Clear Sky Science · en
TGF-β/SMAD4/14-3-3σ/TFEB axis promotes mesenchymal-epithelial transition and inhibits autophagy in colorectal cancer
Why this study matters for colon cancer
Colorectal cancer is one of the leading causes of cancer death because tumor cells often spread from the intestine to distant organs. This study uncovers a previously unrecognized safety circuit inside intestinal cells that can restrain this spread. By mapping how several molecules talk to each other, the researchers show how normal signals in the gut can push cancer cells back toward a more orderly state and cut off one of their survival tricks, a self‑cleaning process called autophagy.
A protective message in the gut
Our intestines are constantly exposed to food, bacteria, and mechanical stress, so cells in the gut rely on strong control systems to keep growth and repair in balance. One major signal, called TGF‑β, usually acts as a brake on early tumor development. It works through a protein named SMAD4, which moves into the cell nucleus and switches genes on or off. In colorectal cancers, SMAD4 is frequently lost, and patients whose tumors lack this protein tend to have more metastases and worse survival. The authors set out to understand which specific genes SMAD4 controls that might explain its ability to block invasion and spread.
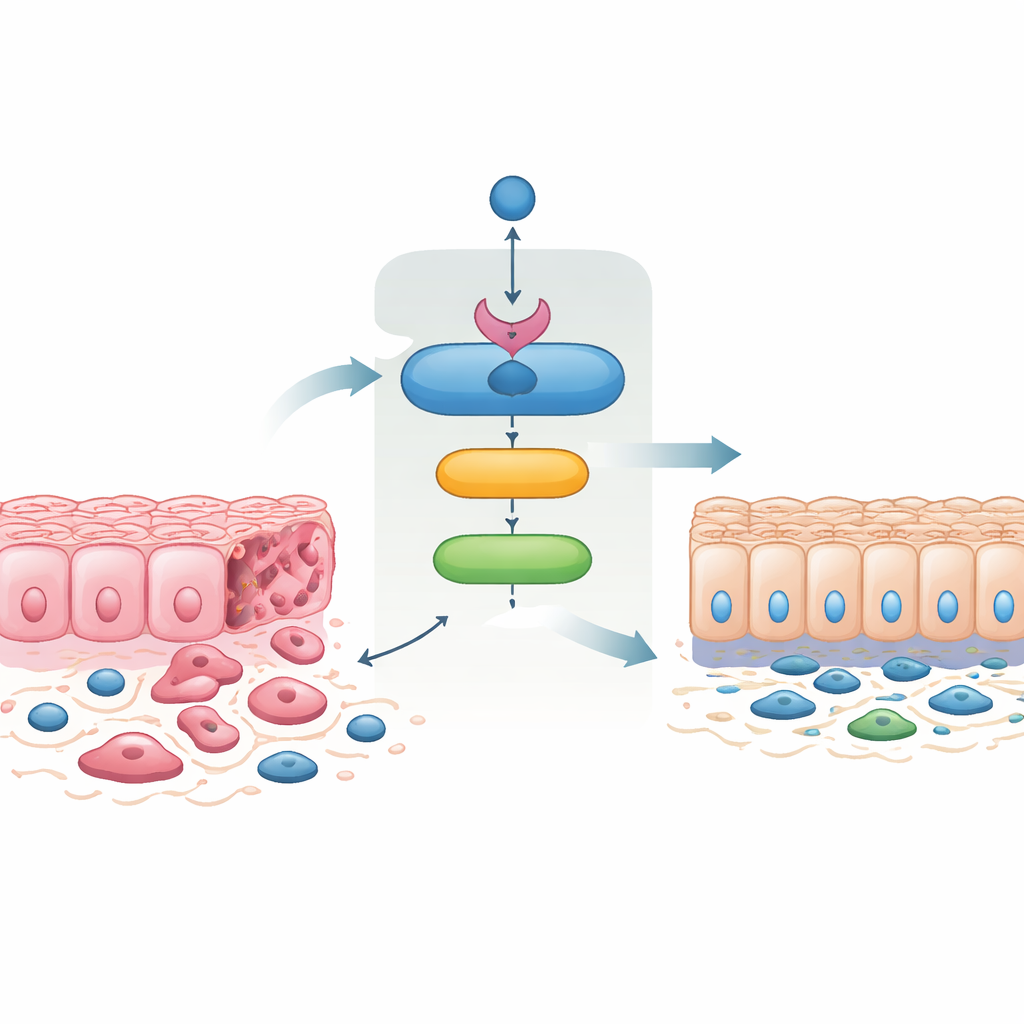
A chain of guardians inside cancer cells
The team discovered that SMAD4 directly activates a gene called 14‑3‑3σ, long known as a tumor suppressor. Using human colorectal cancer cell lines, patient‑derived mini‑tumors grown in the lab, and mouse models, they showed that turning on SMAD4 reliably boosts 14‑3‑3σ levels, while removing SMAD4 prevents this increase. They pinpointed docking sites for SMAD4 in the 14‑3‑3σ control region and confirmed that SMAD4 physically occupies these sites, firmly establishing 14‑3‑3σ as a direct target in both human and mouse intestinal cells. In normal mouse intestine, 14‑3‑3σ is especially abundant at the tips of the villi, but this pattern disappears when Smad4 is deleted, highlighting how closely the two are linked during tissue differentiation.
Pushing cancer cells back toward order
Cancer cells often undergo an "epithelial‑mesenchymal transition," changing from tightly connected, sheet‑like cells into more mobile, spindle‑shaped ones that more easily invade and seed metastases. The reverse process, mesenchymal‑epithelial transition, restores tight cell–cell contacts and reduces invasiveness. When the researchers reintroduced SMAD4 into aggressive colorectal cancer cells that normally lack it, the cells shifted toward an epithelial appearance, regained membrane‑bound cell adhesion proteins, and lost features associated with mobility and invasion. These SMAD4‑restored cells migrated less, invaded less through artificial membranes, and formed fewer colonies. Remarkably, forcing cells to produce 14‑3‑3σ alone produced very similar changes, and blocking 14‑3‑3σ largely erased the effects of SMAD4. This shows that 14‑3‑3σ is not just a bystander but a key mediator of SMAD4’s ability to tame invasive behavior.
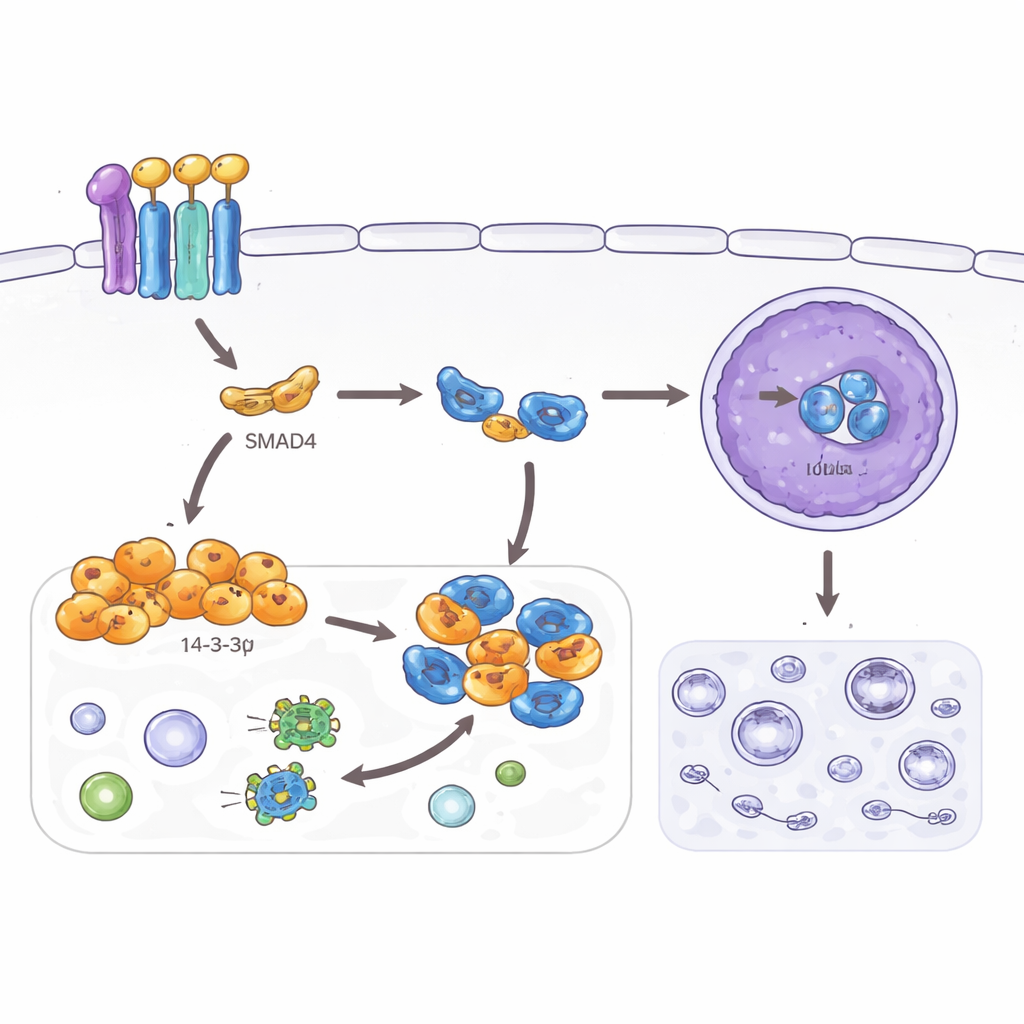
Switching off a survival pathway
The study also examined autophagy, a recycling program that helps cells survive stress by breaking down internal components. While useful in normal tissues, heightened autophagy can help cancer cells cope with hostile environments and treatment. The authors found that SMAD4 and 14‑3‑3σ both lower markers of autophagy in colorectal cancer cells and reduce the number of recycling vesicles observed under the microscope. They traced this effect to another protein, TFEB, a master controller of autophagy and lysosomes. Normally, when TFEB enters the nucleus it activates many autophagy‑related genes. Here, SMAD4‑driven 14‑3‑3σ binds TFEB in the cell’s outer compartment and keeps it from reaching the nucleus, but only when a specific site on TFEB has been chemically tagged. When this site is mutated, 14‑3‑3σ can no longer hold TFEB back, autophagy resumes, and the suppressive effects on invasion and cell shape are weakened.
What this means for patients
Taken together, the findings outline a linear protective axis in the intestine: TGF‑β activates SMAD4, SMAD4 turns on 14‑3‑3σ, and 14‑3‑3σ traps TFEB outside the nucleus. This combination pushes colorectal cancer cells toward a more orderly, less mobile state and dampens their self‑cleaning machinery, which may make them less able to spread and more vulnerable to treatment. When SMAD4 or 14‑3‑3σ are lost, TFEB becomes more active, potentially boosting both cell plasticity and stress resistance. Although further work in living animals and clinical samples is needed, the pathway revealed here highlights new molecular points where future drugs could reinforce the body’s own defenses against metastatic colorectal cancer.
Citation: Chen, X., Winter, M., Rokavec, M. et al. TGF-β/SMAD4/14-3-3σ/TFEB axis promotes mesenchymal-epithelial transition and inhibits autophagy in colorectal cancer. Cell Death Dis 17, 397 (2026). https://doi.org/10.1038/s41419-026-08733-x
Keywords: colorectal cancer, metastasis, autophagy, epithelial plasticity, TFEB