Clear Sky Science · en
Lactylation stabilizes PD-L1 to promote tumor immune evasion and cell growth
Why this cancer story matters
Cancer immunotherapy has transformed treatment for many patients, yet only a minority enjoy long-lasting benefits. This study uncovers a hidden chemical trick that tumors use to shield themselves from the immune system—and suggests a surprisingly simple way to turn that trick against the cancer. By revealing how a common metabolic by-product, lactate, helps tumors hide from killer T cells, the work points to new strategies to make current immune therapies work better and for more people.
How cancer cells talk to the immune system
Our immune system relies on molecular "handshakes" between cells to decide when to attack and when to stand down. One of the most important of these is between a protein on tumor cells called PD-L1 and its partner PD-1 on T cells. When PD-L1 is abundant on cancer cells, it tells T cells to back off, allowing the tumor to grow unchecked. Drugs that block PD-1 or PD-L1 can release this brake, but their success often depends on how much PD-L1 the tumor displays. Understanding what controls PD-L1 levels on cancer cells is therefore central to predicting and improving responses to these treatments.
Fuel waste that becomes a weapon
Many tumors rely heavily on a sugar-burning process called glycolysis, which produces large amounts of lactate. Long viewed mainly as metabolic waste, lactate is now recognized as an active signaling molecule. The researchers found that adding lactate to lung and breast cancer cells increased the amount of PD-L1 protein, while blocking glycolysis reduced it. This effect appeared in both human and mouse cells and could occur even when lactate did not boost PD-L1 gene activity, implying that lactate was stabilizing existing PD-L1 protein rather than just turning on its production.
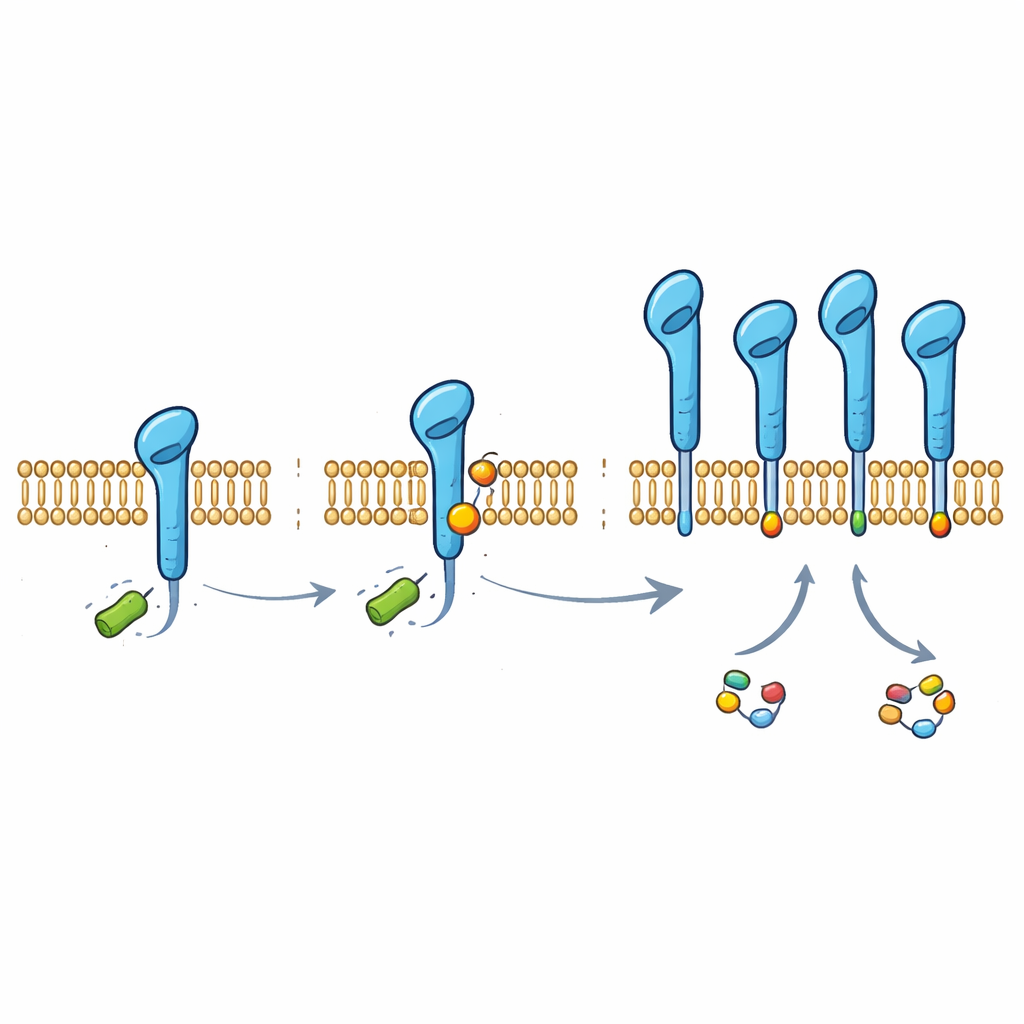
A tiny chemical tag that protects PD-L1
Digging deeper, the team discovered that PD-L1 carries a small chemical tag derived from lactate, attached to one particular building block of the protein (a lysine at position 280). This tag, called lactylation, was found on PD-L1 inside several lung cancer cell lines and normal airway cells. When the scientists changed that single lysine so it could no longer be modified, PD-L1 lost most of its lactate-dependent protection and became easier for the cell to destroy. Normally, a molecular machine named HUWE1 marks PD-L1 for disposal by adding chains of another small molecule, ubiquitin, at a nearby position. The new lactate-based tag physically interferes with HUWE1’s access, preventing this “trash label” from being attached and thereby shielding PD-L1 from breakdown.
The enzyme that writes the lactate tag
To understand how this tag is added, the researchers looked for enzymes that could use lactate directly as a donor. They identified alanyl-tRNA synthetase 1 (AARS1), a normally routine protein-synthesis enzyme, as the key “writer” of PD-L1 lactylation. In test-tube experiments and cells, AARS1 could attach a lactate group to the PD-L1 tail, whereas a related enzyme, AARS2, could not. Overproducing AARS1 increased the lactate tag on PD-L1, reduced HUWE1 binding and ubiquitin tagging, and stabilized the checkpoint protein. Silencing AARS1 had the opposite effect and erased the stabilizing impact of lactate, indicating that this enzyme is the crucial link between tumor metabolism and PD-L1 protection.
From cell tricks to treatment opportunities
The biological consequences of this chemistry were tested in mice. Tumor cells engineered to carry a non-lactylatable version of PD-L1 were more easily killed by CD8+ T cells in culture and formed much smaller tumors in animals, with greater infiltration of these killer cells. The authors then explored sodium lactate (NaLa), a medically used salt of lactate already common in intravenous fluids. In mouse lung cancer models, NaLa alone did not accelerate tumor growth, but when combined with anti-PD-L1 therapy it significantly boosted the treatment’s ability to control tumors and increased the presence of CD8+ T cells inside them. In patient lung cancer samples, higher levels of PD-L1 carrying the lactate tag were associated with more advanced disease and poorer survival, suggesting that this modification could serve as a biomarker for aggressive tumors and possibly for response to immunotherapy.
What this means for patients and future therapies
Taken together, the findings reveal a direct chain of events: tumor metabolism produces lactate; lactate is used to tag PD-L1 at a single spot; this tag blocks the protein’s destruction and helps tumors silence T cells. Yet that same stabilization may render tumors more visible to PD-L1–targeting drugs. In practical terms, this work proposes lactylation—and the AARS1 enzyme that drives it—as new levers to adjust how strongly PD-L1 shields cancer cells. By measuring this tag in patient tumors and designing treatments that fine-tune it, clinicians may one day better predict who will benefit from immunotherapy and pair checkpoint blockers with metabolic interventions to turn cancer’s own fuel waste into a therapeutic weakness.
Citation: Liang, L., Zong, Y., Huang, J. et al. Lactylation stabilizes PD-L1 to promote tumor immune evasion and cell growth. Cell Death Dis 17, 335 (2026). https://doi.org/10.1038/s41419-026-08589-1
Keywords: PD-L1, lactate, tumor immunity, post-translational modification, non-small cell lung cancer