Clear Sky Science · en
Antagonistic SMAD2/3 control of TIMP-1, VEGF-A, and hypoxia signaling in myofibroblasts shapes histotype-specific angiogenesis in lung cancer
Why blood vessels in lung tumors matter
Lung cancer is not a single disease, and patients with different tumor types often respond very differently to the same drugs. This study asks a simple but crucial question: why do some common lung tumors grow rich networks of blood vessels while others suffocate in low-oxygen pockets—and how does the surrounding scar-like tissue help decide the outcome? Understanding this hidden conversation between cancer cells, support cells, and blood vessels could guide more precise treatments and improve the success of both chemotherapy and immunotherapy.
Two common lung cancers, two vascular landscapes
The authors focus on the two major forms of non-small cell lung cancer: adenocarcinoma, which often arises in the outer lung, and squamous cell carcinoma, which tends to form nearer the central airways. By analyzing thousands of patient samples and images from large public databases, they show that adenocarcinomas are generally better supplied with blood vessels, with wider vessel channels and stronger activity of genes linked to vessel growth. In contrast, squamous tumors more often show signs of poor blood supply, including extensive dead tissue and high levels of hypoxia markers, which signal lack of oxygen. This split in vascular behavior persists even after accounting for clinical variables such as age, tumor stage, and smoking history. 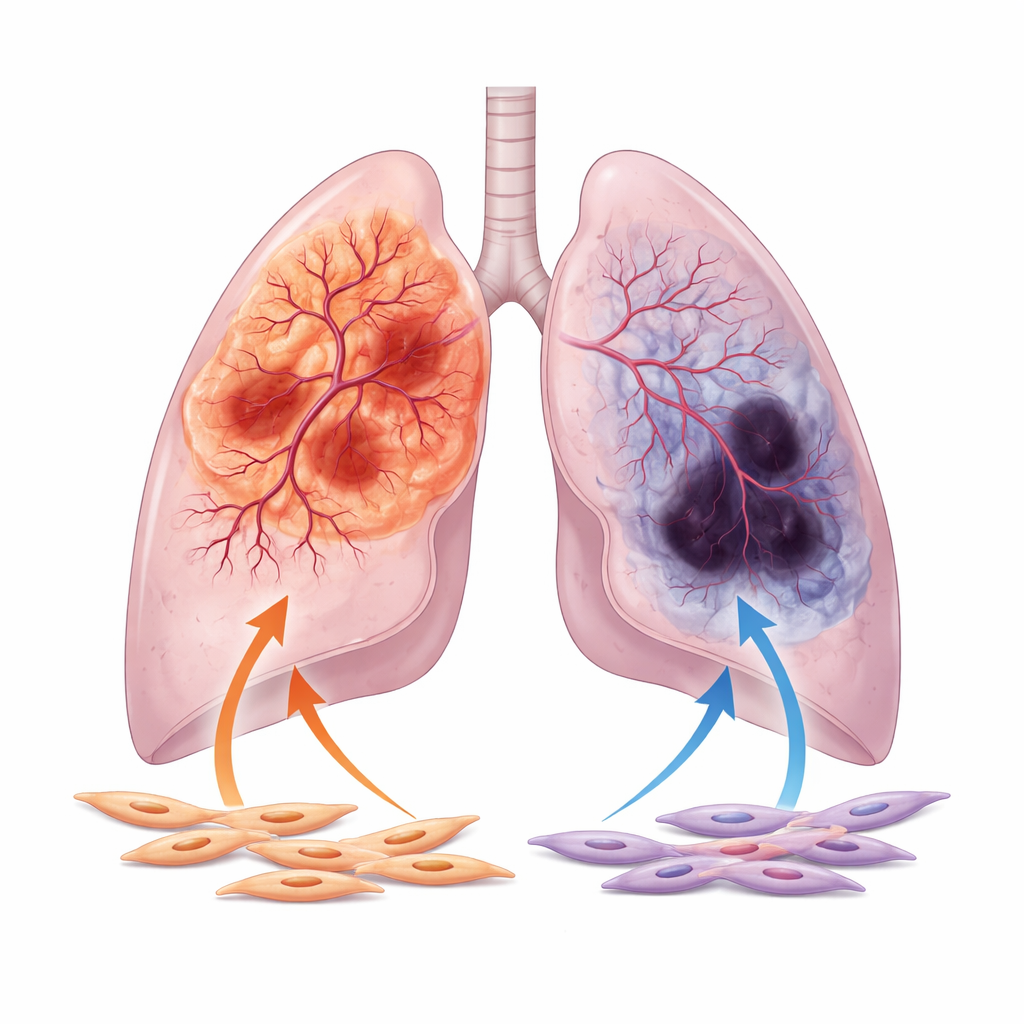
The quiet power of tumor fibroblasts
Attention then turns to tumor-associated fibroblasts, the contractile, myofibroblast-like cells that surround and infiltrate lung tumors. These cells, better known for producing scar tissue, also secrete many substances that can either encourage or restrain vessel growth. Using single-cell RNA sequencing, the researchers find that fibroblasts in adenocarcinoma interact more intensely with nearby endothelial cells, the building blocks of blood vessels, than fibroblasts in squamous tumors. In lab dishes, fluid collected from adenocarcinoma fibroblasts makes endothelial cells migrate and form dense, intertwined capillary-like networks, while fluid from squamous fibroblasts triggers much weaker and sparser structures.
Key messengers: VEGF, TIMP-1, and a molecular switch
To uncover which signals are responsible, the team profiles dozens of secreted factors. Adenocarcinoma fibroblasts produce particularly high levels of VEGF-A, a classic driver of new vessel growth, and TIMP-1, a protein better known as a brake on tissue-degrading enzymes. Here, TIMP-1 shows a different face: in controlled experiments, adding TIMP-1 boosts the branching complexity of endothelial networks, creating the tangled architectures typical of tumor blood vessels. Removing TIMP-1 from adenocarcinoma fibroblasts sharply reduces vessel branching in culture and diminishes blood-vessel density and tumor growth in mice. Crucially, this branching effect depends on a baseline level of VEGF signaling, suggesting that VEGF acts as the gas pedal while TIMP-1 fine-tunes how chaotic the resulting vasculature becomes.
Opposing controls of growth versus hypoxia
Underlying these differences is a molecular "switch" inside fibroblasts that balances two related proteins, SMAD2 and SMAD3, which relay signals from the growth factor TGF-β. Adenocarcinoma fibroblasts favor SMAD3, and this high SMAD3 to SMAD2 ratio drives the overproduction of VEGF-A, TIMP-1, and other vessel-promoting factors. When the researchers dial down SMAD3 in these cells, their ability to stimulate endothelial migration, branching, and tumor vascularization collapses. Squamous fibroblasts, often shaped by smoking-induced epigenetic changes, show the opposite pattern: SMAD3 is silenced and SMAD2 is increased. This state boosts hypoxia-related genes and HIF-1α, a master regulator of low-oxygen responses, but paradoxically does not translate into strong VEGF-A output. As a result, these tumors remain more hypoxic and necrotic, with weaker support for new vessels despite a strong internal “low oxygen” alarm. 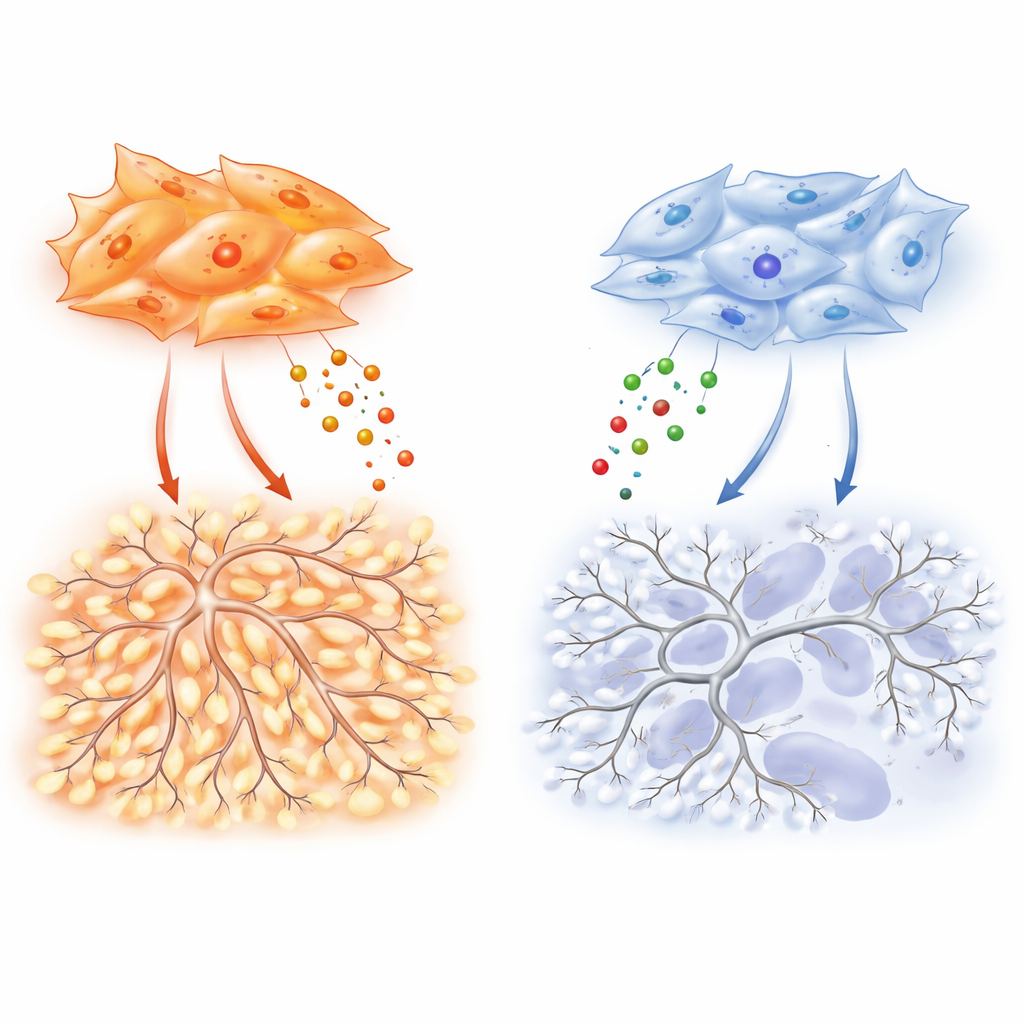
What this means for patients and future therapies
By tying the behavior of blood vessels to fibroblast programs in each tumor type, the study helps explain why anti-angiogenic drugs have worked better in adenocarcinoma than in squamous lung cancer. In adenocarcinoma, targeting the SMAD3–VEGF–TIMP-1 axis could both normalize the abnormal vasculature and relieve local immune suppression, making checkpoint-blocking immunotherapies more effective. In squamous tumors, where fibroblasts foster a harsh, oxygen-poor and acidic environment rather than strong vessel growth, therapies that ease hypoxia, neutralize acidity, or block specific inflammatory signals may be more promising than classic anti-angiogenic approaches. For patients, this work underscores that the success of vascular and immune-targeted treatments depends not just on cancer cells, but on the fibrous stroma that quietly orchestrates the tumor’s blood supply.
Citation: Díaz-Valdivia, N., Duch, P., Ikemori, R. et al. Antagonistic SMAD2/3 control of TIMP-1, VEGF-A, and hypoxia signaling in myofibroblasts shapes histotype-specific angiogenesis in lung cancer. Cell Death Dis 17, 431 (2026). https://doi.org/10.1038/s41419-026-08677-2
Keywords: lung cancer angiogenesis, tumor-associated fibroblasts, VEGF and TIMP-1, tumor microenvironment, SMAD signaling