Clear Sky Science · en
Synthesis, biological evaluation and molecular docking studies of N-propylsulfonyl indole-linked hydrazinecarbothioamides as selective ecto-5′-nucleotidase and NTPDase inhibitors
Why this research matters for future cancer treatments
Our immune system constantly patrols for cancer cells, but tumors have evolved clever tricks to switch those defenses off. One of those tricks uses chemical messengers made from ATP, the same molecule that powers our cells. This paper describes a new family of small molecules designed to interrupt that chemical trick, with the long‑term goal of helping the immune system better recognize and attack tumors.
The chemical switch that calms immune cells
Cells talk to each other using many signals, and one important channel is based on nucleotides such as ATP and its breakdown product, adenosine. Outside the cell, a chain of enzymes sitting in the cell membrane steadily converts ATP into adenosine. Two key players, known as CD39 and CD73, are often overproduced in oxygen‑poor tumor tissue. There, they rapidly transform ATP released by stressed or dying cells into adenosine, which acts like a chemical "tranquilizer" on nearby immune cells. High adenosine levels dampen T‑cell activity, promote blood vessel growth, and help tumors spread. Blocking the enzymes that create adenosine is therefore an attractive strategy in modern cancer immunotherapy.
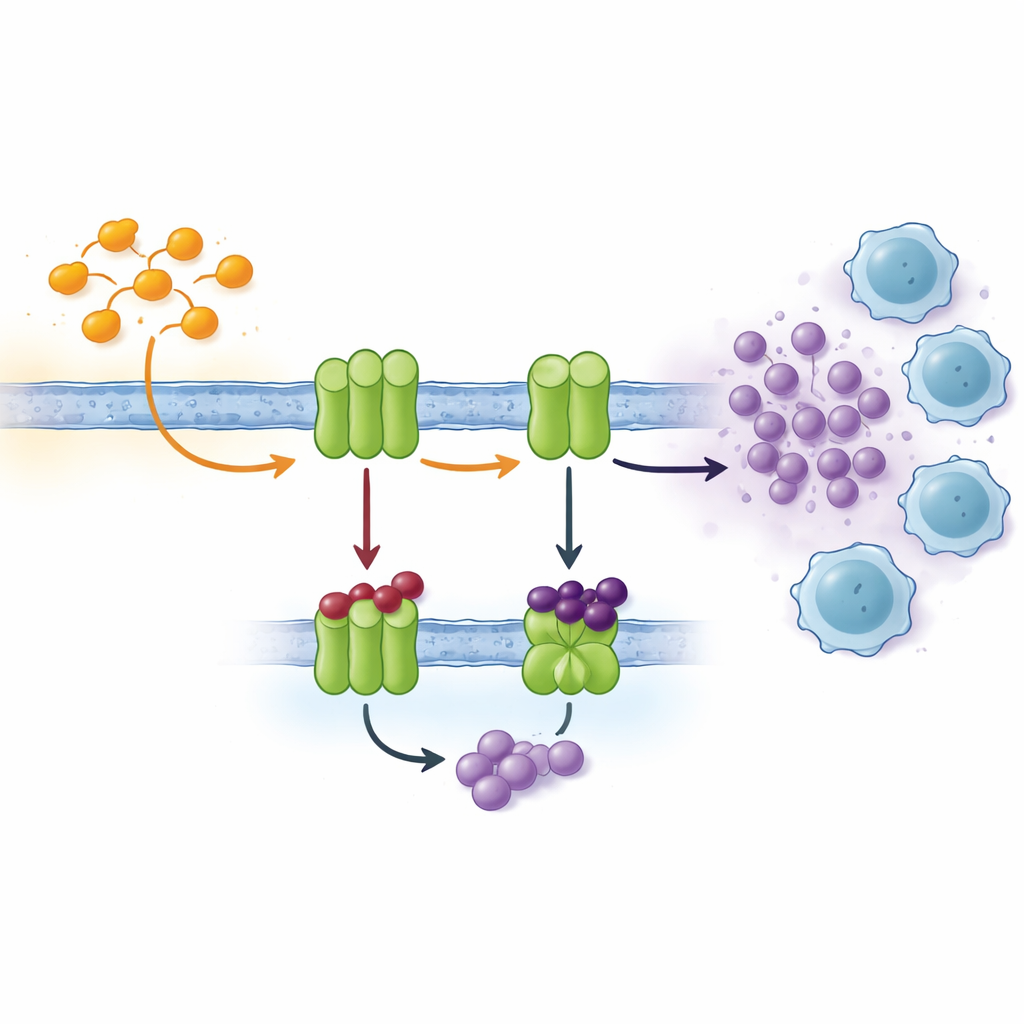
Designing smarter blockers using a proven ring system
Drug designers often start from ring systems that have already shown medical value. The indole ring, found in several approved anticancer drugs, is one such structure. Another useful component, thiosemicarbazone, can bind metals and interacts strongly with many biological targets. In this work, the researchers combined these two motifs into a single hybrid scaffold and fine‑tuned it by attaching different chemical groups, creating a small library of 22 related compounds. They changed where on the indole ring the thiosemicarbazone unit sits and varied the attached side groups to see how these changes affected the ability to block CD73 and several related enzymes called NTPDases.
Testing which molecules hit which enzyme
The team first confirmed the structure and purity of all new molecules using standard laboratory techniques such as infrared and nuclear magnetic resonance spectroscopy, mass spectrometry, and high‑performance liquid chromatography. They then measured how well each compound slowed down five human enzymes in test‑tube assays: CD73 and four NTPDase types (1, 2, 3 and 8). Several members of the series blocked their targets at low micromolar concentrations, meaning they were far more potent than commonly used reference inhibitors. Different chemical decorations on the scaffold steered each compound toward particular enzyme types. For example, one molecule labeled 5f was especially effective against NTPDase2 and NTPDase8, while 5i favored NTPDase1. Two related molecules, 5n and 5o, stood out for their strong action on CD73 and selected NTPDases, marking them as especially promising lead candidates.
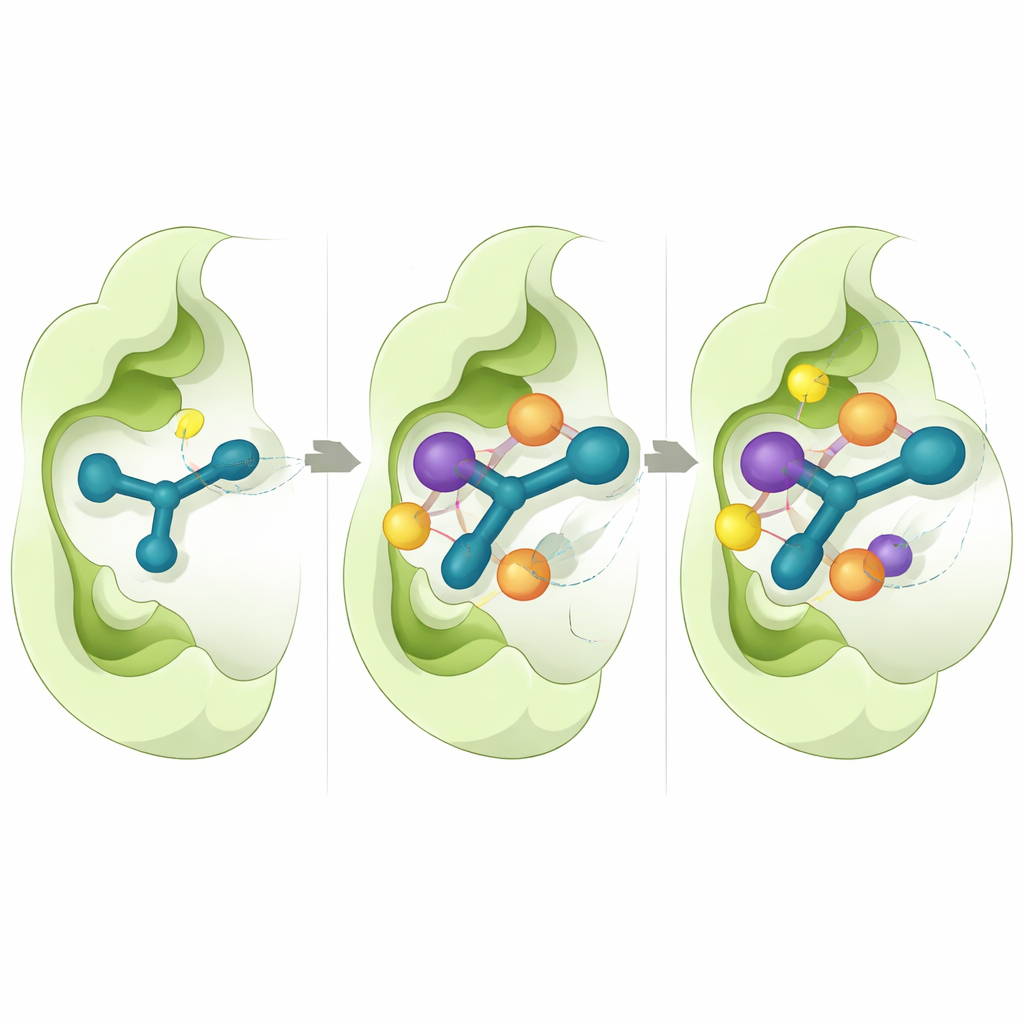
Peering into the binding pocket on a computer
To understand why some molecules worked better than others, the researchers used detailed computer simulations. They focused on CD73, because a high‑resolution three‑dimensional structure of this enzyme is available. Virtual docking experiments showed that the active compounds slip into the enzyme’s catalytic pocket in a consistent way, where the indole ring stacks against aromatic amino acids and the thiosemicarbazone segment forms hydrogen bonds with key residues. Calculations of binding energy suggested that snug hydrophobic contacts and van der Waals forces are major contributors to the strong binding seen for 5n and 5o. Long molecular‑dynamics simulations, each running the motion of atoms over half a microsecond, indicated that these complexes remain stable, with only small fluctuations in both the protein and the inhibitor positions over time.
Early hints of drug‑like behavior
Beyond hitting their targets, potential drugs must move through the body in a suitable way. The authors therefore predicted absorption, distribution, and other pharmacokinetic properties in silico. Most compounds in the series, and especially 5n and 5o, showed a favorable balance of size, solubility, and membrane permeability, with good predicted oral uptake and only minor rule‑of‑thumb violations. These data, combined with their strong and selective enzyme inhibition and stable binding in simulations, support viewing 5n and 5o as starting points for further optimization rather than finished drug candidates.
What this could mean for patients
When tumors use CD39 and CD73 to flood their surroundings with adenosine, immune cells become sluggish and less able to attack. This study introduces a new set of indole‑based molecules that selectively interfere with that pathway in the test tube and behave encouragingly in computer models. While much work remains—including tests in cells, animals, and eventually humans—the findings suggest that this chemical scaffold could be shaped into future medicines that help lift the chemical "brakes" tumors place on the immune system, potentially making existing cancer therapies more effective.
Citation: Batool, Z., Dutt, S.M., al-Rashida, M. et al. Synthesis, biological evaluation and molecular docking studies of N-propylsulfonyl indole-linked hydrazinecarbothioamides as selective ecto-5′-nucleotidase and NTPDase inhibitors. Sci Rep 16, 14419 (2026). https://doi.org/10.1038/s41598-026-50728-3
Keywords: ecto-5-nucleotidase inhibition, NTPDase inhibitors, adenosine signaling, cancer immunotherapy, indole thiosemicarbazones