Clear Sky Science · en
Unstructured, disulfide-bridged C-terminus in helminth α-helical antimicrobial peptides enhances and modulates their activity
Why tiny parasite molecules matter
As antibiotic resistance rises, researchers are hunting for new ways to kill dangerous bacteria without harming our own cells. One promising source is antimicrobial peptides—short protein fragments that many organisms use as natural antibiotics. This study looks at an unusual peptide from a parasitic flatworm and shows how a small, floppy tail at one end, tied together by a chemical bond, can fine‑tune how fast and how forcefully the peptide kills bacteria. Understanding this built‑in control knob could help scientists design smarter, more precise antibiotic alternatives.
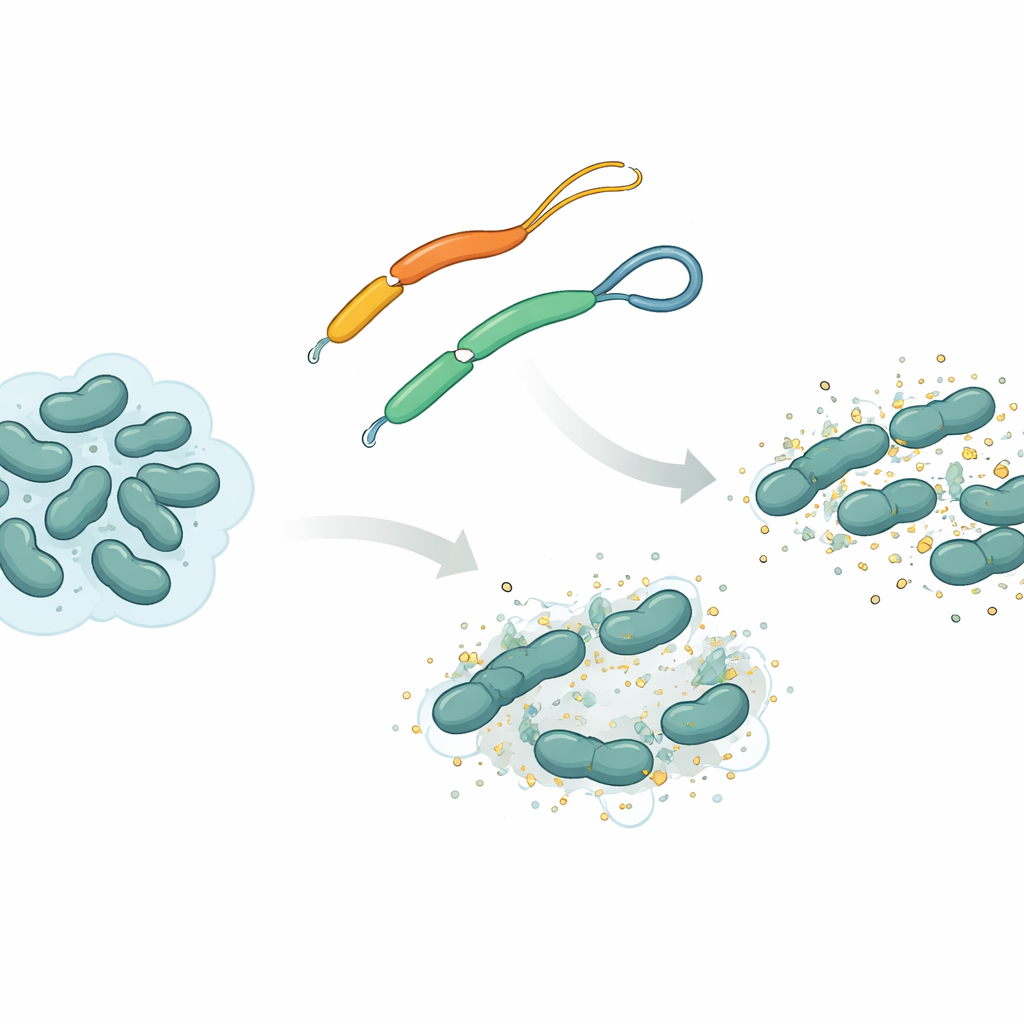
A natural weapon from flatworms
The team focused on an antimicrobial peptide called mesco‑2, found in the flatworm Mesocestoides corti. Like many such peptides, mesco‑2 has a rigid, spiral‑shaped “front” section that interacts with bacterial membranes, plus a looser “tail” at the back. In this tail, two sulfur‑containing building blocks are linked by a chemical tie known as a disulfide bridge, forming a tiny loop. The researchers compared mesco‑2 with a nearly identical version, mesco‑2A, in which that bridge is removed so the tail is fully open. This small change does not alter the overall chemical makeup much, but it changes how the peptide can move and how it behaves as it attacks bacteria.
Hidden patterns in parasite defenses
Before zooming in on mesco‑2 itself, the scientists searched large databases of predicted antimicrobial peptides from flatworms and from many other organisms. They looked for specific patterns where two sulfur‑containing units (cysteines) are separated by a few other building blocks near the end of the molecule. They found that a "CXXXC" pattern—the same spacing as in mesco‑2—is surprisingly common in flatworm peptides and often sits in flexible end segments rather than in well‑ordered regions. Computer‑generated 3D models of several such peptides showed that these cysteine‑rich motifs usually sit in floppy tails, suggesting that nature deliberately places these loops in movable regions that can modulate how the peptide behaves.
How a tiny loop shapes motion and teamwork
Using molecular dynamics simulations, the researchers modeled how mesco‑2 and mesco‑2A move near a simplified bacterial membrane. Both versions quickly attach to the membrane surface, driven by attractive forces between positive charges on the peptide and negative charges on the bacterial lipids. But the looped tail in mesco‑2 helps the central spiral region bend at a specific spot and adopt an orientation that favors partial insertion into the membrane. It also encourages several mesco‑2 molecules to gather into small clusters, especially when their tails start close together, and these assemblies remain together as they bind the membrane. In contrast, the open‑tailed mesco‑2A is more structurally flexible at its end, does not cluster as readily, and tends to stay more on the surface as individual molecules rather than as a coordinated group.
From structure to bacterial killing
Laboratory experiments backed up the computer models. When the peptides were mixed with simple membrane mimics, both changed shape in oily or detergent‑like environments, as expected for membrane‑targeting molecules. However, mesco‑2 gained regular spiral structure more easily and adopted a different shape than mesco‑2A on negatively charged, bacteria‑like membranes. In tests against several bacterial species, both peptides showed strong antibacterial effects at similar overall potency. The important differences appeared below the fully killing dose. Mesco‑2 slowed or stopped bacterial growth for many hours even at low levels, while bacteria exposed to the open‑tailed mesco‑2A recovered sooner. Time‑resolved measurements of membrane damage showed that mesco‑2 punches holes in bacterial membranes much more quickly, whereas mesco‑2A acts more slowly unless used at higher doses. High‑resolution imaging revealed distinct patterns of surface damage, again hinting at different ways the two versions disturb the bacterial envelope. Crucially, both peptides showed low damage to human immune‑cell models at bactericidal levels, suggesting a favorable safety margin.
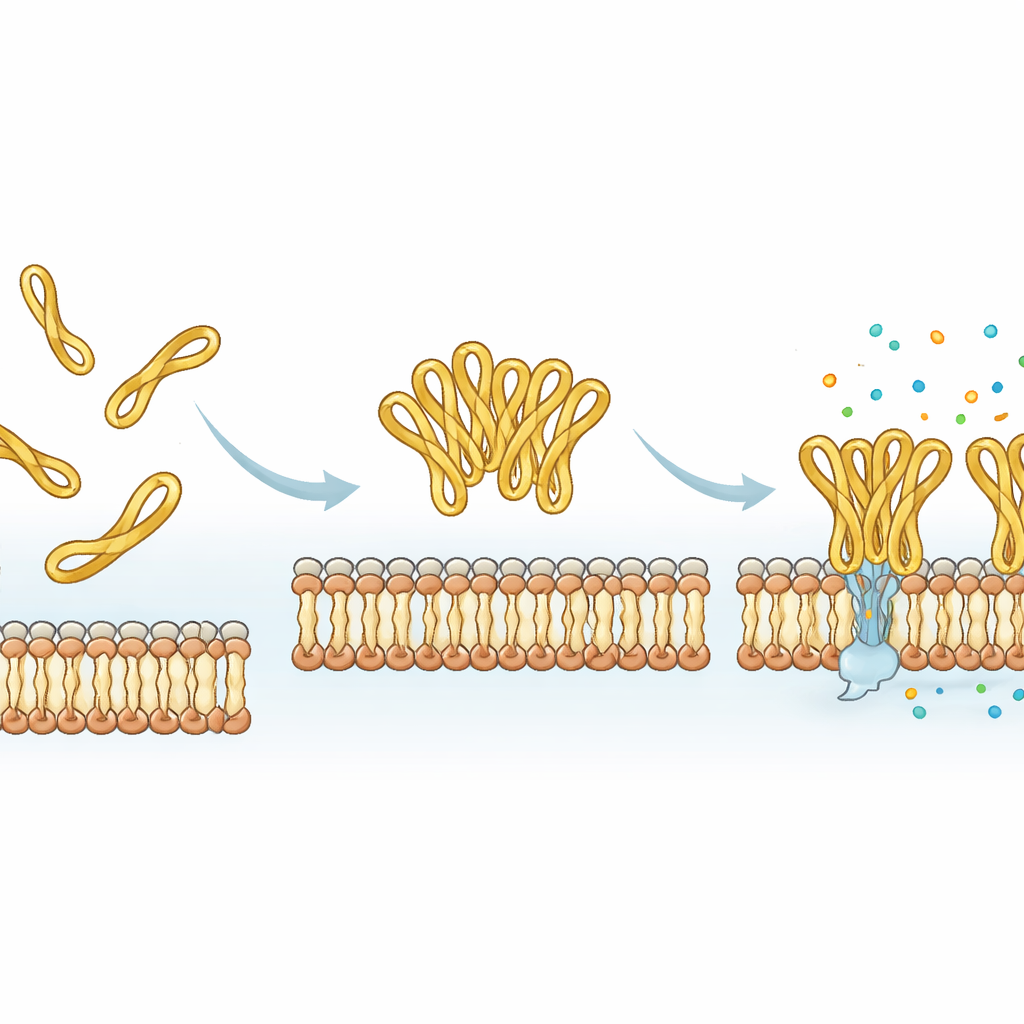
A built‑in dial for tuning peptide drugs
Putting all the evidence together, the study shows that the small disulfide‑tied tail at the end of mesco‑2 acts as a molecular dial: it stabilizes a particular bent shape, encourages peptides to work cooperatively as clusters, and speeds up membrane disruption, all without greatly increasing harm to human cells. Removing the loop makes synthesis easier and can even increase how many molecules stick to a model membrane, but it slows down the actual killing process and alters how the membrane is damaged. For future antibiotic‑like drugs based on such peptides, this means that seemingly minor, flexible end segments—rather than just the main spiral core—can be deliberately tweaked to control how fast, how strongly, and how cooperatively a peptide attacks bacteria.
Citation: Budimir, A., Stojan, I., Primorac, K. et al. Unstructured, disulfide-bridged C-terminus in helminth α-helical antimicrobial peptides enhances and modulates their activity. Sci Rep 16, 14472 (2026). https://doi.org/10.1038/s41598-026-44795-9
Keywords: antimicrobial peptides, flatworm peptides, disulfide bridge, membrane disruption, antibiotic resistance