Clear Sky Science · en
Pancreatic ductal adenocarcinoma: integrating molecular insights for targeted interventions
Why this cancer matters to all of us
Pancreatic ductal adenocarcinoma is one of the deadliest common cancers, with most patients living less than two years after diagnosis. Standard treatments like chemotherapy and radiation have made only modest gains, and many of the immunotherapy breakthroughs that transformed melanoma and lung cancer have largely failed here. This review pulls together what scientists have recently learned about the biology of pancreatic tumors and their surroundings, and how that knowledge may finally open the door to more effective, tailored treatments.
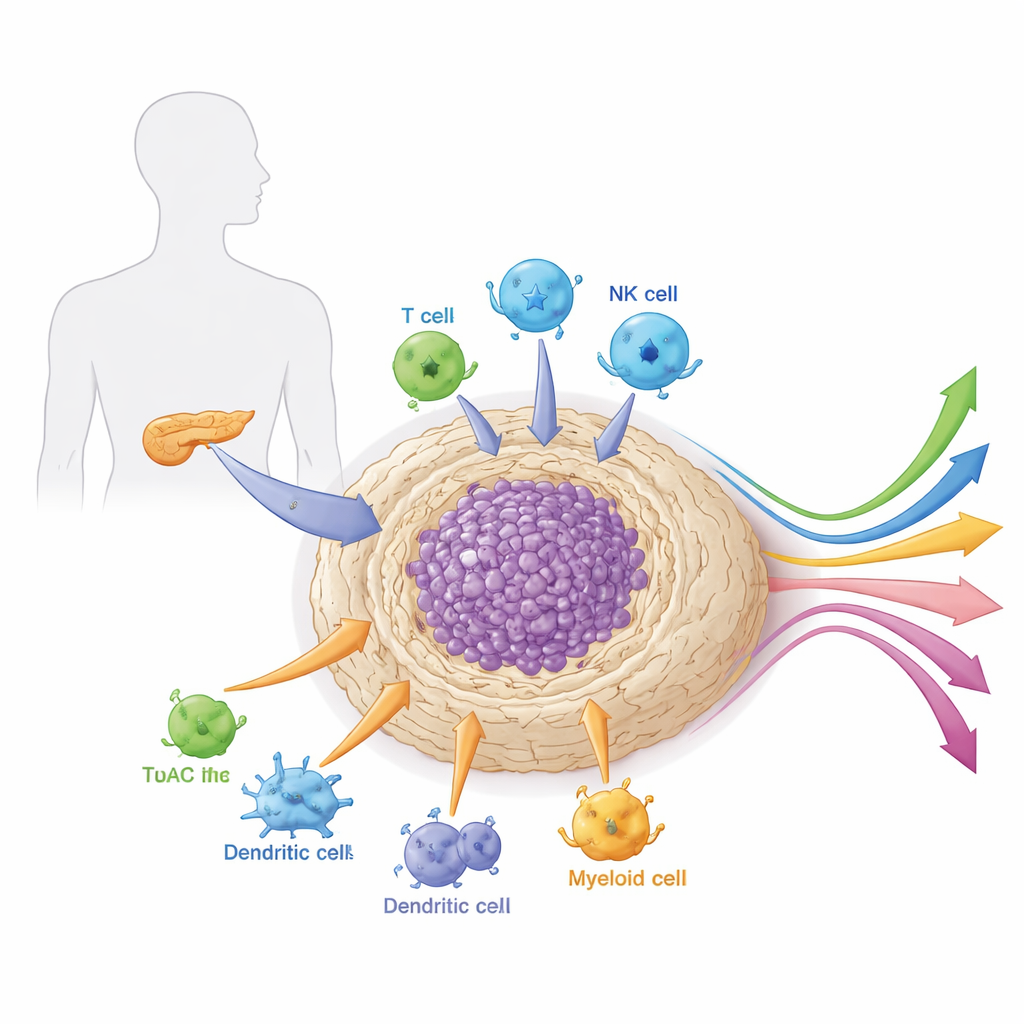
The neighborhood around the tumor
Pancreatic tumors do not grow in isolation. They are embedded in a dense, scar-like tissue called the stroma that is packed with support cells, immune cells, blood vessels, and connective material. This local “neighborhood,” or microenvironment, is unusually tough and immunosuppressive. It acts as both armor and life-support system for the cancer, physically blocking drugs and immune cells while supplying growth signals and nutrients. The article explains how different cell types within this microenvironment—especially cancer-associated fibroblasts, myeloid cells, and specialized lymphocytes—cooperate to shield the tumor from attack and shape how it responds to therapy.
The many faces of support cells
A central focus of the review is a diverse family of cells called cancer-associated fibroblasts. Once thought of as a single type of structural “helper” cell, these fibroblasts are now recognized as multiple subgroups with distinct roles. Some proliferate quickly and build the fibrous shell that stiffens the tumor and blocks drug penetration. Others contract and lay down collagen, further tightening this barrier. Inflammatory fibroblasts secrete clouds of signaling proteins that attract and reprogram immune cells, often pushing them toward tolerance instead of attack. Yet other fibroblasts alter their own metabolism to feed the tumor or even present fragments of tumor proteins to T cells, sometimes supporting immunity and sometimes driving immune suppression. This rich mix means that bluntly eliminating all fibroblasts can backfire; future treatments will need to selectively disarm the harmful subtypes while preserving or even boosting protective ones.
Immune cells turned into unwitting allies
The immune system inside pancreatic tumors is equally complex. Myeloid-derived suppressor cells and tumor-associated macrophages are drawn into the tumor by fibroblast- and cancer-derived signals. Once there, they strip away nutrients needed by killer T cells, release factors that blunt immune activity, and help build new blood vessels that nourish the cancer. Key lymphocyte populations are also reshaped. Regulatory T cells cluster near cytotoxic T cells and dampen their function through checkpoint molecules and metabolic tricks. Dendritic cells, which should prime anti-tumor immunity, are blocked by the stiff stroma and reprogrammed by fatty acid buildup, so they present tumor material poorly. Natural killer cells, normally adept at recognizing stressed cells, are silenced by tumor and stromal signals. Together, these changes turn a potentially hostile immune infiltrate into a tolerant one, explaining why standard checkpoint drugs alone rarely work in pancreatic cancer.
Genes, signals, and why therapies fail
On the genetic and molecular level, pancreatic tumors are driven by a characteristic set of changes, especially mutations in KRAS, TP53, CDKN2A, and SMAD4. Copy-number alterations and epigenetic changes further rewire growth pathways, cell cycle control, and DNA repair. These alterations feed into major signaling cascades—such as the MAPK and PI3K–AKT–mTOR pathways—that drive proliferation and help tumors adapt when one route is blocked. They also influence how cancer cells repair damaged DNA, opening the door to drugs like PARP inhibitors in tumors with specific repair defects. At the same time, dense stroma, abnormal metabolism, and cancer stem-like cells make tumors intrinsically resistant to chemotherapy and targeted drugs, and quick to develop acquired resistance.
New strategies and what comes next
Rather than relying on a single “magic bullet,” the review argues for rational combinations that tackle both the cancer cells and their protective environment. These include pairing checkpoint inhibitors with drugs that remodel fibroblasts or deplete suppressive myeloid cells, engineering T cells or natural killer cells to home into fibrotic tumors, using vaccines and mRNA-based approaches to boost recognition of tumor-specific targets, and exploiting weaknesses in DNA repair or KRAS signaling. Early clinical trials of such combinations, along with sophisticated genetic and immune profiling, are beginning to identify which patients might benefit from which strategy. For a lay reader, the key message is cautiously hopeful: by understanding pancreatic cancer as a complex ecosystem—rather than just a mass of malignant cells—researchers are finally assembling multi-pronged treatment plans that may turn this historically intractable cancer into one that is more responsive to therapy.
Citation: Nagaraju, G.P., Nellipudi, H., Ganji, C. et al. Pancreatic ductal adenocarcinoma: integrating molecular insights for targeted interventions. Sig Transduct Target Ther 11, 165 (2026). https://doi.org/10.1038/s41392-026-02705-5
Keywords: pancreatic cancer, tumor microenvironment, immunotherapy, cancer-associated fibroblasts, KRAS mutations