Clear Sky Science · en
p53: from understanding its structure to advances in therapeutic targeting
Why this guardian protein matters
The protein p53 is often called the “guardian of the genome” because it helps healthy cells repair damage and stops damaged cells from becoming cancerous. This review article explains how p53 works, why it fails so often in cancer, and how scientists are now learning to drug what was long thought to be “undruggable.” It also explores surprising links between p53 and conditions such as Alzheimer’s disease, diabetes, and heart disease, and surveys new treatments that try to fix or bypass p53’s failures.
The cell’s internal safety inspector
Under normal conditions, cells keep p53 levels low through a built‑in recycling system. When stress hits—such as DNA damage, low oxygen, or cancer‑promoting signals—p53 rapidly accumulates and switches on many genes. These genes pause cell division, trigger DNA repair, adjust metabolism, or, if the damage is too great, drive the cell into permanent retirement (senescence) or controlled death (apoptosis). In this way p53 keeps the genetic code stable and prevents damaged cells from multiplying. It also influences newer forms of cell death, like ferroptosis, and shapes the immune environment around tumors, helping immune cells recognize and attack cancerous cells.
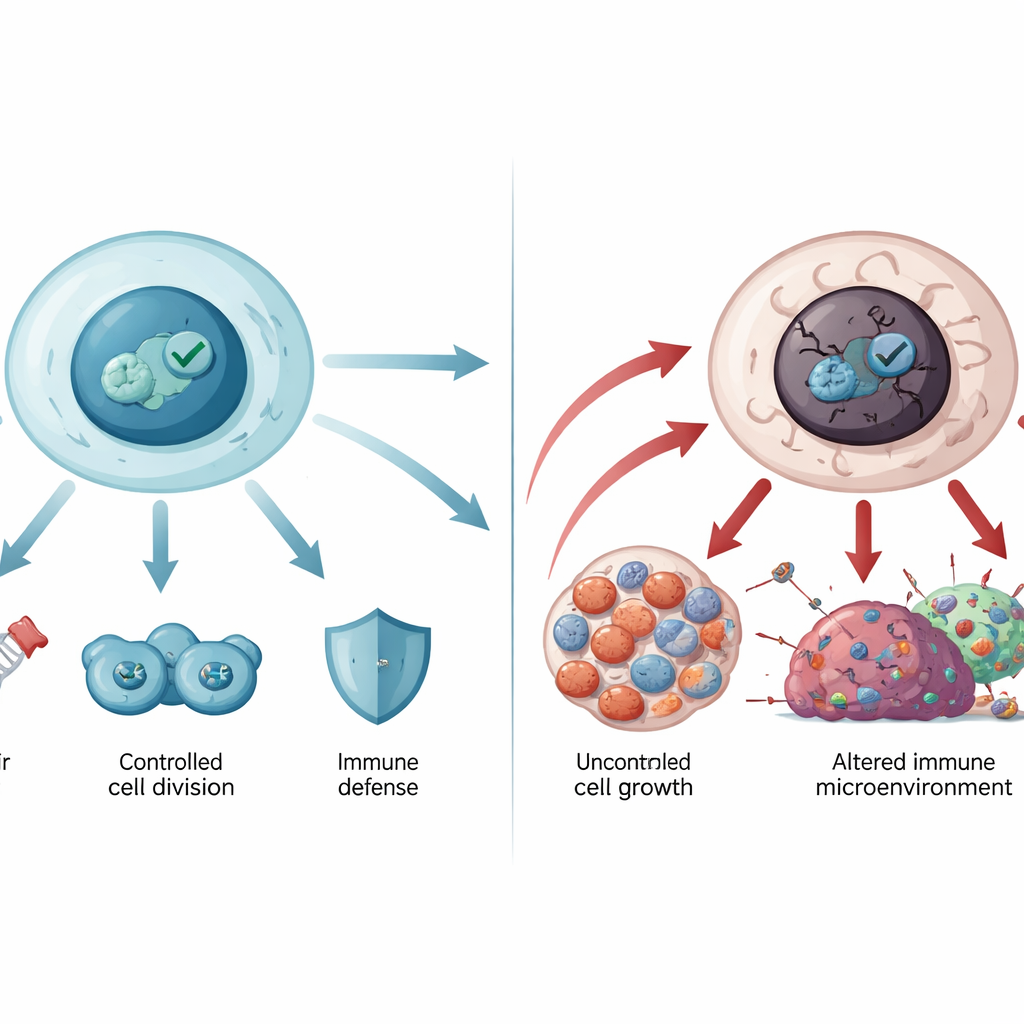
When the guardian goes rogue
The gene that encodes p53, TP53, is the most frequently mutated gene in human cancers. Most of these mutations are single amino‑acid changes in the central DNA‑binding part of the protein. Some mutations weaken p53’s grip on DNA, while others destabilize its structure so it unfolds and clumps into aggregates resembling amyloid, as seen in neurodegenerative diseases. Mutant p53 not only loses its protective role but can also gain new cancer‑promoting activities: it can encourage unchecked growth, help tumor cells resist cell death and chemotherapy, reshape metabolism to feed tumors, and weaken anti‑tumor immunity. Germline TP53 mutations underlie Li‑Fraumeni syndrome and other rare inherited disorders with high cancer risk, showing how crucial this pathway is for lifelong protection.
Beyond cancer: links to brain, metabolism, and heart
Because p53 controls cell death, stress responses, and energy use, its influence extends beyond cancer. In the brain, abnormal p53 activity has been tied to Alzheimer’s, Parkinson’s, Huntington’s disease, ALS, and multiple sclerosis, often through interactions with disease‑related proteins and by promoting excessive loss of vulnerable neurons. In metabolism, p53 helps regulate fat storage, blood sugar, and liver fat; depending on context, it can either protect against obesity and diabetes or contribute to insulin resistance and fatty liver disease. In the heart and blood vessels, p53 affects cell survival, scarring, and plaque formation, playing complex roles in heart failure, diabetic heart damage, and atherosclerosis. These wide‑ranging effects mean that any attempt to boost or block p53 must balance anti‑cancer benefits with potential harm to normal tissues.
New ways to drug the “undruggable”
For decades, p53 was seen as too complex and too central to safely target with drugs. That view is changing. One major strategy aims to “rescue” mutant p53 by stabilizing its shape so it behaves more like the normal protein; examples include PRIMA‑1/APR‑246, arsenic trioxide, and molecules tailored to specific hotspot mutants such as Y220C or R175H. Another approach is to raise levels of normal p53 in tumors that still have an intact TP53 gene by blocking its negative regulators MDM2 and MDM4; several oral inhibitors of these proteins are in clinical trials. A third tactic tries to strip tumor cells of their dependence on mutant p53 by encouraging its degradation using heat‑shock and histone‑deacetylase inhibitors or commonly used statin drugs. In parallel, vaccines, engineered dendritic cells, and highly specific antibodies are being developed to help the immune system recognize cells overloaded with abnormal p53.
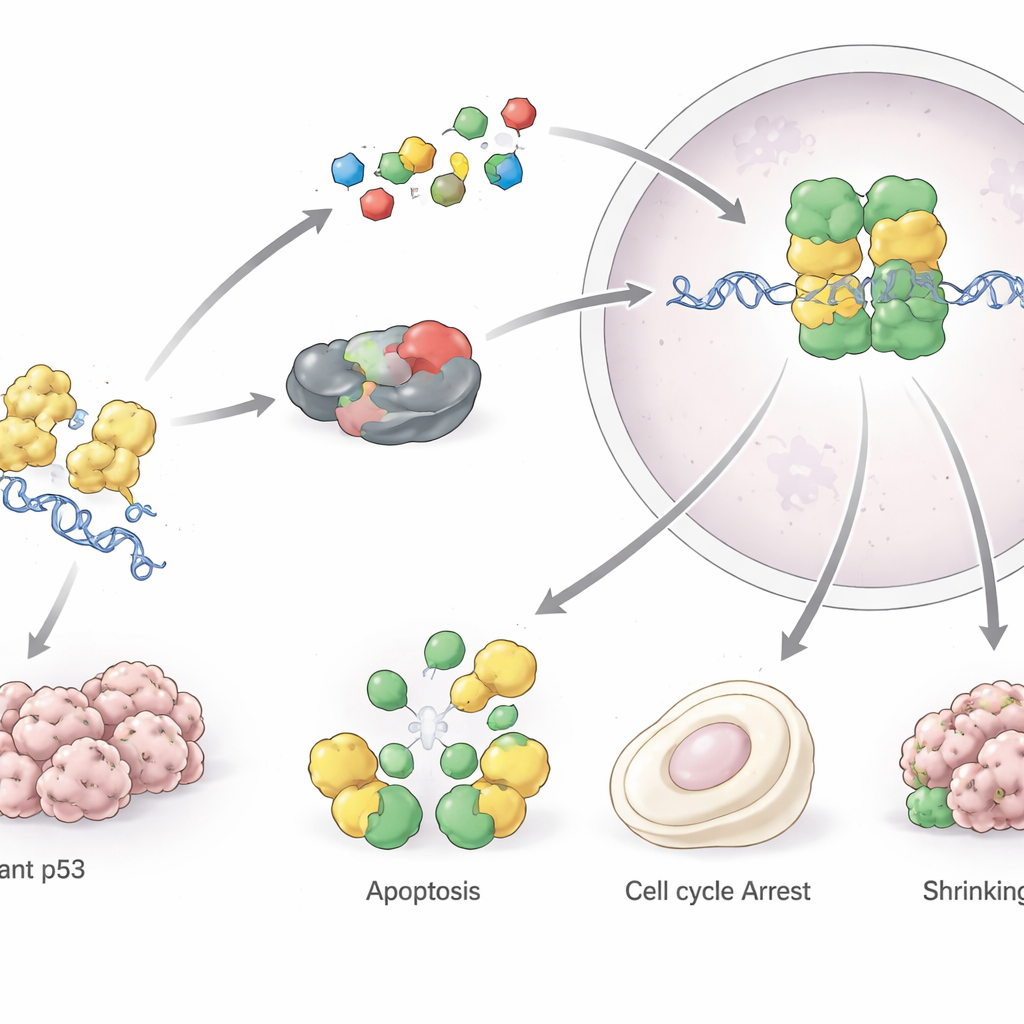
Looking ahead: from maps to medicines
The article concludes that p53 has moved from being a purely biological curiosity to a realistic therapeutic target, but one that demands precision. The diversity of TP53 mutations and the double‑edged nature of p53’s roles in normal tissues make one‑size‑fits‑all treatments unlikely. Instead, the authors envision combinations of small molecules, immunotherapies, and gene‑editing tools guided by systems biology and machine learning to match the right p53‑targeted strategy to each tumor’s wiring. If these efforts succeed, drugs that restore or compensate for p53 function could transform care not only for many cancers, but potentially for some non‑cancer diseases linked to this pivotal guardian protein.
Citation: Wang, W., Liu, X., Liu, H. et al. p53: from understanding its structure to advances in therapeutic targeting. Sig Transduct Target Ther 11, 121 (2026). https://doi.org/10.1038/s41392-025-02549-5
Keywords: p53, TP53 mutations, cancer therapeutics, tumor suppressor pathways, p53-targeted drugs