Clear Sky Science · en
Multi-omics profiling reveals tumor microenvironment characteristics linked to immunotherapy response and prognosis in non-small cell lung cancer
Why this study matters for people with lung cancer
Many people with advanced lung cancer now receive immunotherapy drugs that help the immune system attack tumors. Yet a large number of patients either do not benefit or see their cancer return. This study asks a simple but crucial question: what is different inside tumors that respond to treatment compared with those that do not, and can that knowledge guide better care for patients with non-small cell lung cancer?
Looking inside tumors one cell at a time
The researchers combined several powerful genetic tools to build a detailed map of lung tumors from patients treated with immunotherapy, often together with chemotherapy. Instead of averaging signals across the whole tumor, they analyzed individual cells, identifying immune cells, cancer cells and support cells. By comparing tumors that shrank deeply after treatment with those that did not, they found that successful responses were tightly linked to the presence of a particular group of killer T cells and to how friendly or hostile the surrounding tumor microenvironment was to immune attack.
A special group of killer T cells linked to better outcomes
Among all the immune cells they studied, one subset of CD8 T cells stood out. These cells were marked by high levels of a gene called ZNF683. Tumors from patients who responded well to immunotherapy were rich in these ZNF683-positive CD8 T cells, while non-responding tumors had far fewer of them. The team showed, across many different cancer types and large patient collections, that higher ZNF683 levels usually went hand in hand with more immune cell presence and better survival. They then built a risk score, based on 19 genes related to this T cell group, that could sort lung adenocarcinoma patients into lower-risk, immune "hot" tumors and higher-risk, immune "cold" tumors.
Cold tumors, a suppressive signal, and a new target
High-risk tumors, as defined by this ZNFRS score, had fewer helpful immune cells and more cancer and myeloid cells, a pattern typical of "cold" tumors that resist immunotherapy. When the researchers examined how cells in these tumors talked to each other, they found that many signals flowed from myeloid and cancer cells toward T cells and natural killer cells. A key player was SPP1, a secreted protein that traveled from myeloid or cancer cells to receptors on immune cells. This SPP1 signaling network was strong in non-responding and high-risk tumors, but weak or absent in responding and low-risk tumors, suggesting that SPP1 helps create an immune-suppressive niche that keeps killer T cells away or blunts their activity.
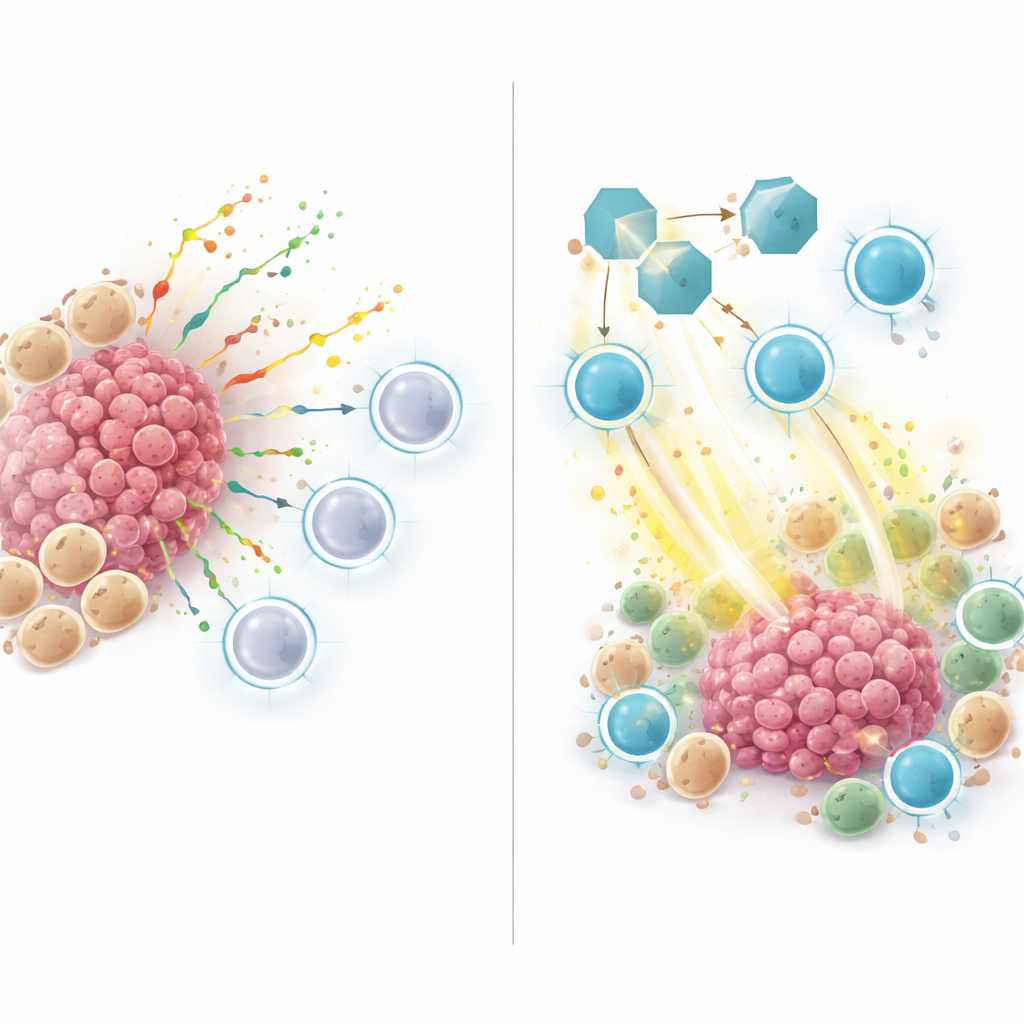
Testing SPP1 blocking in animal models
To move beyond patterns in human data, the team turned to mouse models of lung cancer. They treated tumor-bearing mice with an antibody that blocked SPP1, with or without an anti-PD-1 immunotherapy drug. Blocking SPP1 alone slowed tumor growth, and the combination with anti-PD-1 worked best of all. Under the microscope and by single-cell analysis, tumors from SPP1-blocked mice contained more active CD8 T cells producing toxic molecules that kill cancer cells, and fewer of the so-called M2-like macrophages that usually promote tumor growth. In other words, inhibiting SPP1 shifted the microenvironment from a suppressive state toward an immune-activated one.
What this means for future treatments
For lay readers, the main message is that not all immune cells in a tumor are equal, and that the surrounding neighborhood can either welcome or repel them. This study pinpoints a special type of killer T cell linked to successful immunotherapy in lung cancer and builds a gene-based score that reflects how warm or cold the tumor’s immune climate is. It also highlights SPP1 as a key harmful signal that helps tumors hide from the immune system. By blocking SPP1 in mice, researchers were able to wake up killer T cells and make standard immunotherapy work better. While more clinical testing is needed, these findings outline a path toward blood and tissue tests that identify who is most likely to benefit from immunotherapy and suggest combination treatments that could turn resistant lung tumors into ones the immune system can attack more effectively.
Citation: Zhang, L., Zeng, J., Wen, J. et al. Multi-omics profiling reveals tumor microenvironment characteristics linked to immunotherapy response and prognosis in non-small cell lung cancer. npj Precis. Onc. 10, 179 (2026). https://doi.org/10.1038/s41698-026-01474-2
Keywords: non-small cell lung cancer, immunotherapy, tumor microenvironment, CD8 T cells, SPP1 signaling