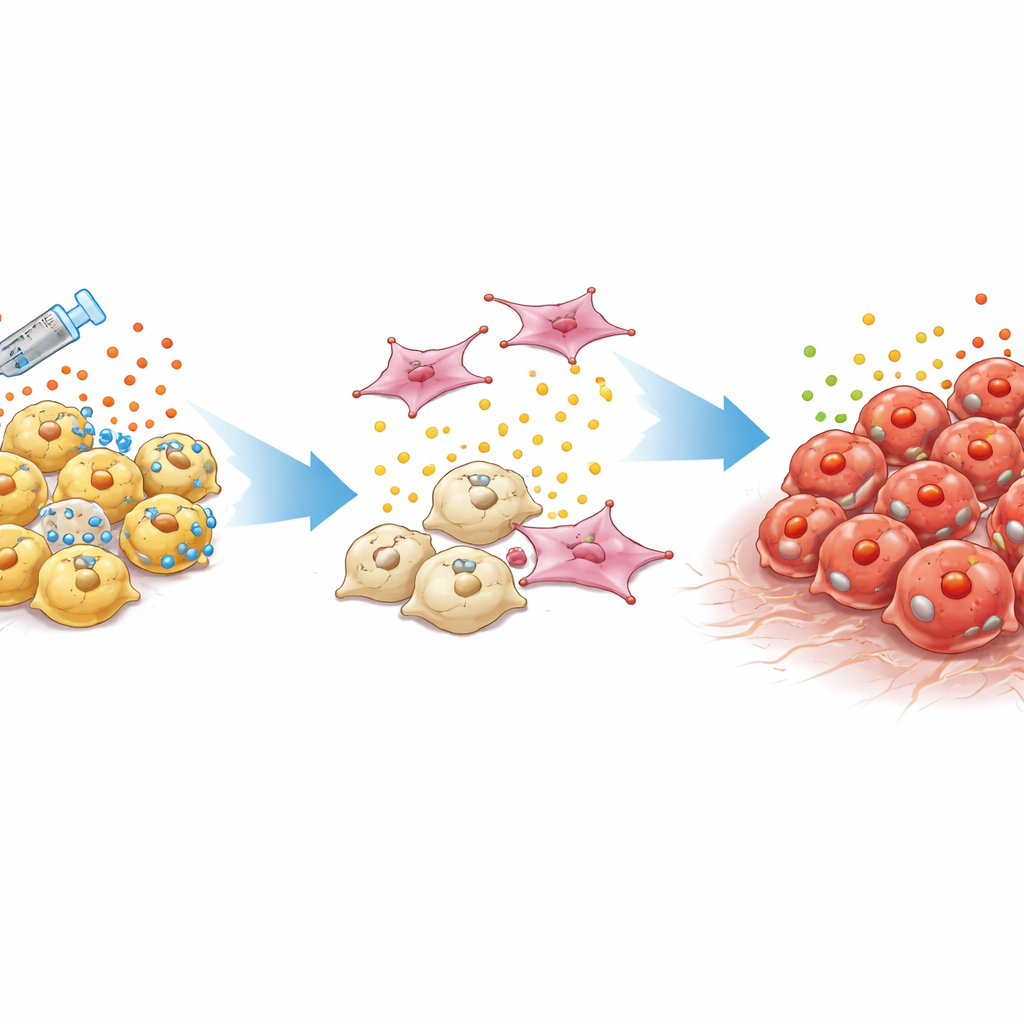
A hard-to-detect, hard-to-treat tumor type
Standard prostate cancers depend on male hormones acting through a protein called the androgen receptor. Doctors suppress this signal with hormone therapy and newer targeted drugs. Over time, however, some tumors evolve into castration-resistant prostate cancer that no longer relies on this pathway. An especially dangerous subtype, called double-negative prostate cancer, lacks both androgen receptor activity and features of nerve-like “neuroendocrine” cells. These tumors often grow back locally or spread despite low levels of the usual markers in blood tests, leaving clinicians with few clues and no proven treatments.
A growth engine that turns on when hormones are shut off
The researchers focused on KRAS, a well-known molecular switch that drives growth in lung and colon cancers when mutated. In prostate cancer, such mutations are rare, but patient DNA profiling hinted that KRAS might be involved in double-negative cases. By comparing different prostate cancer cell lines, they found that KRAS signaling did very little in hormone-dependent cells but became crucial once cells no longer relied on androgen signals. In DNPC-like cells, reducing KRAS sharply cut their ability to grow, move, and invade, while hormone-sensitive cells barely noticed the loss—suggesting KRAS becomes a key engine only in the late, hormone-independent state.
How nearby support cells feed the cancer switch
Tumors are not just rogue cancer cells; they are communities that include fibroblasts—connective-tissue cells that help shape the local environment. The team discovered that when androgen signaling is blocked in prostate cancer cells, the cancer cells change the types of receptors they display on their surface, becoming more receptive to a growth signal called FGF8. At the same time, the hormone-starved cancer cells release a distress signal, the chemokine CCL2, into their surroundings. Fibroblast-like stromal cells in advanced prostate tumors carry receptors for CCL2. When CCL2 reaches these stromal cells, it switches on an internal pathway that boosts their production and release of FGF8. That FGF8 then circles back to the cancer cells, activates their altered receptors, and in turn flips on KRAS, driving powerful growth and survival signals.
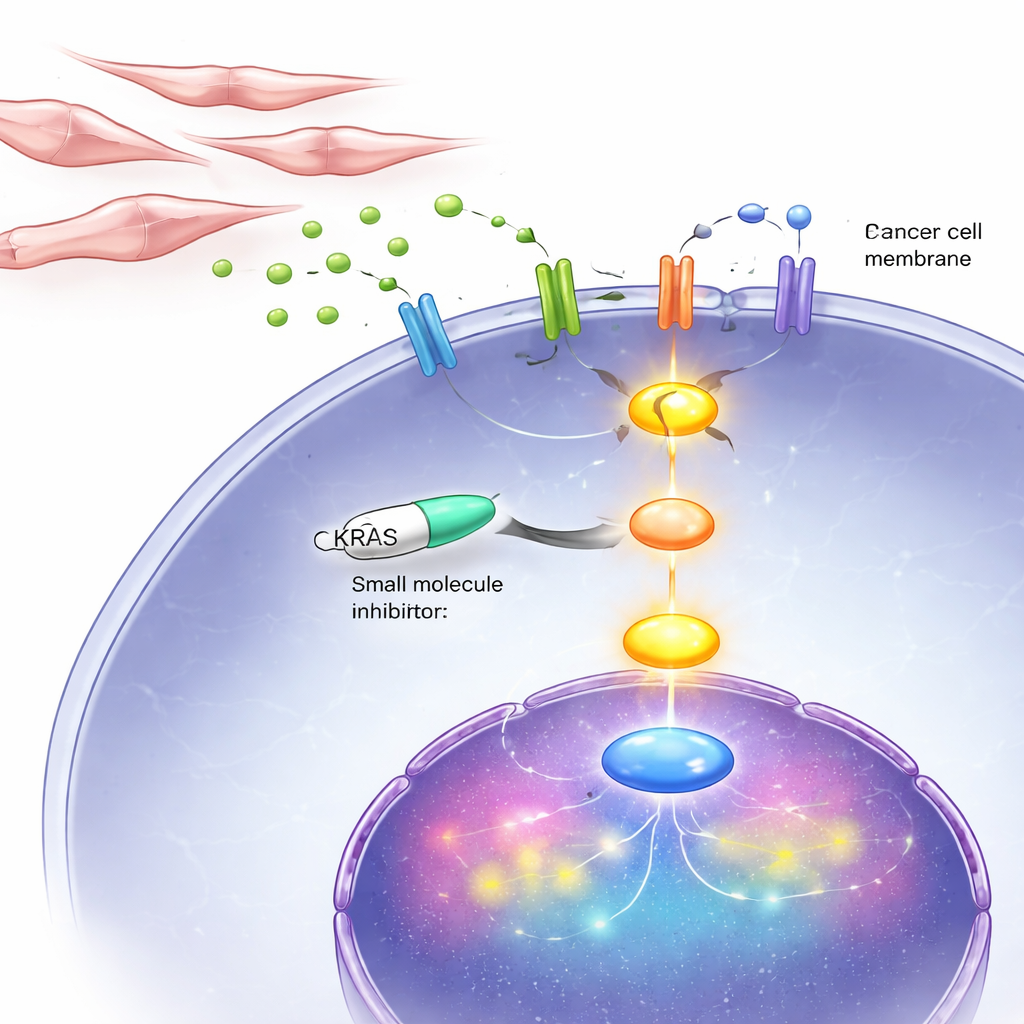
Blocking the KRAS lifeline in cells and in mice
Because this vicious cycle ultimately funnels through KRAS, the researchers tested a drug, BI-3406, that interferes with how KRAS is turned on inside cells. In lab dishes, this inhibitor strongly slowed the growth and movement of androgen-independent prostate cancer cells, including those with abnormal KRAS and those with normal KRAS activated by signals from fibroblasts. In mice implanted with DNPC-like tumors, daily treatment with the inhibitor shrank tumors and reduced markers of cell division, while boosting signs of programmed cell death. Importantly, the drug had much less impact on hormone-dependent cancer cells, echoing the idea that KRAS is especially critical in the late, double-negative stage.
What this means for future treatment
To a layperson, the takeaway is that when hormone therapy drives prostate cancer into a corner, the tumor can escape by recruiting nearby fibroblasts to feed it a different growth signal that ultimately flips on KRAS. This study maps that escape route in detail and shows that cutting off KRAS signaling can kill these resistant cells in models. While more work is needed in patients, especially with drugs that can safely block KRAS or its upstream helpers, the findings highlight a new strategy: targeting the CCL2–FGF8–KRAS chain to treat the most stubborn, hormone-independent forms of prostate cancer.
Citation: Kamijima, T., Izumi, K., Hiratsuka, K. et al. Fibroblast-mediated KRAS activation in double-negative prostate cancer. Cell Death Dis 17, 403 (2026). https://doi.org/10.1038/s41419-026-08800-3
Keywords: double-negative prostate cancer, tumor microenvironment, KRAS signaling, fibroblasts, androgen-independent cancer