Clear Sky Science · en
SARS-CoV-2 nonspike structural proteins hijack mucosa epithelial cell fate
When the Body’s Linings Lose Their Identity
COVID-19 is known for attacking the lungs, but many patients also develop stubborn ulcers in the mouth, gut, and other organs lined by delicate mucous membranes. This study asks a deceptively simple question: before we see visible wounds and inflammation, what exactly is happening to the cells that form these protective linings—and could that early damage point to new treatment strategies?
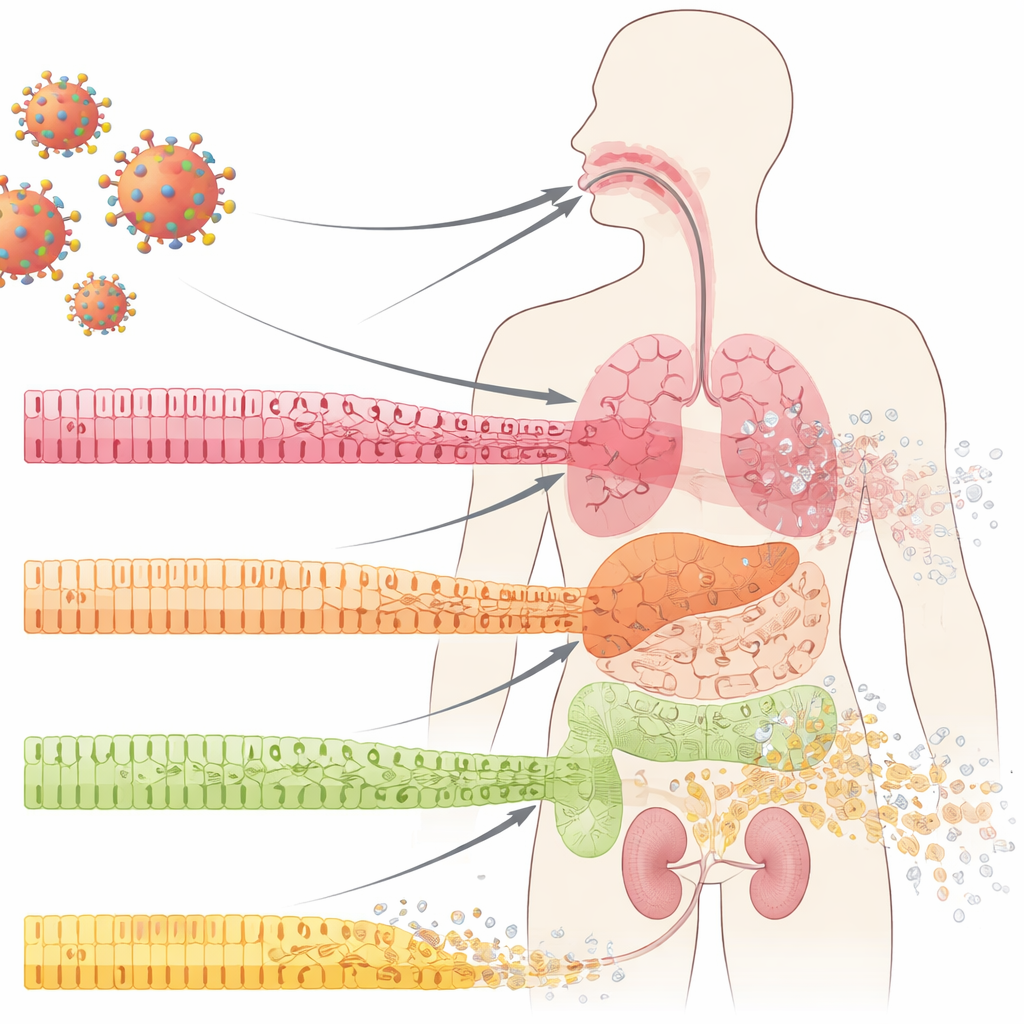
The Frontline Cells That Shield Our Organs
Our internal surfaces—inside the mouth, lungs, intestines, and kidneys—are covered by epithelial cells arranged in orderly layers. These cells act like tiles in a waterproof wall: tightly joined, specialized, and constantly renewing from a pool of underlying progenitor cells. In COVID-19, doctors have long noticed dramatic breakdowns of these mucosal barriers, including ulcers and tissue erosion. The authors examined samples from the oral cavity, lungs, and kidneys of COVID-19 patients and found something striking even in areas that still looked normal under routine inspection: the mature, specialized cells were losing their identity, and markers of less mature, “stem-like” cells were rising. This process, called dedifferentiation, usually appears during controlled tissue repair—but here it was appearing early in infection, before obvious inflammation or scarring.
Viral Shell Proteins That Reprogram Cell Fate
The coronavirus is best known for its spike protein, which it uses to enter cells. But the virus also carries other structural proteins that form its shell and interior. In this work, the researchers focused on three of these “nonspike” proteins: envelope (E), membrane (M), and nucleocapsid (N). Using sophisticated 3D models of human oral mucosa and cultured mouse tongue tissue, they delivered each of these proteins into epithelial cells to see what would happen. All three could push the cells toward a less differentiated state, but the envelope protein stood out. It rapidly reduced markers of mature epithelial cells, increased markers of more primitive cells, and weakened E-cadherin, a molecule that helps cells stick tightly together. At the same time, cells began to show DNA damage signals such as micronuclei, became stuck in a checkpoint early in the cell cycle, and were more likely to undergo programmed cell death. Together, these changes suggest that the virus’s structural shell is not just a passive coat but an active saboteur of epithelial stability.
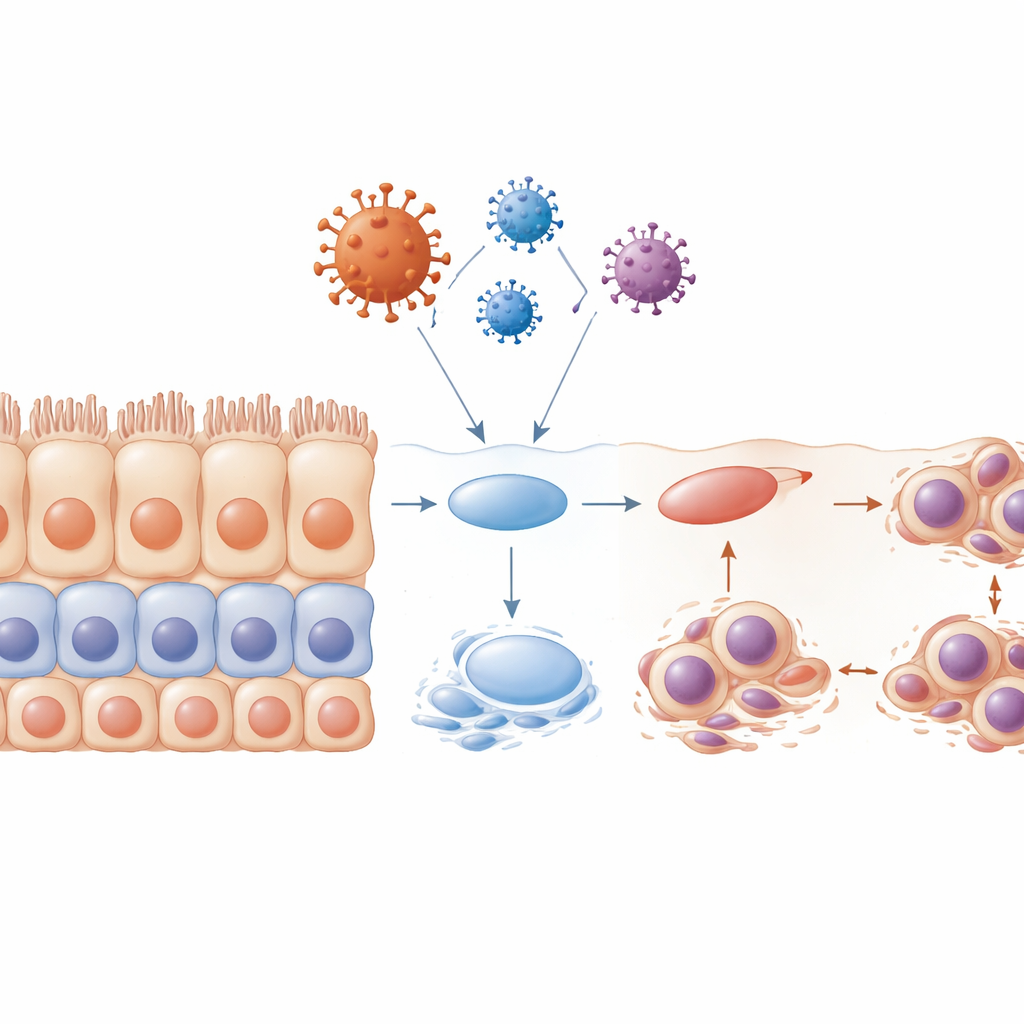
Disrupted Layering and Damaged Cellular “Brushes”
Healthy stratified epithelia renew themselves through a carefully choreographed program: progenitor cells divide near the base, then their descendants move upward and mature into highly specialized surface cells. When the team exposed cells to the envelope protein before they had formed full layers, this program essentially collapsed. The resulting tissue was disorganized, with poor layering, fewer mature markers, and many cells trapped in an early cell-cycle phase. Many of these cells carried misshapen or multiple nuclei and underwent apoptosis, leaving gaps where a continuous barrier should be. The researchers also examined tiny hair-like structures called cilia, which help move mucus and debris. They found that cilia shrank or disappeared, especially under the influence of the envelope protein, and that this occurred at key stages of the cell cycle. This loss of cilia would further weaken the tissue’s ability to clear viruses and contaminants.
A Hidden Molecular Switch and a New Drug Target
To uncover how these viral proteins rewire cell behavior, the authors compared the protein profiles of infected versus uninfected epithelial cells. One molecule, calponin 2 (CNN2), consistently stood out: it was strongly increased by all three nonspike proteins and was elevated in the oral, lung, and kidney tissues of COVID-19 patients. CNN2 helps organize the cell’s internal scaffolding and responds to mechanical stress. When the scientists reduced CNN2 levels using targeted genetic tools, many of the harmful effects of the envelope protein were blunted: epithelial markers were preserved, tissue layering improved, and fewer cells died. They then traced CNN2 regulation upstream to a transcription factor called GLIS2, which normally keeps CNN2 in check. In patient samples and in virus-protein–treated cells, GLIS2 levels were noticeably decreased, releasing the brake on CNN2. This GLIS2–CNN2 axis appears to be a key pathway through which SARS-CoV-2’s shell proteins derail epithelial cell fate.
What This Means for Future COVID-19 Care
To a non-specialist, the core message is that SARS-CoV-2 can quietly destabilize the body’s protective linings long before obvious tissue damage appears. Its nonspike structural proteins, especially the envelope protein, push frontline epithelial cells to lose their specialization, disrupt their orderly layering, damage their DNA, and kill them off. At the molecular level, the virus seems to execute a “double hijack”: it both dedifferentiates mature cells and blocks the normal maturation of progenitor cells, largely through a GLIS2–CNN2 control switch. Because CNN2 sits at a crucial point in this pathway and its reduction can reverse much of the damage in lab models, it emerges as a promising target for therapies aimed at protecting mucosal barriers in COVID-19 and possibly reducing stubborn ulcers and multi-organ injury.
Citation: Gao, Y., Souza, L.L., Kang, H.S. et al. SARS-CoV-2 nonspike structural proteins hijack mucosa epithelial cell fate. Cell Death Dis 17, 340 (2026). https://doi.org/10.1038/s41419-026-08611-6
Keywords: SARS-CoV-2 envelope protein, mucosal epithelium, cell dedifferentiation, CNN2, GLIS2