Clear Sky Science · en
Mesenchymal to epithelial transition (MET) in cancer progression: insights from logical modeling
Why cancer cells changing shape matters
Cancer cells do not just divide; they also change their shape and behavior to spread through the body. To escape a tumor, they loosen their ties to neighboring cells and become more mobile. To grow new tumors elsewhere, they often need to regain their original, tightly connected form. This back and forth, known as cellular plasticity, is at the heart of metastasis. The study summarized here uses computer-based logic models to understand how cancer cells switch back from a mobile to a more settled state, and why this reversal is so dependent on the surrounding conditions.
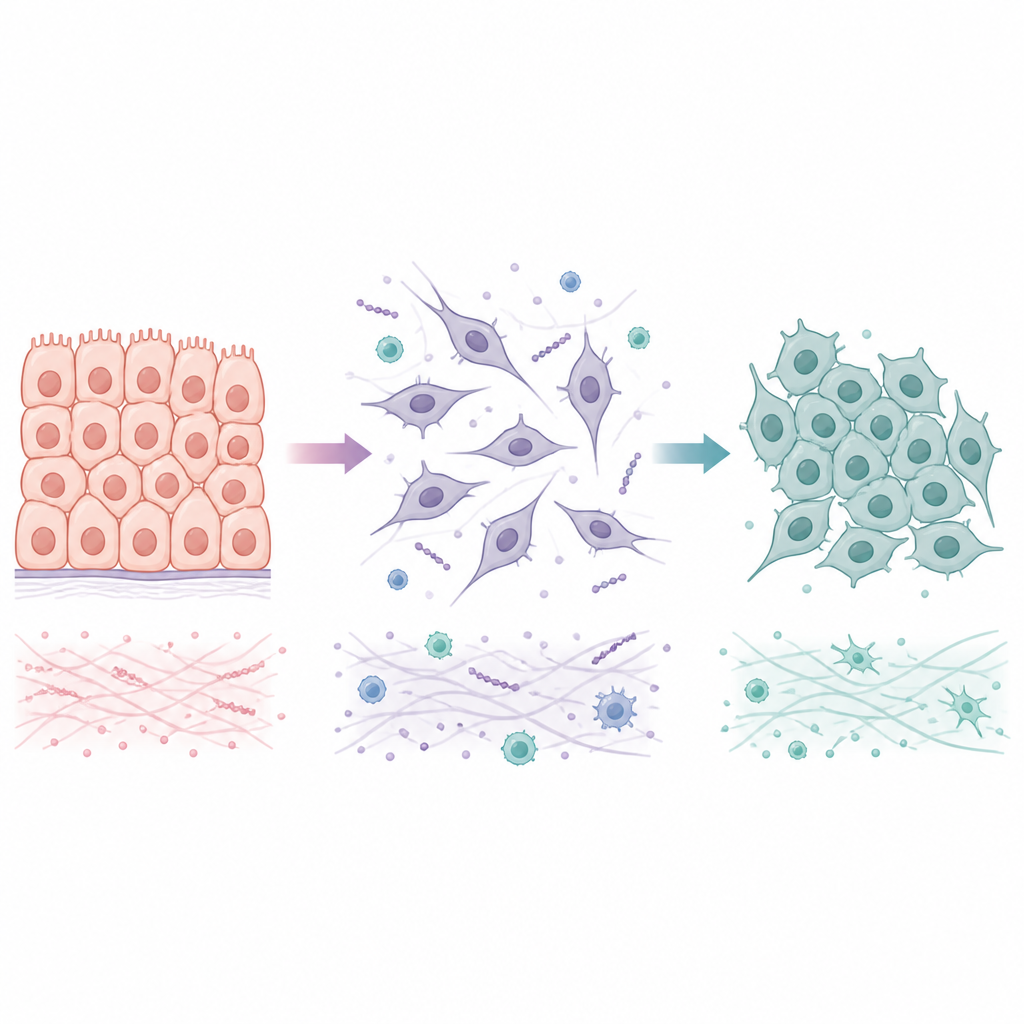
Two ways cells travel between settled and mobile states
Many tumors begin as epithelial cells, which are tightly linked into orderly sheets. During a process called epithelial to mesenchymal transition, these cells lose their tight junctions, change shape, and gain the ability to move and invade nearby tissue and blood vessels. When they arrive at a new organ, they often undergo the reverse shift, mesenchymal to epithelial transition, which helps them form stable colonies. Rather than flipping between only two endpoints, cancer cells often occupy hybrid states that mix settled and mobile features. These intermediate states seem especially good at surviving stress and seeding metastases, making it important to understand not just how the first transition occurs, but also how and when it can be reversed.
Signals that push or pull on cancer cell identity
Outside signals from the tumor environment strongly influence whether a cell behaves more like an epithelial or mesenchymal cell. Well-studied drivers such as TGFβ and various growth factors tend to encourage the mobile, invasive side, while other signals, including a protein family called BMP, can favor a return toward an epithelial state. Inside the cell, networks of genes and small RNAs reinforce one identity or the other. Certain transcription factors promote the breakdown of cell junctions and remodeling of the surrounding matrix, while another group of factors works to restore tight contacts and an epithelial character. Evidence from experiments shows that there is no single, universal route through these programs; instead, the outcome depends on which signals are present, for how long, and in which cell type.
Building a logical map of cellular decisions
Because so many molecules and feedback loops are involved, it is hard to guess how a cancer cell will respond in a particular setting. The authors gathered published information on key pathways linked to both directions of the transition and organized it into a regulatory network. They then translated this network into a Boolean, or on–off, logical model and ran many computer simulations. This approach does not try to measure exact molecule amounts; instead, it explores which combinations of signals and internal regulators lead to stable patterns of gene activity. The model produced 25 long-term states that naturally grouped into epithelial, mesenchymal, hybrid, and naive categories, reflecting the spectrum seen in laboratory studies.
Context shapes how far cells can revert
By simulating epithelial-like and mesenchymal-like starting conditions under different mixes of TGFβ, BMP, and growth factors, the model showed that the same external cue can have very different effects depending on context. In many cases, BMP was able to weaken a TGFβ-driven mesenchymal program and push cells toward more epithelial or hybrid states, but often stopped short of a full return. The simulations also highlighted the importance of self-sustaining feedback loops, in which cancer cells produce their own TGFβ or growth factor signals. These loops can maintain a mesenchymal state even after outside stimulation disappears. Breaking specific parts of these loops, or boosting certain pro-epithelial regulators, was predicted to shift cells toward hybrid or epithelial states, but the exact outcome again depended on the combination of signals and starting conditions.
What this means for understanding metastasis
Overall, the work suggests that mesenchymal to epithelial transition in cancer is not governed by a single master switch, but by overlapping networks of signals whose impact is highly context dependent. Some molecules appear capable of nudging cells back toward an epithelial-like state only in narrow circumstances, while others act more broadly but often still leave cells in hybrid states that retain invasive traits. For non-specialists, the main message is that metastasis relies on flexible cell identities, and that attempts to force cells back into a safer, less mobile form will need to account for the many feedback loops and environmental cues that stabilize these dangerous intermediate conditions.
Citation: Orozco-Ruiz, S., Ruscone, M., Barillot, E. et al. Mesenchymal to epithelial transition (MET) in cancer progression: insights from logical modeling. Sci Rep 16, 15032 (2026). https://doi.org/10.1038/s41598-026-44905-7
Keywords: epithelial mesenchymal transition, cancer metastasis, cell plasticity, logical modeling, bone morphogenetic protein