Clear Sky Science · en
Hypoxia-induced XBP1s-MYDGF axis suppresses ferroptosis through UBQLN1-mediated stabilization of LCN2 in gastric cancer
Why starving tumors of iron matters
Gastric cancer is one of the world’s deadliest cancers, in part because tumor cells become remarkably good at surviving in low‑oxygen, or hypoxic, pockets deep inside the stomach wall. Many new drugs aim to kill cancer cells by triggering a form of iron‑driven cell death called ferroptosis. This study asks a crucial question for future patients: how do hypoxic gastric tumors dodge this iron‑dependent death, and can that escape route be blocked?
A hidden rescue line inside oxygen‑poor tumors
Using large single‑cell RNA sequencing datasets from human stomach tissue, the researchers mapped gene activity cell by cell across the progression from normal lining to full‑blown gastric cancer. They found that malignant epithelial cells switch on a secreted protein called myeloid‑derived growth factor (MYDGF) far more strongly than nearby healthy cells. These MYDGF‑high cancer cells also showed strong signatures of protein handling and disposal systems inside the endoplasmic reticulum, suggesting that MYDGF might be tied to how tumor cells manage the life and death of key proteins.
Turning off iron‑driven cell death
The team then grew human gastric cancer cells under hypoxic conditions that mimic the starved interior of solid tumors. Low oxygen raised MYDGF levels and, as expected, reduced cell survival. When MYDGF was experimentally reduced, hypoxia became much more lethal, especially through ferroptosis: markers of lipid damage and iron‑driven oxidative stress spiked, mitochondria took on a damaged, shrunken appearance, and a potent ferroptosis inhibitor largely rescued the cells. In mice bearing gastric cancer grafts, boosting MYDGF reduced the tumor‑shrinking effect of a ferroptosis‑inducing drug, while blocking a partner protein called LCN2 had the opposite effect, confirming that this pathway operates in living tumors.
How cancer cells shield their iron‑binding guardian
Digging deeper, the authors identified LCN2, an iron‑binding protein, as the critical downstream player. Under hypoxia, LCN2’s protein levels fell even though its gene activity did not, pointing to active destruction rather than reduced production. They showed that LCN2 is normally tagged with ubiquitin and fed into the cell’s protein shredder, the proteasome, via an endoplasmic‑reticulum quality‑control route known as ER‑associated degradation. A transport adaptor called UBQLN1 delivers ubiquitinated LCN2 to this degradation machinery. MYDGF enters as a bodyguard: it binds to UBQLN1 at a specific docking domain, crowding out LCN2 and preventing its handover to the proteasome. As a result, LCN2 protein is stabilized, keeps binding reactive iron, and dampens the lipid peroxidation that drives ferroptosis.
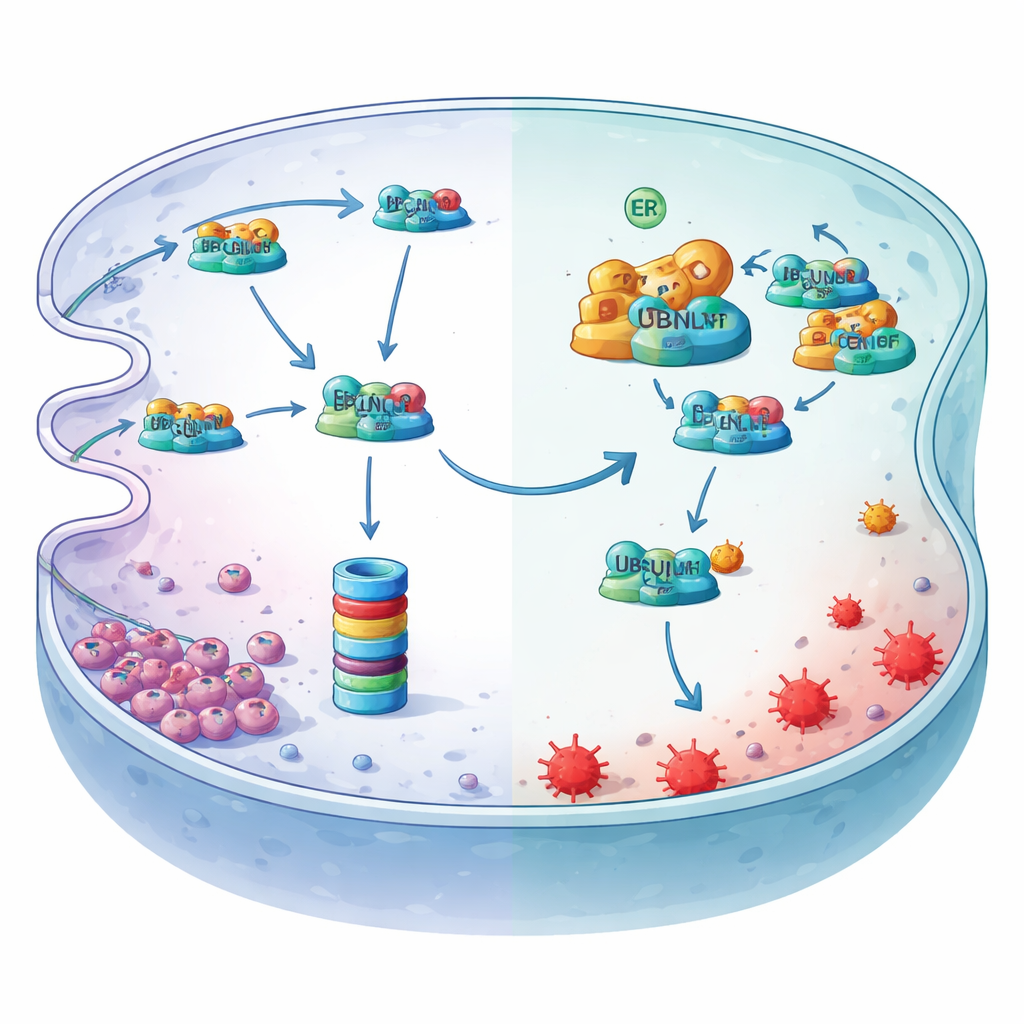
Hypoxia flips the master switch upstream
The study also reveals how hypoxia flips this protective axis on. Low oxygen stresses the endoplasmic reticulum and activates a spliced form of a transcription factor called XBP1s. By scanning the MYDGF gene’s control region and using chromatin‑binding and reporter assays, the researchers showed that XBP1s docks at a specific site and directly cranks up MYDGF production. When XBP1s was reduced, MYDGF levels fell, LCN2 levels dropped, and ferroptosis markers rose—effects that could be undone by restoring MYDGF or LCN2. Together, these experiments outline a linear safeguard: hypoxia activates XBP1s, which boosts MYDGF, which blocks UBQLN1‑driven destruction of LCN2, which then soaks up iron and shields gastric cancer cells from ferroptosis.
New ways to corner stubborn stomach tumors
For non‑experts, the takeaway is that hypoxic gastric tumors have built an internal "emergency line" that keeps an iron‑binding protector protein alive just long enough to avoid a promising new form of cell death. By charting this XBP1s–MYDGF–UBQLN1–LCN2 chain in detail, the work explains why some tumor regions may resist ferroptosis‑based therapies and points to concrete strategies to disarm that defense—such as blocking MYDGF, XBP1s, or the interaction surfaces on UBQLN1. If such interventions can be translated safely to patients, they may help future ferroptosis‑inducing drugs work better in the most treatment‑resistant, oxygen‑starved parts of gastric cancers.
Citation: Zhou, Q., Qi, H., Zou, Y. et al. Hypoxia-induced XBP1s-MYDGF axis suppresses ferroptosis through UBQLN1-mediated stabilization of LCN2 in gastric cancer. Oncogene 45, 1786–1799 (2026). https://doi.org/10.1038/s41388-026-03760-6
Keywords: gastric cancer, hypoxia, ferroptosis, protein degradation, iron metabolism