Clear Sky Science · en
Combining AI to reveal CCDC3-mediated pathways of colorectal cancer liver metastasis
Why this research matters
Colorectal cancer is one of the most common cancers worldwide, and many deaths occur only after the disease has spread to the liver. Doctors know that tumors with highly unstable chromosomes tend to be more aggressive and harder to treat, but it has been unclear how the surrounding non-cancerous cells help push tumors into this dangerous state. This study uncovers a hidden line of communication between support cells around the tumor and cancer cells, revealing a chain of events that drives genetic chaos and fuels liver metastases—and pointing to new ways to stop it.
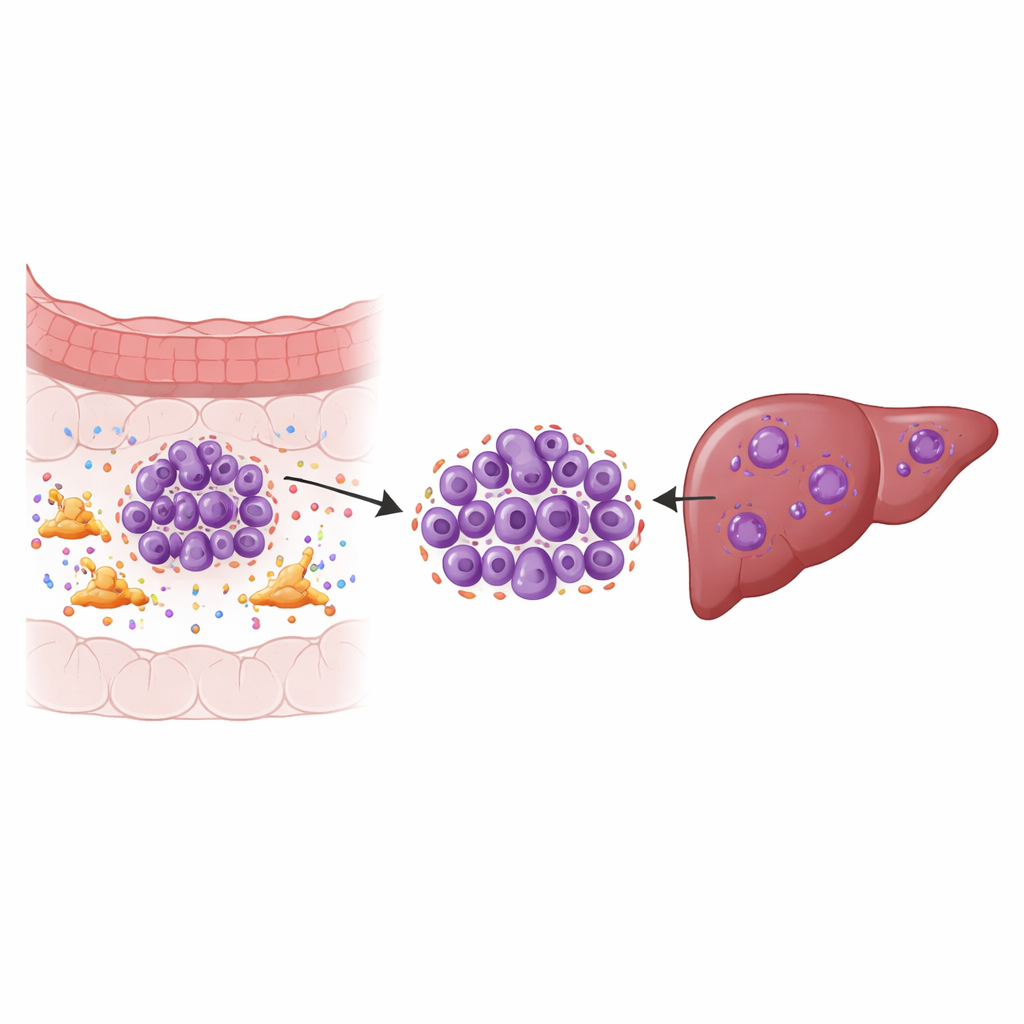
The hidden helpers around the tumor
Solid tumors are not just clumps of cancer cells; they are ecosystems. One key resident of this ecosystem is the cancer-associated fibroblast, a type of connective-tissue cell that remodels the area around the tumor and secretes many signaling molecules. Using single-cell RNA sequencing on samples from primary colorectal tumors, matching liver metastases, nearby normal tissues, and blood, the researchers mapped nine major cell types and then zoomed in on the cancer cells themselves. They discovered specific tumor subgroups in the liver that showed strong signs of chromosomal instability—frequent errors in how chromosomes are copied and divided—which is linked to rapid growth, treatment resistance, and poor survival.
A numerical score for dangerous tumor cells
To understand which cancers are most at risk, the team built a composite “instability index” that captured multiple layers of evidence: gene expression patterns, inferred changes in chromosome copy number, and known instability-related genes. Tumor cells with high scores were far more common in liver metastases than in primary tumors. These high-index cells showed an overactive cell cycle, more DNA damage and repair activity, and features associated with invasion and spread. In other words, the more unstable the chromosomes, the more aggressive and adaptable the cancer cells appeared—supporting the idea that instability is not just a side effect of cancer, but a driver of its progression.
A secret signal from fibroblasts
The next question was what in the tumor environment might be pushing cancer cells toward this unstable state. By analyzing how different cell types "talk" to each other through signaling molecules, the researchers found especially strong communication between fibroblasts and the chromosomally unstable tumor cells. A particular group of fibroblasts was enriched in liver metastases and linked to worse patient survival. When the genes active in these fibroblasts were compared with those in instability-high cancer cells, one stood out: CCDC3, a little-studied protein that was produced mainly by fibroblasts and was much more abundant in liver lesions than in primary tumors. Patients whose tumors had higher levels of CCDC3 tended to live for a shorter time, suggesting this signal might be a key promoter of aggressive disease.
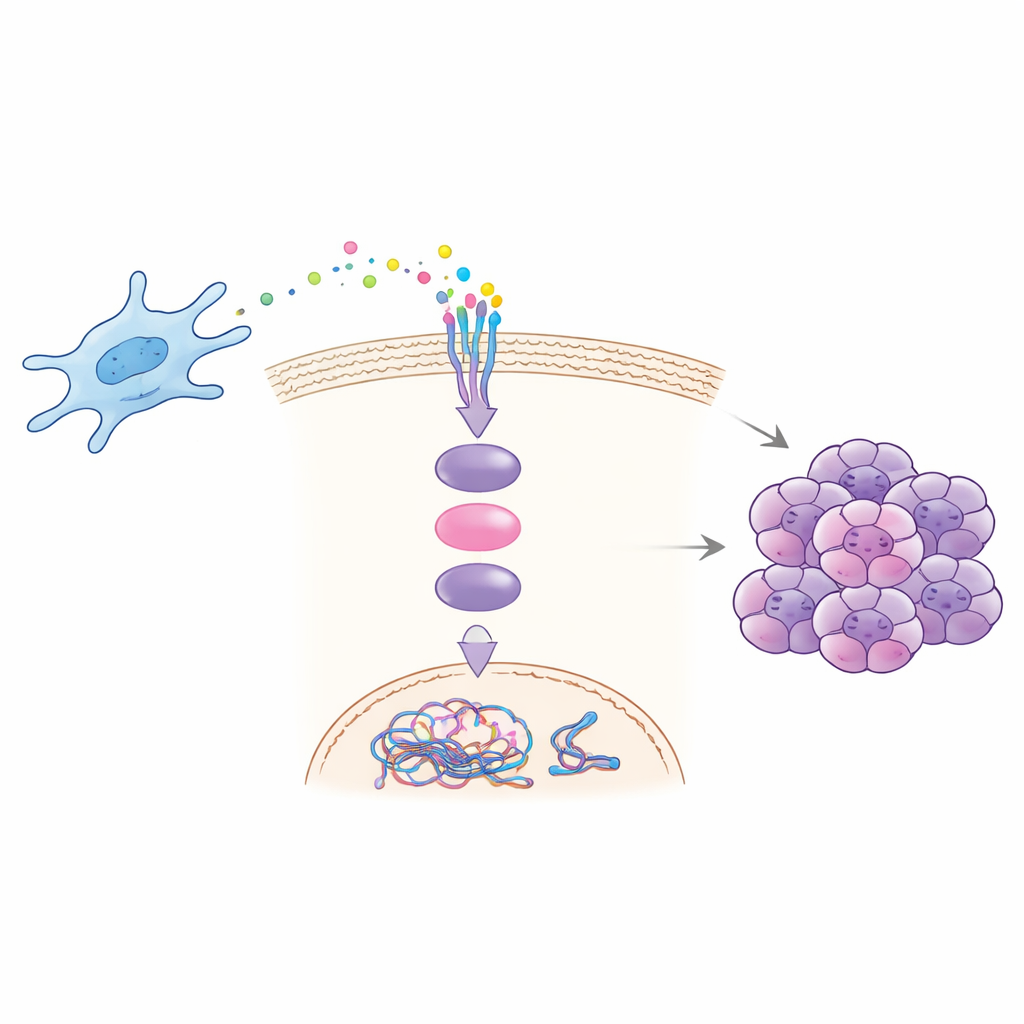
How one signal triggers a chain reaction
Laboratory and animal experiments revealed how CCDC3 drives malignancy. Fibroblasts taken from liver metastases secreted more CCDC3 and, when grown with cancer cells, boosted the cancer cells’ ability to migrate, invade, form colonies, and display signs of chromosomal instability. Adding purified CCDC3 to cancer cells reproduced these effects, while blocking CCDC3 in fibroblasts reduced them. The team showed that CCDC3 binds to a receptor called CXCR3 on cancer cells, which then activates a well-known messenger protein inside the cell, STAT3. Once switched on, STAT3 travels to the nucleus and directly increases production of CDT1, a protein that helps license DNA replication. Excess CDT1 disrupts the normal timing and accuracy of DNA copying, promoting chromosome errors and instability.
From mechanism to potential treatment
By selectively blocking each step of this chain—CCDC3, CXCR3, STAT3, or CDT1—the researchers were able to blunt the cancer cells’ invasive behavior and growth in dishes and in mouse models. Mice given extra CCDC3 developed larger tumors and more liver metastases, whereas those treated with an antibody against CCDC3 or a STAT3 inhibitor had slower tumor growth and fewer liver lesions. These findings define a clear fibroblast-to-tumor signaling route that links the tumor environment to internal genetic chaos in cancer cells. For patients, this work suggests that targeting the CCDC3–CXCR3–STAT3–CDT1 axis, or using CCDC3 levels as a warning sign of aggressive disease, could one day help prevent or treat liver metastases from colorectal cancer.
Citation: Huang, R., Liu, Q., Jin, X. et al. Combining AI to reveal CCDC3-mediated pathways of colorectal cancer liver metastasis. npj Digit. Med. 9, 327 (2026). https://doi.org/10.1038/s41746-026-02457-0
Keywords: colorectal cancer, liver metastasis, tumor microenvironment, chromosomal instability, cancer-associated fibroblasts