Clear Sky Science · en
Asymmetry and redundancy of STAT5 paralogs across CD8+ T cell differentiation states
Why this matters for future immune therapies
Many of today’s most promising cancer and autoimmune treatments work by nudging our own immune cells, especially CD8+ “killer” T cells, to fight harder or stand down. A key molecular switch in these cells is a protein called STAT5, which actually comes in two nearly twin versions. This study asks a deceptively simple question with big therapeutic implications: are these twins truly interchangeable, or does one quietly run the show?
Two similar switches that behave differently
Our genomes carry two closely related copies, or paralogs, of STAT5: STAT5A and STAT5B. Because they look so alike at the molecular level and bind many of the same stretches of DNA, they have often been treated as redundant parts. Yet patients with damaging STAT5B mutations develop serious immune problems, while comparable STAT5A defects are rare. Using a suite of mouse models that systematically remove STAT5A, STAT5B, or both in different combinations, the authors probe how each paralog shapes CD8+ T cell numbers and behavior in living organisms and cells.
Key role for STAT5B in keeping killer T cells balanced
When total STAT5 levels were dialed down, CD8+ T cells dropped sharply in lymph nodes, spleen, and bone marrow, leading to skewed ratios of helper (CD4+) to killer (CD8+) T cells. Mixed bone marrow transplants showed that this is largely due to a problem inside the T cells themselves, not just changes in their environment. The loss was most severe when STAT5B was missing, and mice relying on a single STAT5A copy fared worst. At the same time, the remaining CD8+ T cells shifted away from a resting, naive state toward more experienced effector and memory states, but paradoxically could not fully arm themselves with key killing tools such as specific enzymes and receptors. This reveals STAT5 as a guardian of both the numbers and quality of killer T cells, with STAT5B carrying the greatest weight.
Unequal partners under the hood
Digging deeper, the team measured how much of each paralog is present and how genes respond when either one is missing. In CD8+ T cells, STAT5B made up roughly two-thirds of the total STAT5 protein and message. Loss of STAT5B reduced activation of the pathway’s key phosphorylation step more than loss of STAT5A. When the researchers sequenced RNA from naive, effector, and memory CD8+ T cells stimulated by two cytokines, IL-7 and IL-15, they found far more genes misregulated in the absence of STAT5B than when only STAT5A was lacking. Many of these genes control survival, metabolism, and memory formation, underscoring STAT5B as the main driver of cytokine responses in killer T cells.
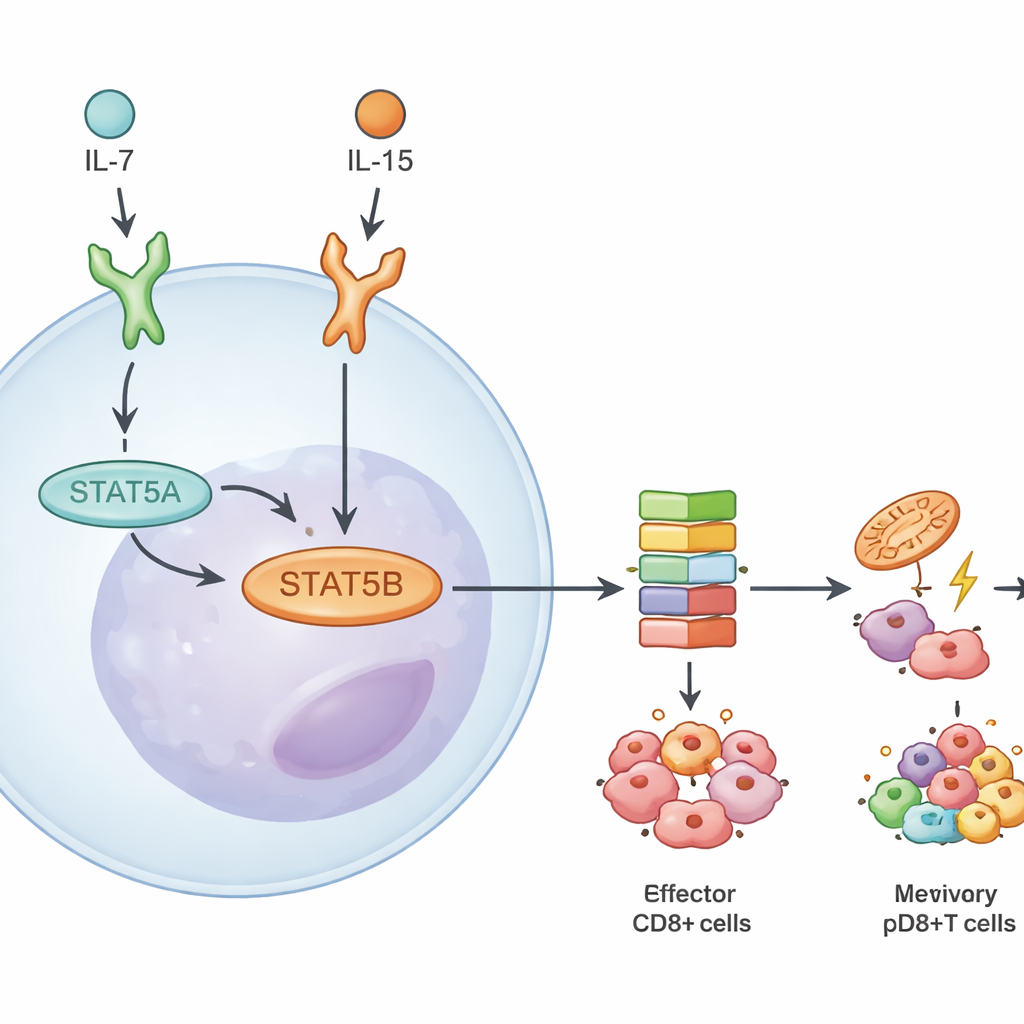
Shared DNA targets but context-specific actions
Despite STAT5B’s dominance, the two paralogs turned out to occupy much of the same genomic real estate. Using binding maps, the authors saw that most high-impact DNA sites were bound by both STAT5A and STAT5B. When they reintroduced an always-on form of STAT5A into deficient cells, it largely boosted genes that were also reachable by STAT5B, suggesting true STAT5A-only targets are rare and relatively weak. Instead, differences arose from how strongly and in what situations STAT5B acts. Responses varied depending on which cytokine signaled (IL-7 vs. IL-15), whether the T cell was naive or memory, and whether it belonged to the CD4+ or CD8+ lineage. IL-15, for example, triggered broader STAT5B-driven gene programs than IL-7, especially in memory CD8+ T cells.
A gene signature to track STAT5 activity in disease
To capture the essence of this pathway, the investigators distilled an 85-gene “core STAT5 signature” that appeared across different cytokines and CD8+ T cell states. When they overlaid this signature onto single-cell RNA data from virus-infected mice, it lit up a distinct cluster of early effector or early exhausted CD8+ T cells, the very cells experiencing strong, fresh cytokine stimulation. The signature was far less active in fully exhausted or resting cells, suggesting it can act as a bioinformatic beacon for intense STAT5 signaling during immune responses and perhaps in cancers driven by aberrant STAT5B activity.
What this means for patients and therapies
For a non-specialist, the take-home message is that STAT5A and STAT5B are like two levers on the same control panel, but one lever, STAT5B, is longer and pulled more often. Both contribute to how killer T cells survive, rest, and remember, yet STAT5B has a uniquely strong influence on their numbers and on critical gene programs. By clarifying this asymmetry, and by offering a gene-based signature of STAT5 activity, the study provides a conceptual map for designing more precise immunotherapies—whether by tuning cytokine drugs, engineering T cells, or interpreting patient genomic data—that harness the power of killer T cells without tipping the immune system into disease.
Citation: Ristin, S., Dalzell, M., Armstrong, C. et al. Asymmetry and redundancy of STAT5 paralogs across CD8+ T cell differentiation states. Commun Biol 9, 529 (2026). https://doi.org/10.1038/s42003-026-09999-9
Keywords: STAT5B, CD8 T cells, cytokine signaling, immunotherapy, T cell differentiation