Clear Sky Science · en
Metabolic networks in the tumor microenvironment: roles of amino acid and lipid metabolism pathways in cancer progression and therapy
Why the Tumor’s Neighborhood Matters
Cancer does not grow in isolation. It lives in a busy neighborhood of support cells, immune defenders and blood vessels, all sharing and fighting over fuel. This article explores how cancers rewire the use of proteins and fats—broken down into amino acids and lipids—not only inside tumor cells, but across their entire surroundings. Understanding this hidden economy of nutrients helps explain why some tumors escape the immune system and resist treatment, and it points toward smarter, more precise therapies.
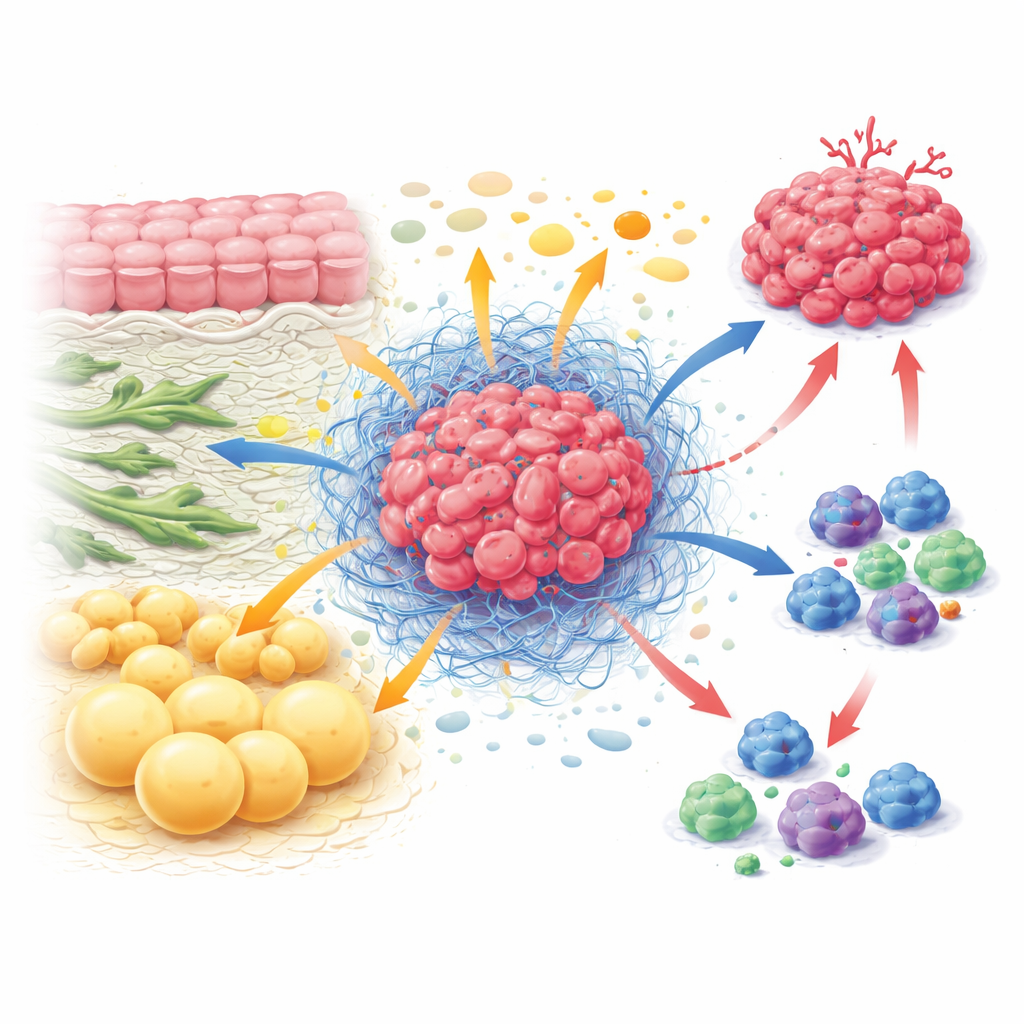
The Hidden Economy Inside a Tumor
Inside a tumor, cancer cells coexist with fibroblasts that shape tissue structure, immune cells that try to attack, blood vessel cells and fat cells. Together they form the tumor microenvironment, a kind of ecosystem with its own rules. Instead of each cell type working alone, they constantly trade and compete for nutrients. Fibroblasts can feed cancer cells with building blocks such as certain amino acids, while also stiffening the surrounding tissue so immune cells cannot easily enter. Immune cells, when active, need large amounts of fuel to divide and kill, yet they often find themselves starved because cancer and stromal cells get priority access. This constant push and pull over nutrients helps tumors grow while weakening the body’s defenses.
Fuel Wars: Amino Acids as Weapons
The review highlights several key amino acids that act as both food and signals. Glutamine, arginine, tryptophan and branched-chain amino acids are crucial for both cancer cells and immune cells. Tumors often boost transport systems and enzymes that let them soak up these nutrients, leaving too little for killer T cells and natural killer cells. At the same time, support cells like myeloid cells and fibroblasts can deliberately break down arginine and tryptophan, generating byproducts that calm immune responses and encourage regulatory T cells that tolerate the tumor. In some niches, cells also share amino acids to help cancer survive treatment, or recycle them from damaged tissue, creating pockets of resistance to chemotherapy and targeted drugs.
Fat as Fuel, Shield and Signal
Lipids—fats, fatty acids, cholesterol and related molecules—play equally important roles in this ecosystem. Cancer cells increase both fat production and fat burning, using fats to build membranes, store energy and resist stress. Nearby fat cells and fibroblasts can release fatty acids that tumors readily import and tuck into lipid droplets, tiny storage depots that protect against toxic stress and many drugs. Immune cells in the tumor are also pushed toward heavy reliance on fat metabolism. Regulatory T cells and certain macrophages use fatty acids to survive and remain suppressive, while overloaded killer T cells become exhausted. Special lipid messengers, such as lysophosphatidic acid, further stimulate tumor growth, guide cancer spread to distant organs and repel immune cells from the tumor core.
Changing Over Space, Time and Treatment
Metabolism in a tumor is not uniform. Oxygen and nutrient levels vary from the well-fed outer rim to the starved core, and different organs that host metastases offer distinct nutrient mixes. As a result, some regions favor sugar burning, others rely more on glutamine or fats, and these patterns shift as the tumor grows or faces therapy. When treatments cut off one fuel, tumors often switch to another or rely more on help from fibroblasts and fat cells. The same metabolic shifts can also alter how genes are turned on or off through epigenetic marks, locking in aggressive or immune-suppressive states. This flexibility makes it hard to stop cancer by blocking a single pathway.
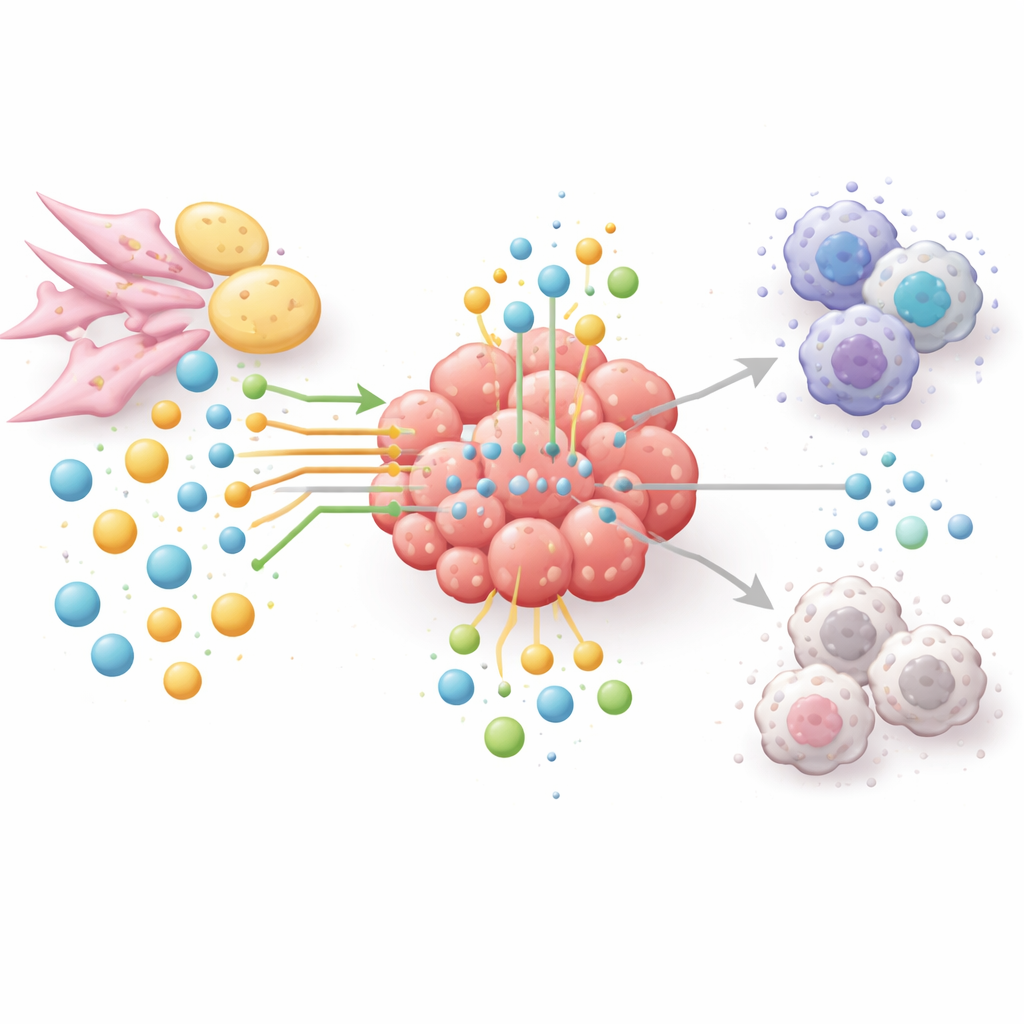
New Ways to Starve the Tumor, Not the Patient
Because amino acid and lipid pathways link tumor growth, immune escape and treatment resistance, the authors argue for a network-based approach rather than one-drug-for-one-enzyme solutions. Experimental therapies now aim to lower arginine or glutamine in carefully chosen tumors, block fat-making or fat-burning enzymes, or shut down tryptophan breakdown and its immune-dampening products. Many of these strategies are being combined with immune checkpoint drugs to both weaken the tumor and re-energize T cells. To choose the right combinations and patients, the field is turning to spatial metabolomics, liquid biopsies and artificial intelligence to map who is eating what, where and when inside tumors. In simple terms, the article concludes that winning against cancer will require not only attacking tumor cells directly, but also rewiring the entire nutrient network that shelters them.
Citation: Sung, Y., Kim, D.K., Kim, J.S. et al. Metabolic networks in the tumor microenvironment: roles of amino acid and lipid metabolism pathways in cancer progression and therapy. Exp Mol Med 58, 1128–1148 (2026). https://doi.org/10.1038/s12276-026-01697-0
Keywords: tumor microenvironment, cancer metabolism, amino acids, lipid metabolism, immunotherapy