Clear Sky Science · en
The RASSF1C-HIF-1α axis drives macrophage lipid metabolism to promote pancreatic cancer
Why this cancer story matters
Pancreatic cancer is one of the deadliest cancers, in part because it hides deep in the body and is often found late. But its aggressiveness is not just about the tumor cells themselves. This study shows how a lack of oxygen inside pancreatic tumors rewires both cancer cells and nearby immune cells in a way that helps the cancer grow and spread. By uncovering this chain of events, the researchers point to new ways to attack the disease by targeting both tumor metabolism and the immune system.
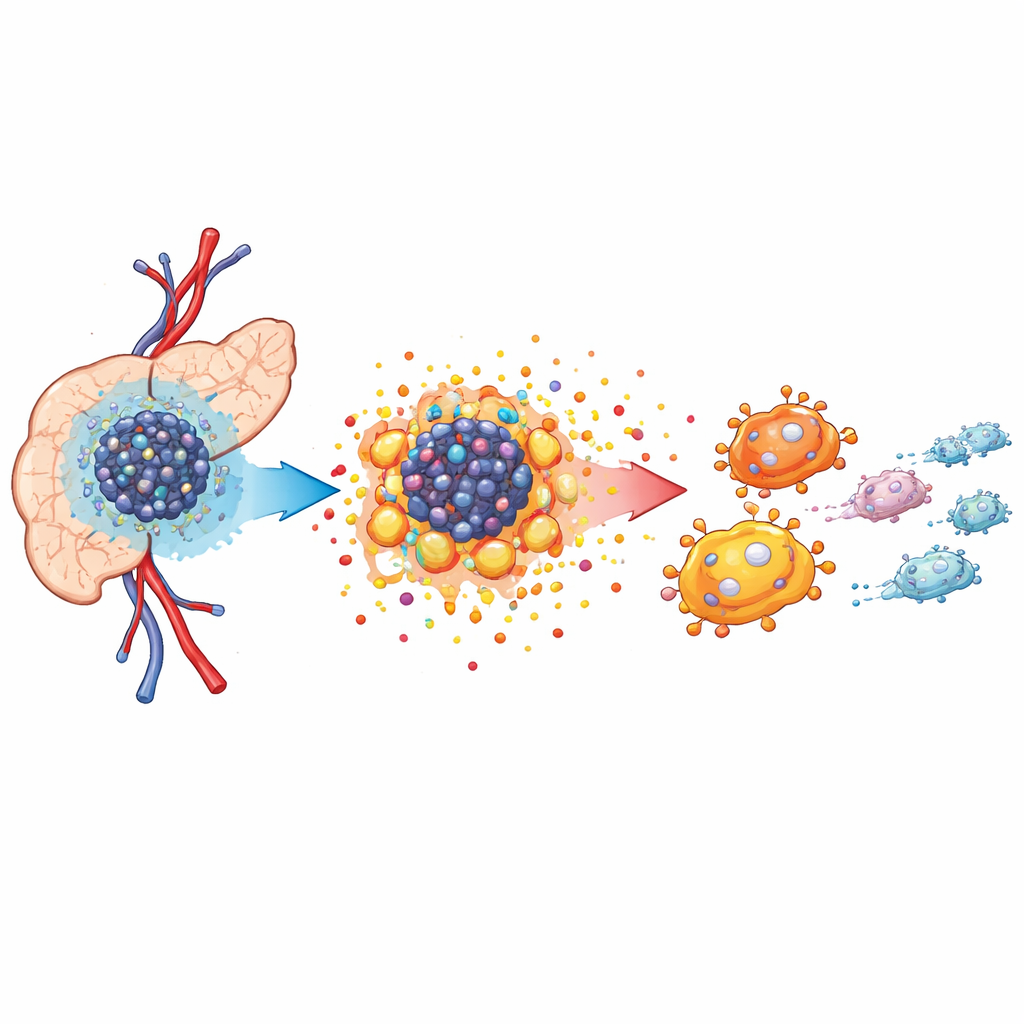
Low oxygen and a hungry tumor neighborhood
Pancreatic tumors grow in a crowded, hostile neighborhood known as the tumor microenvironment. Blood supply is poor, so oxygen levels drop, forcing cancer cells to change how they make energy. Using single-cell RNA sequencing and spatial mapping in mouse models, the authors showed that tumor tissue is packed with cancer cells, cancer-associated fibroblasts, and specialized immune cells called tumor-associated macrophages. These macrophages cluster near cancer cells and tend to adopt a growth-supporting, rather than cancer-fighting, role. The analysis revealed that cancer cells rely heavily on altered metabolism to fuel rapid growth, setting the stage for a broader reshaping of the tissue environment.
Fueling cancer with sugar and acid
When oxygen is scarce, cancer cells lean on a process similar to fast, inefficient sugar burning, often called the Warburg effect. The team found that a protein variant named RASSF1C, previously linked to cancer behavior, becomes strongly activated in low oxygen. Driven by another oxygen-sensing protein, HIF-1α, RASSF1C boosts glycolysis, leading to high production of lactate and acidification of the surroundings. In cell and animal experiments, dialing down RASSF1C reduced acid production and slowed tumor growth, while boosting it had the opposite effect. Importantly, blocking lactate formation with a drug blunted the growth advantage conferred by RASSF1C, showing that this metabolic shift is not just a side effect—it actively fuels disease progression.
How tumor byproducts reprogram immune cells
The lactate released by tumor cells does more than change local chemistry; it acts as a signal. When the researchers bathed macrophages in conditioned fluid from lactate-rich pancreatic cancer cells, the macrophages shifted toward an M2-like, tumor-supporting state. They produced more anti-inflammatory factors and fewer molecules linked to attack and clearance of cancer cells. In mice, tumors with high RASSF1C levels contained more of these pro-tumor macrophages, an effect that could be dampened by blocking lactate production. Further tests showed that lactate also rewired how macrophages handle fats and cholesterol, increasing lipid buildup in ways that track with a more supportive niche for tumor growth and spread.
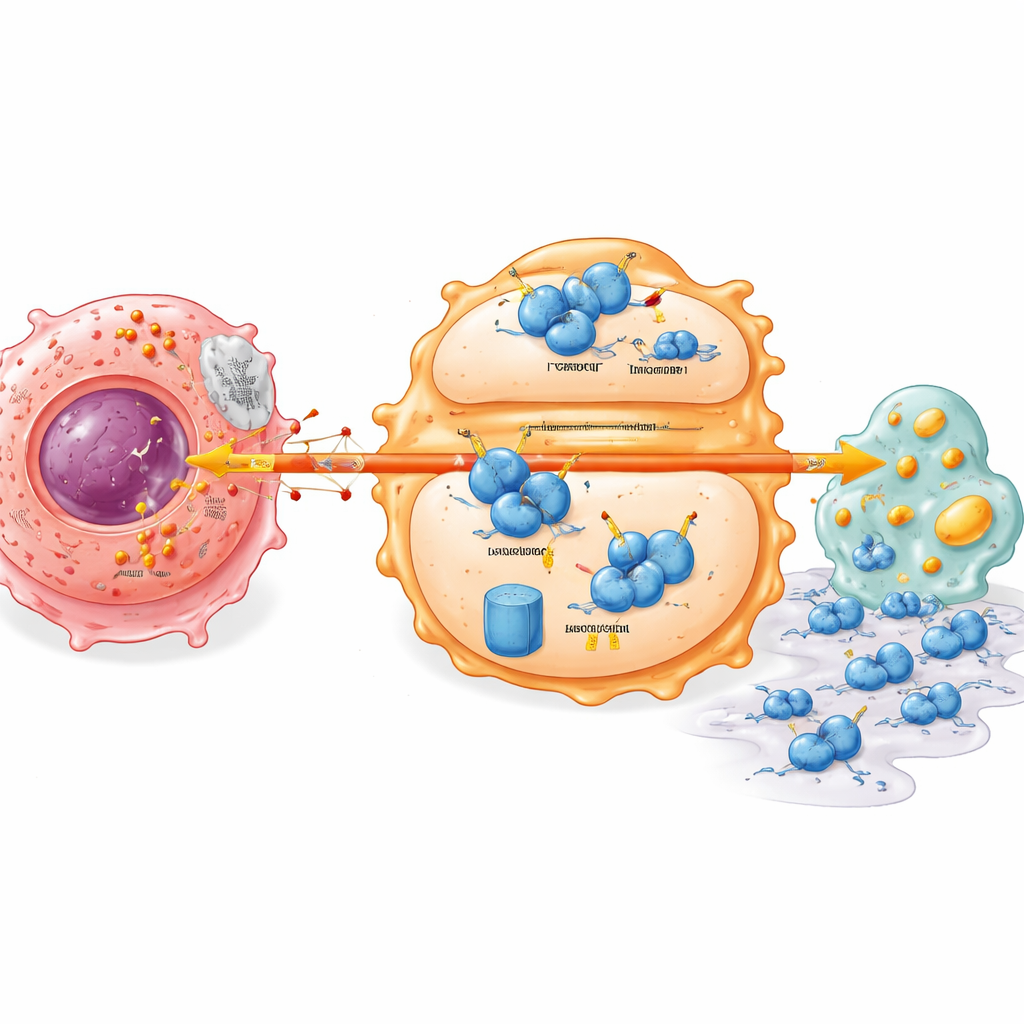
A hidden stability switch inside immune cells
Digging deeper, the study uncovered a molecular switch that links tumor metabolism to immune cell behavior. A key immune regulator called IRF7, which normally supports a more vigilant, attack-ready macrophage state and healthy lipid handling, was found at low levels in pancreatic tumors. Lactate from cancer cells caused IRF7 protein in macrophages to become unstable and break down faster. This depended on another protein, UFL1, which attaches a small tag (UFM1) that protects IRF7 from being marked for destruction. Lactate suppressed UFL1 in macrophages, tilting the balance toward a tagging system that sends IRF7 to the cell’s protein shredder. Without stable IRF7, macrophages accumulate metabolic defects and more readily assist tumor cells in migrating and invading surrounding tissues.
What this means for future treatments
In tumor samples from people with pancreatic cancer, the researchers saw a clear pattern: higher levels of RASSF1C and HIF-1α in tumor cells, and lower levels of UFL1 and IRF7 in the surrounding tissue. These patterns matched more advanced disease and metastatic features. Together, the findings outline a chain in which low oxygen activates RASSF1C and HIF-1α, drives lactate overproduction, and in turn disables a protective UFL1–IRF7 brake inside macrophages. For lay readers, the takeaway is that pancreatic cancer thrives by turning its own waste products into weapons that disarm nearby immune cells. Interrupting this chain—by limiting lactate, blocking RASSF1C–HIF-1α signaling, or stabilizing IRF7—could open new avenues for therapies that combine metabolic drugs with immune-based approaches.
Citation: Zhan, T., Huang, M., Chen, M. et al. The RASSF1C-HIF-1α axis drives macrophage lipid metabolism to promote pancreatic cancer. Cell Death Dis 17, 430 (2026). https://doi.org/10.1038/s41419-026-08609-0
Keywords: pancreatic cancer, tumor microenvironment, tumor metabolism, macrophages, lactate