Clear Sky Science · en
Low-dose radiotherapy synergizes with PD-1 blockade to achieve durable survival in advanced NSCLC through antitumor neutrophil programming
Turning the Body’s First Responders Against Lung Cancer
Most people think of radiation and immunotherapy as separate weapons against cancer. This study shows how carefully tuning radiation doses and pairing them with a popular immunotherapy drug can reprogram a surprising ally—neutrophils, a type of white blood cell—to help patients with advanced lung cancer live much longer.
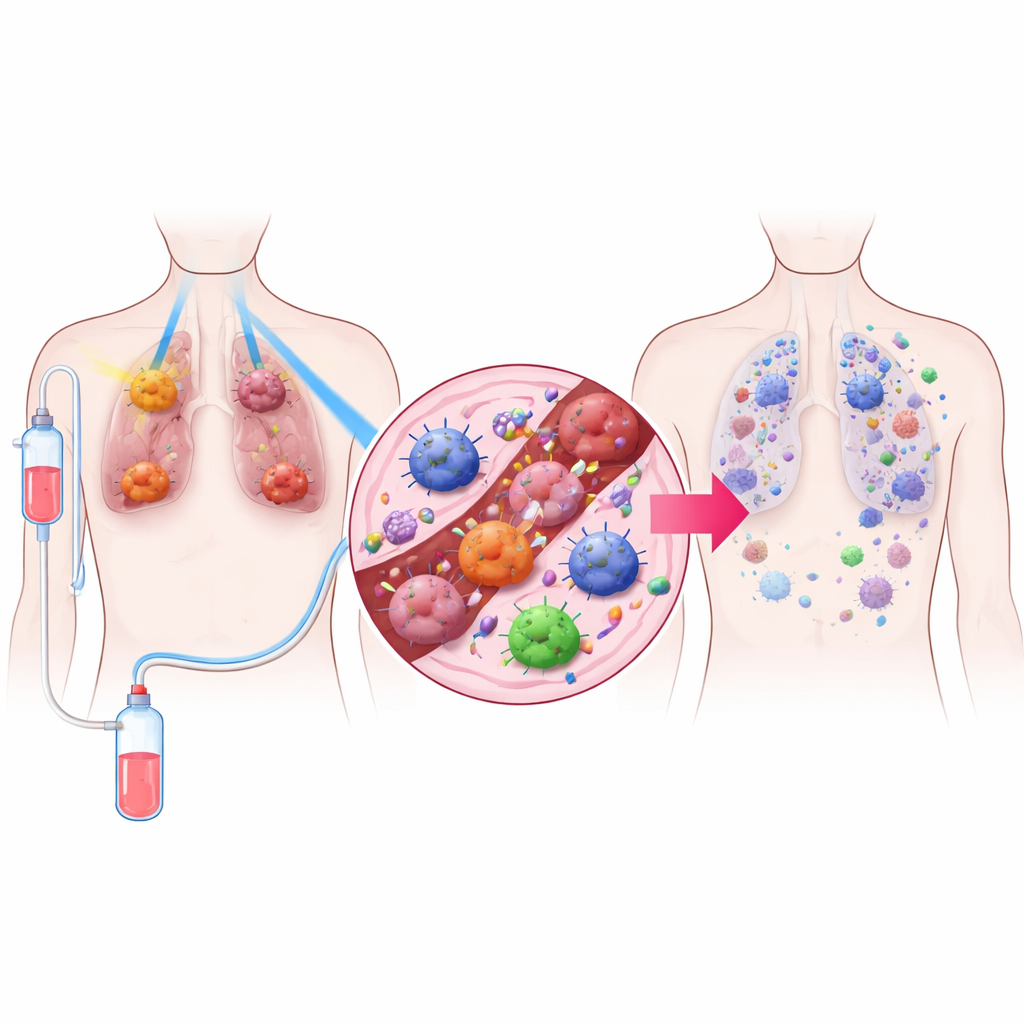
A Three-Part Treatment Strategy
The researchers designed a treatment they call TRIDENT, which combines three elements. First, large lung tumors receive very low doses of radiation that are too weak to kill many cells outright but can change how the tumor environment behaves. Second, smaller tumors are hit with the more familiar high-dose radiation used to destroy cancer locally. Third, patients receive a PD‑1–blocking drug, a kind of immunotherapy that takes the brakes off the immune system’s T cells. By mixing these three parts, the team aimed to both control visible tumors and wake up the body’s immune defenses throughout the body.
Promising Survival in Advanced Lung Cancer
In a phase I clinical trial, 29 people with advanced non-small cell lung cancer who had not received prior treatment and whose tumors made the PD‑L1 protein were given TRIDENT without chemotherapy. With more than five years of follow-up, the median overall survival was 51.3 months—far longer than the roughly 15–23 months usually seen with standard immunotherapy alone or immunotherapy plus chemotherapy. A separate real-world group of 97 patients treated with a TRIDENT-like approach showed similarly encouraging survival. Several patients remained progression-free for more than five years, suggesting that the treatment can spark long-lasting immune memory against the cancer.
How Low-Dose Radiation Helps the Immune System
To understand why this combination works, the team turned to mouse models with tumors on both sides of the body, mimicking spread to multiple sites. Only when low-dose radiation, high-dose radiation, and PD‑1 blockade were used together did tumors shrink not just where high doses were aimed, but also at distant sites, indicating a whole-body immune response. Detailed single-cell analyses revealed that this regimen greatly increased the number of neutrophils entering these distant tumors. Unlike the usual tumor-supporting neutrophils, these cells produced the inflammatory signal TNF‑α and displayed features of professional immune stimulators, including molecules that allow them to show pieces of tumor to T cells and provide “go” signals for attack.
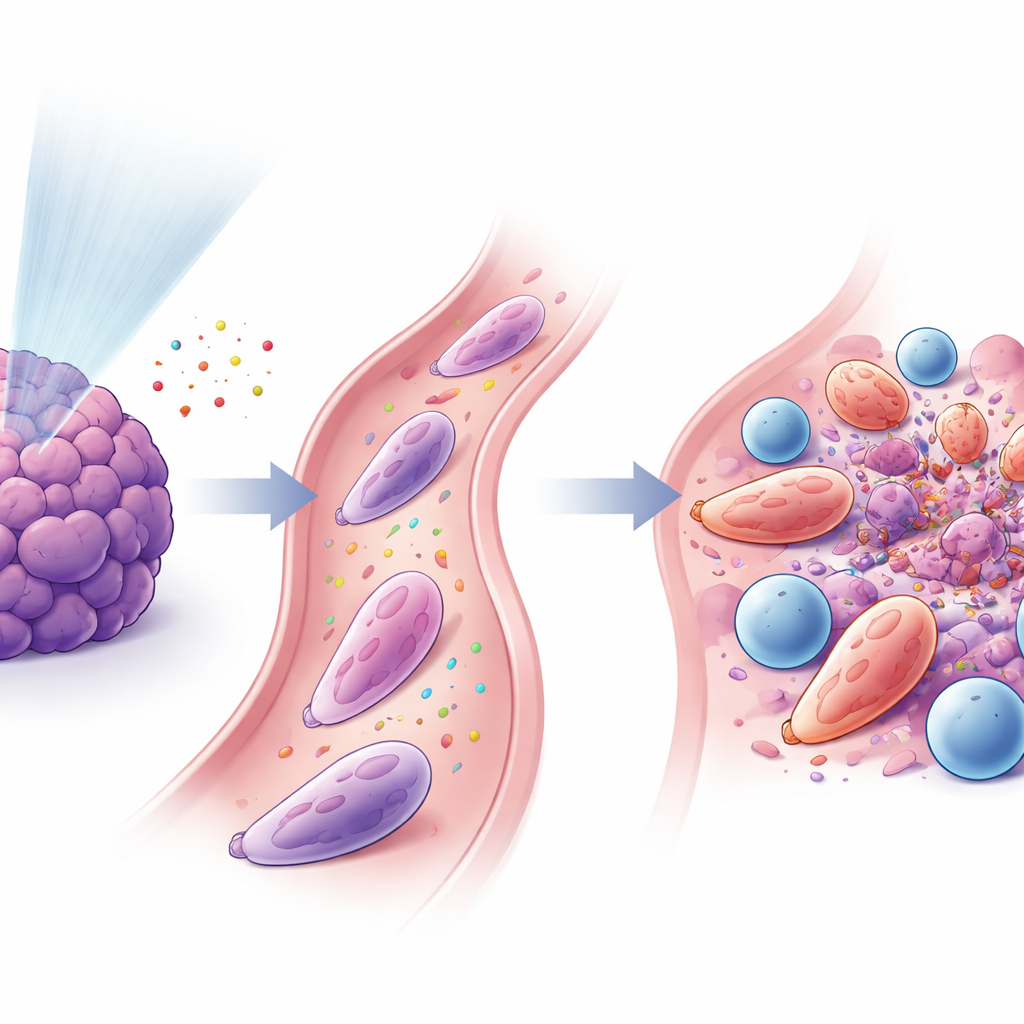
Neutrophils and T Cells Join Forces
The study found that these reprogrammed neutrophils and killer CD8+ T cells were tightly linked. Signals released after low-dose radiation attracted neutrophils through a pathway known as CXCL–CXCR2, and other factors, including GM‑CSF and interferon‑γ, helped convert them into an antitumor state. Once activated, neutrophils expressed sticky surface proteins (such as ICAM‑1) that locked them together with T cells (which carry the matching partner LFA‑1), forming close-contact “immune synapse–like” interfaces. In lab dishes, these neutrophils boosted T‑cell activation and proliferation and helped T cells kill cancer cells more efficiently. When scientists transferred these specialized neutrophils into tumor-bearing mice, especially together with PD‑1 blockade, tumors shrank and survival improved.
Evidence from Patient Tumors and Blood
The team also examined tumor samples from patients before and after TRIDENT using advanced spatial gene-mapping tools. After treatment, tumors showed pockets where these TNF‑α–rich neutrophils and active CD8+ T cells clustered together, along with strong signals related to antigen processing and immune activation. In multiple patient datasets of lung cancer treated with immunotherapy, higher levels of a gene signature linked to these neutrophils were associated with longer time before the cancer worsened. In a small observational group of patients receiving combined radiation and PD‑1 blockade, responders showed clear increases in activated neutrophils in their blood, while non-responders did not.
What This Means for People with Lung Cancer
For someone with advanced lung cancer, these findings suggest that how radiation is given may be just as important as how much is given. By combining small, immune-tuning doses to large tumors with standard high-dose radiation and PD‑1 blockade, TRIDENT appears to turn neutrophils from potential accomplices of the tumor into powerful partners of T cells. This neutrophil–T‑cell teamwork is linked to long-lasting tumor control and better survival, making TRIDENT a promising strategy now being tested in larger randomized trials. The work also points to activated neutrophils themselves as possible future treatment targets and as simple blood-based markers to help predict who is most likely to benefit from radio-immunotherapy.
Citation: Zhou, L., Liu, Y., Xing, Z. et al. Low-dose radiotherapy synergizes with PD-1 blockade to achieve durable survival in advanced NSCLC through antitumor neutrophil programming. Sig Transduct Target Ther 11, 170 (2026). https://doi.org/10.1038/s41392-026-02712-6
Keywords: lung cancer, radiotherapy, immunotherapy, neutrophils, PD-1 blockade