Clear Sky Science · en
Limosilactobacillus reuteri metabolites modulate immune pathways and intestinal barrier repair after 5 fluorouracil exposure
Why protecting the gut during cancer treatment matters
Chemotherapy saves lives, but it often punishes the lining of the mouth and gut, causing pain, diarrhea, and infections that can interrupt or weaken treatment. This study explores whether harmless bacteria, or more precisely the molecules they release, can help the gut wall repair itself after being injured by a common cancer drug. The work focuses on a well-known probiotic strain, Limosilactobacillus reuteri DSM 17938, and asks: can its secreted sugars and tiny particles support healing without needing to give live bacteria to vulnerable patients?
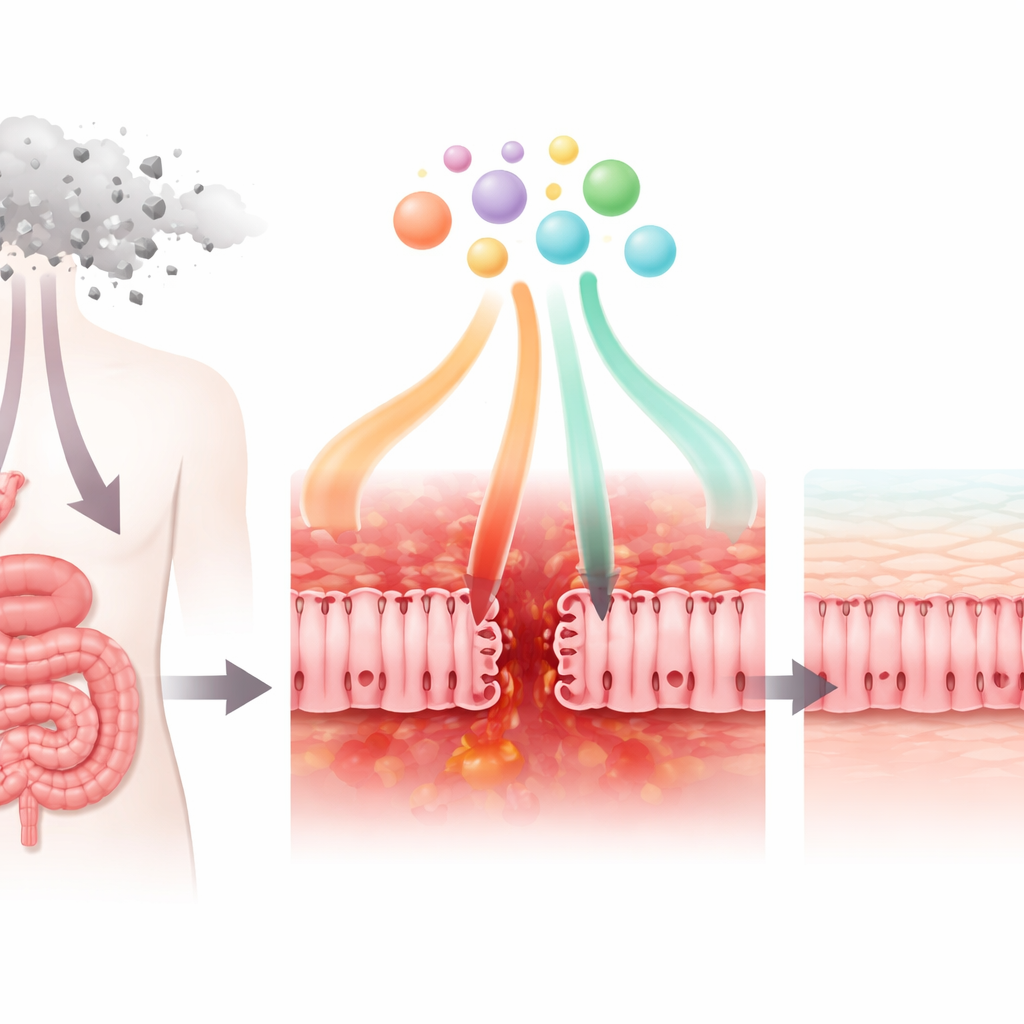
How a common cancer drug harms the gut wall
The researchers first examined how the chemotherapy drug 5-fluorouracil (5-FU), widely used against breast, stomach, and colon cancers, affects cells that resemble those lining the small intestine. In lab dishes, 5-FU reduced how well these cells survived and carried out normal metabolism. It also weakened the “tight junctions” that hold neighboring cells together, making the cell layer more leaky. Microscopy revealed that key junction proteins lost their usual neat, web-like arrangement and became disorganized. At the same time, the cells began to secrete higher levels of inflammatory signals, such as the chemokine CXCL8, while producing less of the regulatory factor TGF-β1. Together, these changes mirror what happens in patients when the gut barrier is damaged, inflamed, and less able to keep out microbes.
Helpful sugars from friendly bacteria
The team then turned to products made by L. reuteri DSM 17938: a cell-free supernatant containing many secreted molecules, purified exopolysaccharides (EPS, long sugar chains), and tiny membrane vesicles. These were added only after the chemotherapy drug had been washed away, mimicking a support treatment rather than a shield given beforehand. In both the intestinal-like cancer cell line and primary human small intestinal cells from a donor, EPS clearly improved barrier function. Electrical resistance across the cell layer rose, and the passage of fluorescent tracer across the barrier fell, indicating tighter sealing. Interestingly, EPS and the other bacterial products also boosted the release of CXCL8 and another chemokine, CCL20, especially on the side facing the gut lumen. Thus, the same treatment that strengthened the barrier also stirred up signals typically linked to inflammation.
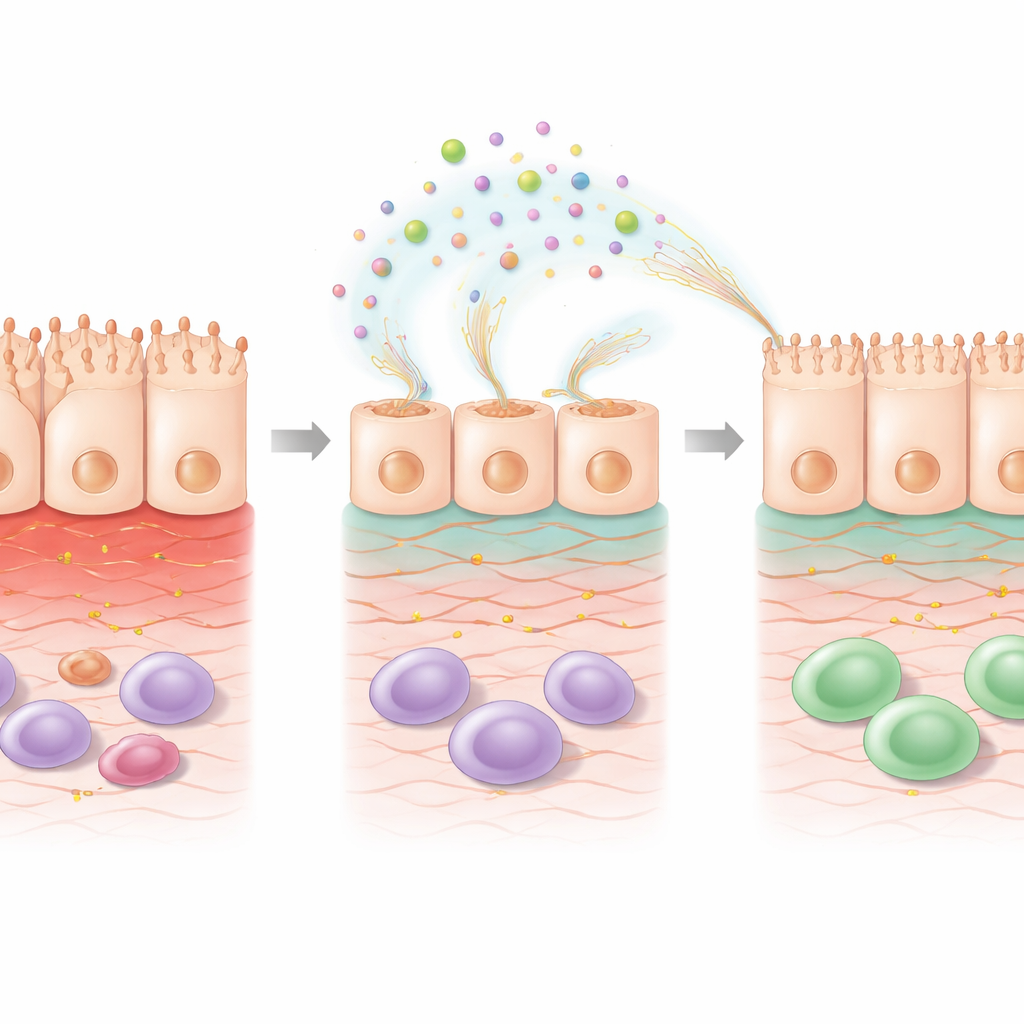
Inside the cell: repair programs switch on
To understand what was changing inside the cells, the scientists analyzed global gene activity after 5-FU treatment with or without EPS. Chemotherapy alone altered many genes related to cell survival, stress handling, and immune signaling. Adding EPS on top of 5-FU shifted the pattern further, in a way that pointed toward repair. Genes involved in organizing the extracellular matrix—the scaffold that supports cells—and in pathways linked to retinoic acid, a vitamin A–derived molecule known to promote gut barrier health, were upregulated. Other genes that help detoxify harmful byproducts of oxidative stress were turned on, while some genes associated with stress-induced damage were dialed down. These changes suggest that EPS do not simply coat the surface; they actively reprogram the injured cells to rebuild their structure and restore function.
Crosstalk with immune cells during healing
Because the gut barrier does not heal in isolation, the team asked how these epithelial changes might affect immune cells. They took the liquid from 5-FU–damaged, bacteria-product–stimulated epithelial cultures and used it to grow human monocytes into macrophages. Conditioned medium from EPS-treated cells pushed macrophages toward an M1-like profile, a state typically linked to early inflammatory responses and tissue clean-up. In contrast, medium from cultures treated with the cell-free supernatant tended to favor an M2-like profile, often associated with calming inflammation and tissue remodeling, while membrane vesicles produced a mixed pattern. Notably, even when surface markers suggested an M1-like skew, classic aggressive inflammatory cytokines such as IL‑1β and TNF‑α were not increased, hinting at a more balanced, repair-focused activation rather than runaway inflammation.
What this could mean for future cancer care
This work shows that specific molecules released by a probiotic bacterium—especially its exopolysaccharides—can help gut cells recover from chemotherapy-induced injury in the lab. EPS tightened the barrier, turned on internal repair programs, and shaped immune-cell behavior in ways consistent with orchestrated tissue healing. Although the study was done in cell cultures and will need confirmation in animals and patients, it points toward a future where carefully defined bacterial products, rather than live microbes, might be used alongside chemotherapy to reduce painful gut side effects and help patients stay on life-saving treatments.
Citation: Lasaviciute, G., López Plana, M., Sundberg Örtegren, S. et al. Limosilactobacillus reuteri metabolites modulate immune pathways and intestinal barrier repair after 5 fluorouracil exposure. Sci Rep 16, 11376 (2026). https://doi.org/10.1038/s41598-026-45524-y
Keywords: chemotherapy mucositis, gut barrier repair, probiotic metabolites, Limosilactobacillus reuteri, exopolysaccharides