Clear Sky Science · en
PIAS4 inhibition induces cell cycle arrest and exhibits a synergistic effect in combination with CDK4/6 inhibitor in breast cancer treatment
Why this research matters for patients
Breast cancer is one of the most common and deadly cancers in women, and many patients eventually stop responding to standard drugs that slow down tumor cell division. This study uncovers a previously underappreciated helper protein inside cancer cells, called PIAS4, that quietly pushes cells through a key checkpoint in their growth cycle. By showing that blocking PIAS4 not only slows breast cancer cells but also makes an existing class of drugs more powerful, the work points to a potential new combination therapy that could keep tumors under control for longer.
A hidden switch in breast tumors
The researchers began by comparing tumor samples with normal breast tissue using large-scale cancer databases and patient tissue microarrays. They found that PIAS4 levels were consistently higher in breast tumors than in healthy tissue. Patients whose tumors made more PIAS4 had poorer survival, suggesting that this protein is linked to aggressive disease. Importantly, PIAS4 levels did not strongly track with common diagnostic markers like estrogen, progesterone, or HER2 receptors, indicating that its role may cut across several breast cancer subtypes.
How PIAS4 helps cells race through the cycle
Cancer cells grow by repeatedly passing through a cycle of phases, with an early “G1” checkpoint deciding whether a cell will copy its DNA and divide again. Using cell culture models, the team reduced PIAS4 in breast cancer cells and in mouse cells. Across systems, turning down PIAS4 slowed cell growth and caused many cells to accumulate in the G1 phase, failing to move on to DNA replication. The scientists confirmed this stall using flow cytometry and DNA-labeling assays, which showed fewer cells entering the replication phase when PIAS4 was blocked.
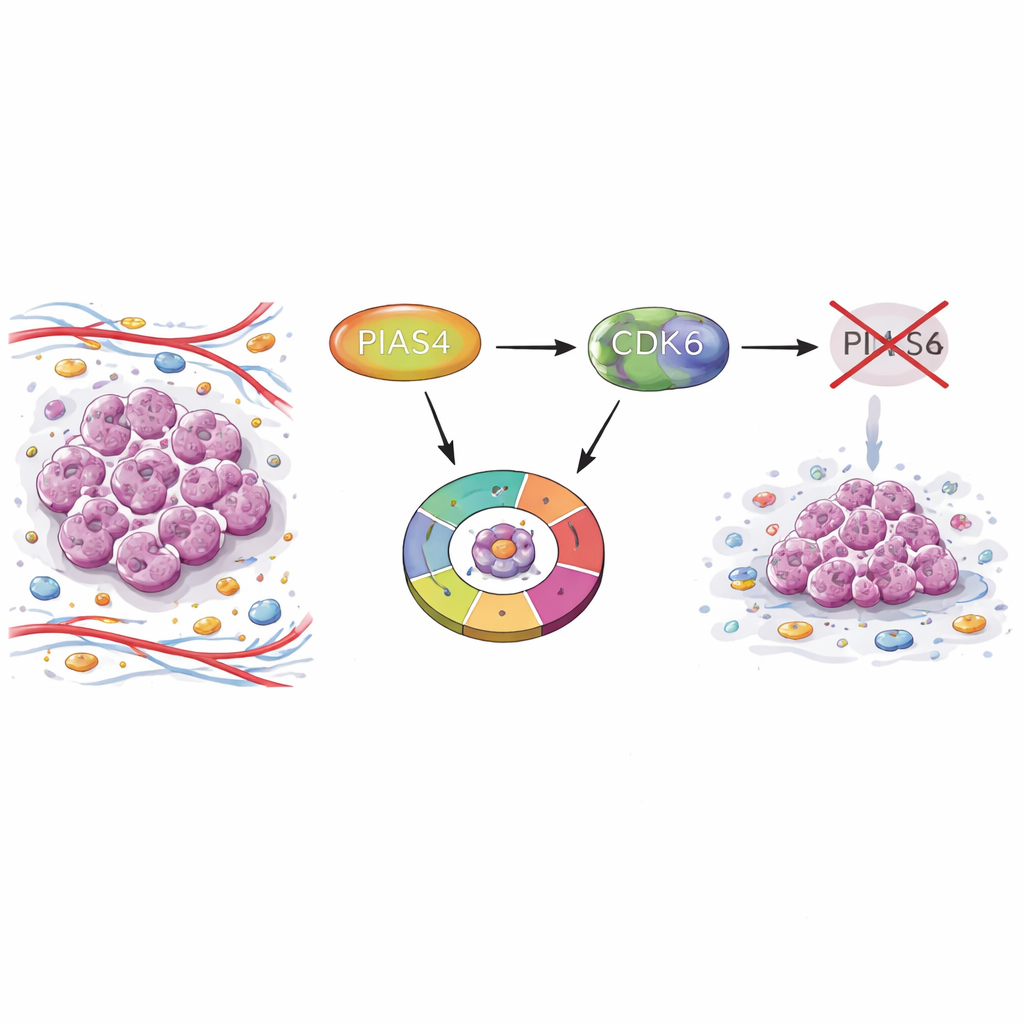
The partnership between PIAS4, CDK6, and the gatekeeper RB1
To understand the mechanism, the team focused on the molecular machinery that drives the G1 checkpoint, especially a protein kinase called CDK6 and its famous target, the tumor suppressor RB1. Normally, active CDK6 adds phosphate groups to RB1, switching it off and freeing a family of transcription factors to turn on genes needed for DNA copying. The researchers showed that PIAS4 physically associates with CDK6 inside the nucleus and chemically decorates CDK6 with small “SUMO” tags. These tags enhance CDK6’s ability to modify RB1 and to promote the expression of cell cycle genes such as CDK1, E2F1, CCNE2, and CCNA2. When PIAS4 was blocked, RB1 phosphorylation fell, RB1 target genes were dialed down, and the cell cycle slowed.
Fine-tuning CDK6: chemical tags and partner proteins
Diving deeper, the team mapped specific sites on CDK6 that receive SUMO tags and created mutant forms of CDK6 that cannot be SUMO-modified. These mutants were much less effective at activating RB1 or pushing cells through G1, confirming that SUMO tagging is not just decorative but functionally important. At the same time, PIAS4 knockdown made CDK6 more stable but paradoxically less active: an inhibitory phosphate mark on CDK6 increased, and its partnership with its activator protein, cyclin D1, weakened. Biochemical assays showed that, despite having more CDK6 protein, cells with low PIAS4 had lower CDK6 enzyme activity, underscoring that how CDK6 is modified and whom it binds to matters more than sheer quantity.
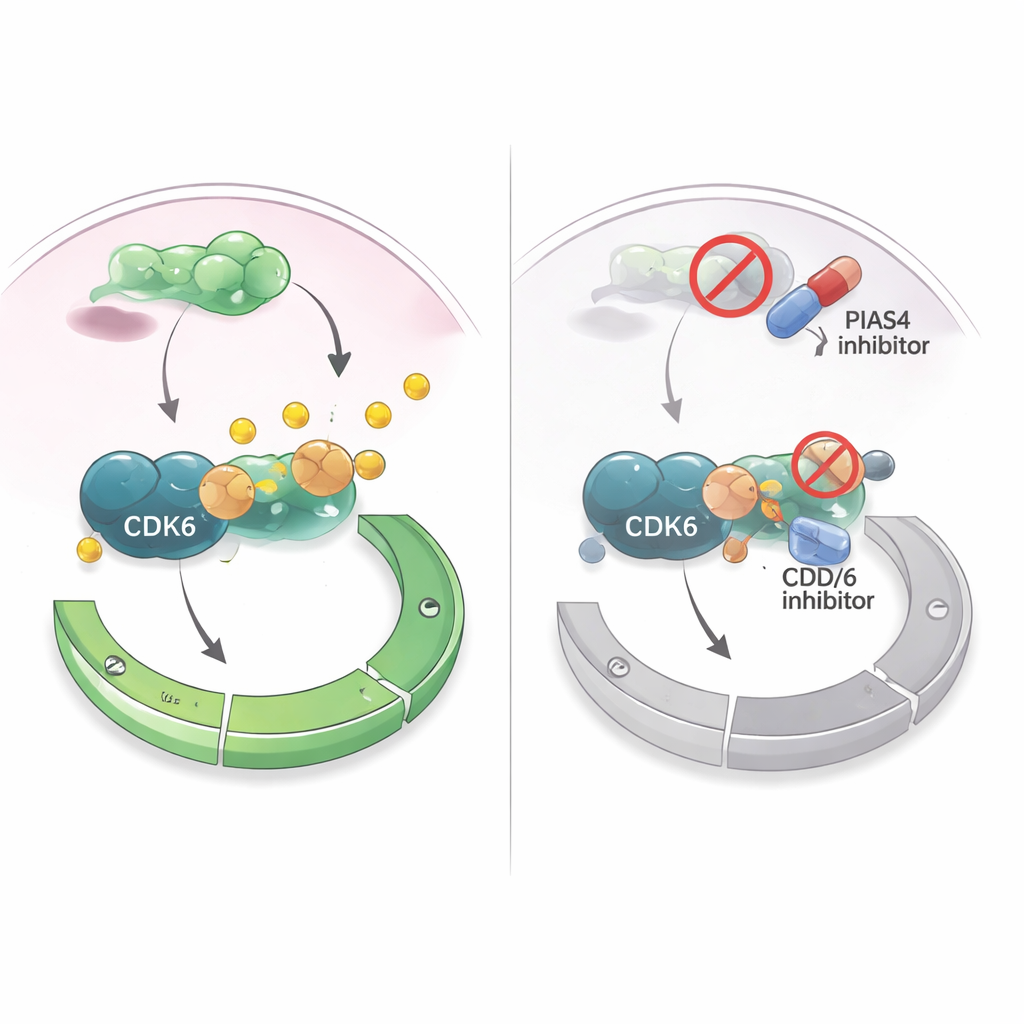
Turning a weakness into a treatment strategy
Because drugs that inhibit CDK4/6 are already used widely in hormone receptor–positive breast cancer, the authors asked whether targeting PIAS4 could boost these treatments. In cultured breast cancer cells, a drug that broadly interferes with SUMO tagging (including the actions of PIAS4) made CDK4/6 inhibitors more effective at suppressing cell growth. In mouse models where breast cancer cells were implanted into mammary tissue, simultaneously reducing PIAS4 and CDK6 shrank tumors more than blocking CDK6 alone, without obvious harm to the animals. Together, the findings suggest that PIAS4 acts as an internal accelerator for CDK6 and the G1 checkpoint, and that disabling this accelerator could sensitize tumors to existing cell-cycle drugs. For patients, this raises the prospect that future therapies combining PIAS4 or SUMO-pathway inhibitors with CDK4/6 inhibitors might better control breast cancer and delay resistance.
Citation: Chen, H., Hu, X., Feng, L. et al. PIAS4 inhibition induces cell cycle arrest and exhibits a synergistic effect in combination with CDK4/6 inhibitor in breast cancer treatment. Oncogene 45, 1756–1770 (2026). https://doi.org/10.1038/s41388-026-03753-5
Keywords: breast cancer, cell cycle, CDK4/6 inhibitors, SUMOylation, PIAS4