Clear Sky Science · en
CircFN1 modulates IL-1β-induced osteoarthritis through the miR-19b-3p/PTEN signaling axis
Why Aching Joints Matter
Osteoarthritis is often dismissed as “wear and tear,” but for millions of people it means daily pain, stiffness, and loss of independence. This study looks under the hood of sore joints and asks a timely question: why do some knees and hips deteriorate so badly, and could we slow that process by changing how certain tiny RNA molecules behave inside cartilage cells?
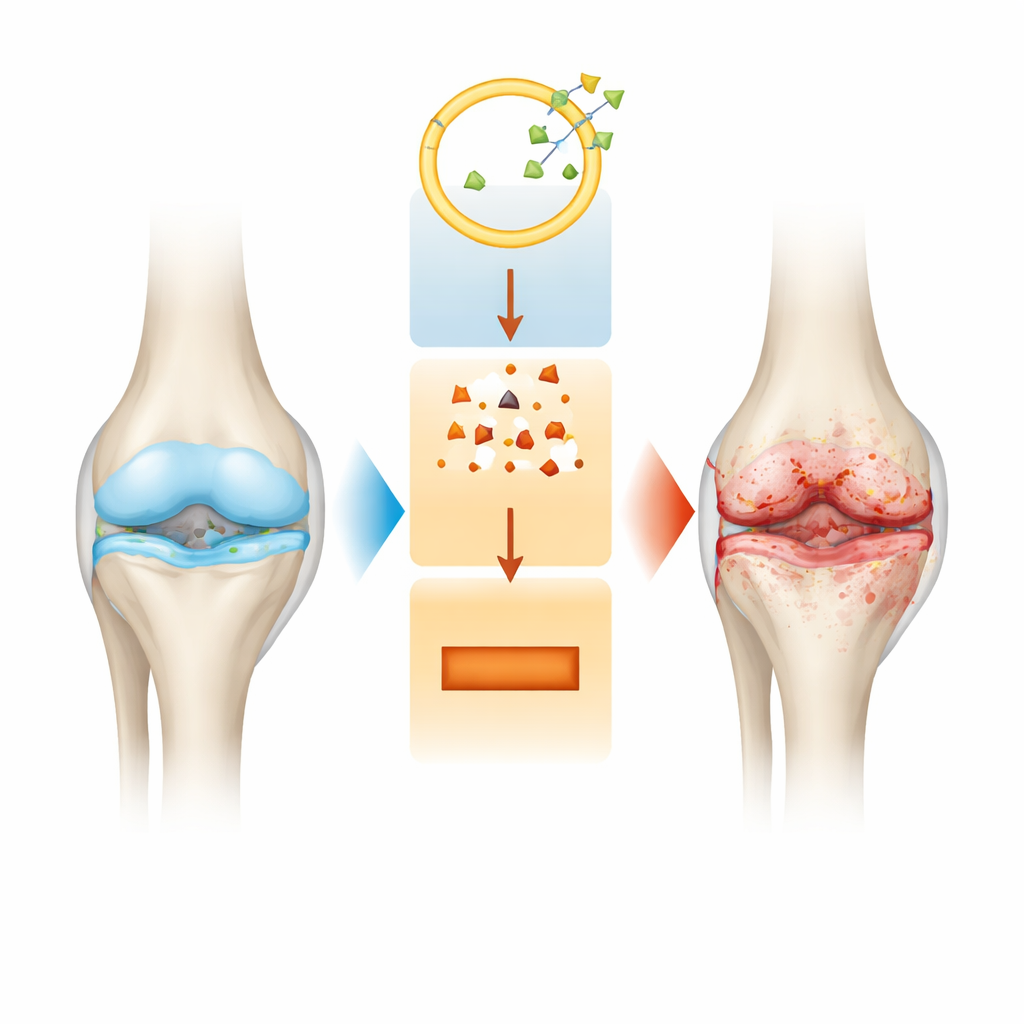
A Closer Look at Joint Breakdown
Osteoarthritis is more than thinning cartilage. As the disease advances, the whole joint changes: the smooth cushion on the ends of bones erodes, the lining of the joint becomes inflamed, and the underlying bone remodels in ways that can worsen pain. Current treatments mainly ease symptoms but do not halt this slow structural damage. Researchers therefore search for molecular switches inside cartilage cells, called chondrocytes, that might explain why these cells start dying and why the supporting matrix around them breaks down.
The Hidden World of Circular RNA
In recent years, scientists have discovered that many stretches of our genetic material never become proteins but still play powerful regulatory roles. Among them are circular RNAs—looped molecules that are unusually stable and often abundant in specific tissues. The team behind this paper focused on one such molecule, circFN1, found at higher levels in human cartilage cells exposed to the inflammatory signal IL‑1β, a chemical commonly used to mimic osteoarthritis in the lab. Sequencing experiments comparing normal and IL‑1β‑treated chondrocytes showed that circFN1 was one of the most strongly increased circular RNAs, making it a prime suspect in fueling joint damage.
A Three-Step Molecular Tug-of-War
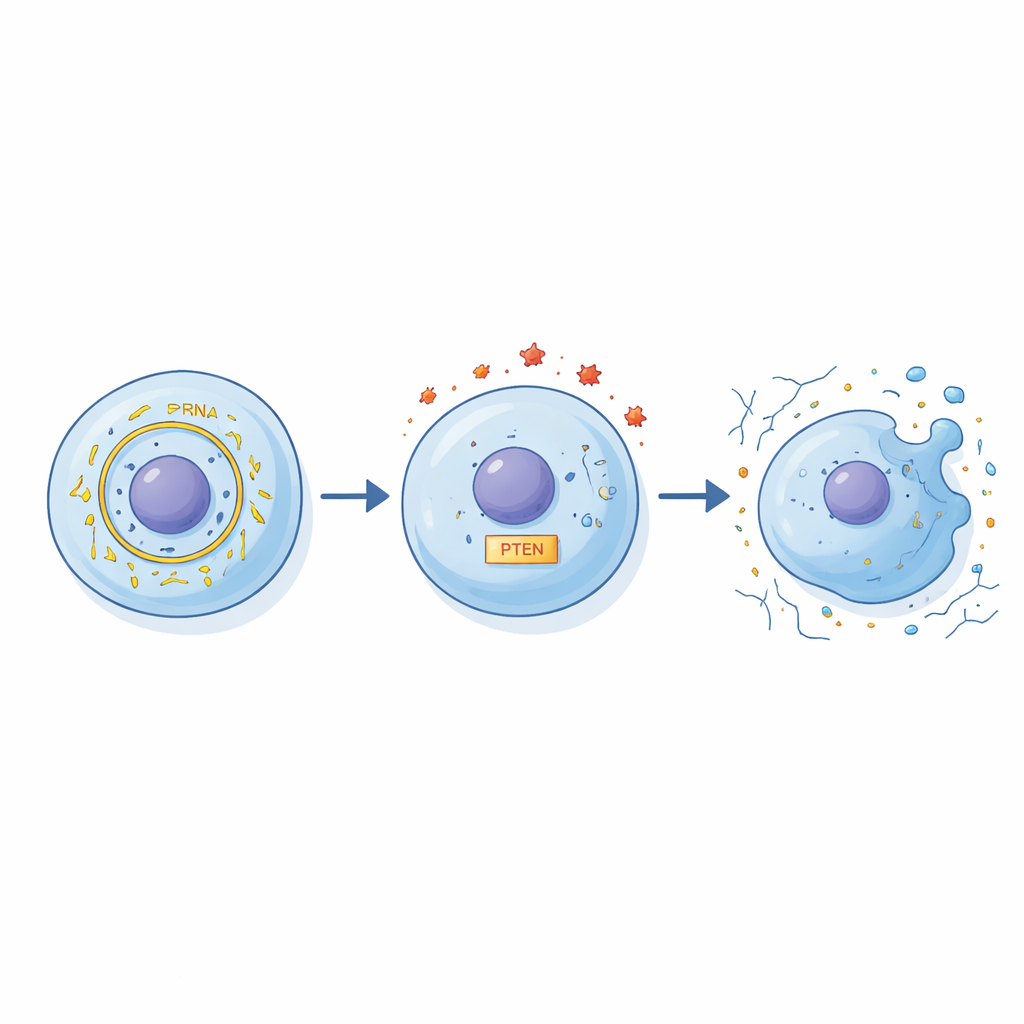
The researchers uncovered a chain of interactions linking circFN1 to cell injury. First, they showed that circFN1 can act like a sponge for a small regulatory RNA called miR‑19b‑3p, binding and sequestering it. Normally, miR‑19b‑3p helps control the levels of PTEN, a protein that influences cell survival, inflammation, and matrix maintenance. When circFN1 levels rise, more miR‑19b‑3p is soaked up, which in turn alters PTEN’s activity and disrupts the balance between protective and harmful signals in the cell. Through a series of genetic “dial up and dial down” experiments, the authors demonstrated that this circFN1–miR‑19b‑3p–PTEN chain strongly shapes how chondrocytes respond to inflammatory stress.
What Happens When the Switches Are Flipped
To test the functional impact of this pathway, the team deliberately reduced circFN1 in IL‑1β‑treated chondrocytes. When circFN1 was lowered, more cells survived, fewer underwent programmed death, and key building blocks of cartilage, such as the matrix protein aggrecan, were better preserved. At the same time, molecules that chew up cartilage, like MMP‑13, dropped, and the surrounding “soup” of inflammatory substances such as TNF‑α and IL‑6 became less intense. When the researchers also altered miR‑19b‑3p and PTEN, these protective changes strengthened or weakened in predictable ways, reinforcing the idea that all three partners form a connected regulatory circuit rather than acting in isolation.
From Cell Dishes to Future Therapies
Although these experiments were performed in cultured human cartilage cells rather than in live joints, the findings point toward a new conceptual lever for osteoarthritis treatment. The study suggests that elevated circFN1 amplifies inflammation‑driven cartilage damage by rearranging the balance between miR‑19b‑3p and PTEN. In simple terms, a surplus of this circular RNA makes cartilage cells more vulnerable to inflammatory insults and more prone to die and shed their supporting matrix. If future work in animals and patients confirms these results, measuring circFN1 could help detect early joint damage, and therapies designed to block circFN1 or boost miR‑19b‑3p might one day slow the slide from occasional stiffness to chronic, disabling osteoarthritis.
Citation: Jiang, Y., Wang, Q., Yang, X. et al. CircFN1 modulates IL-1β-induced osteoarthritis through the miR-19b-3p/PTEN signaling axis. Sci Rep 16, 10710 (2026). https://doi.org/10.1038/s41598-026-45897-0
Keywords: osteoarthritis, cartilage, noncoding RNA, inflammation, joint degeneration