Clear Sky Science · en
Numb mitigates intestinal epithelial cell senescence induced by radiation through a PLK1-dependent pathway
Why Radiation Can Hurt the Gut
Radiation therapy is a lifesaving treatment for many cancers, but it can be brutal on the gut. The lining of our intestines renews itself quickly, making it especially sensitive to radiation. When this lining is damaged, the barrier that normally keeps bacteria and toxins inside the gut and away from the bloodstream begins to leak. This study explores how a little-known protein called Numb helps shield the intestinal barrier from radiation damage, and how another protein partner, PLK1, fits into that protection. Understanding this relationship could point the way toward gentler cancer treatments with fewer long-term digestive side effects. 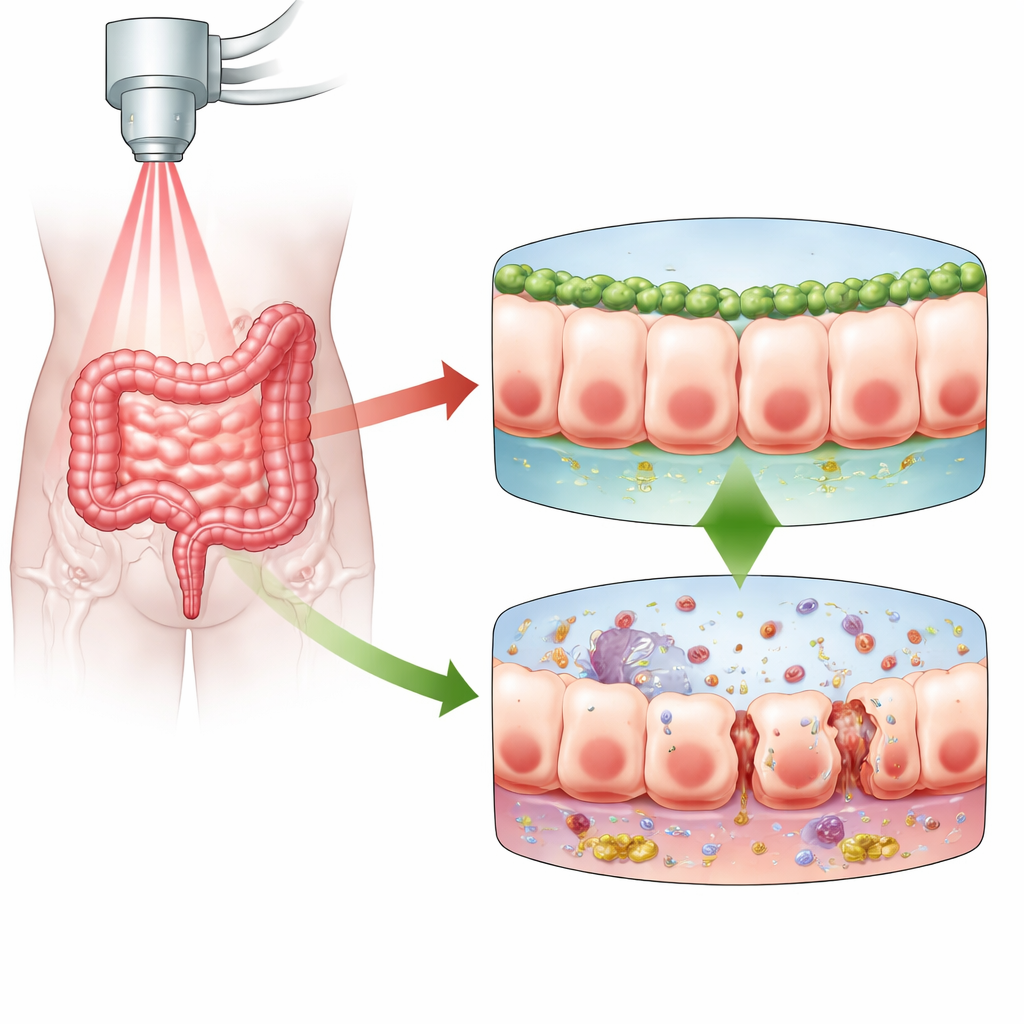
The Gut’s Protective Wall Under Attack
The intestinal mucosal barrier is a single, tightly packed sheet of cells that separates the contents of the gut from the rest of the body. When radiation hits this barrier, it damages the DNA inside these cells. In response, cells either die, pause to repair themselves, or enter a state called senescence, where they stop dividing but remain alive and often become highly inflammatory. The authors focused on radiation enteritis, a painful and sometimes disabling complication of pelvic radiotherapy. They suspected that Numb, a protein already known to help maintain cell polarity, adhesion, and repair in the gut lining, might influence how intestinal cells respond to radiation and whether the barrier stays tight or becomes leaky.
A Key Protein Makes Damage Worse When Silenced
To test Numb’s role, the researchers used mice and reduced Numb levels in their intestinal lining before exposing them to abdominal radiation. Mice lacking Numb in these cells fared much worse: more of them died, they lost more weight, and measures of gut leakiness in the blood were higher. Microscopy showed that the normally neat, continuous junctions between neighboring cells were disrupted, and the mucus layer that coats and protects the surface became thin and patchy. At the same time, inflammatory molecules in the gut tissue rose sharply, and the tiny crypts that house stem cells and drive intestinal renewal were fewer and more damaged. Together, these changes painted a picture of a barrier that could no longer repair itself efficiently or keep harmful contents confined to the gut.
When Cells Grow Old Too Soon
Digging deeper, the team looked at what was happening inside the epithelial cells themselves. They found that, with Numb reduced, radiation triggered more persistent DNA damage signals and a strong rise in markers of cellular senescence. More cells took on the classic senescent appearance and tested positive for a standard senescence enzyme stain, yet overall apoptosis—outright cell death—did not increase. In cultured human colon cell lines, removing Numb did not make cells die more after radiation, but it did push many of them into a prolonged stall in the cell cycle, especially in the phase just before division. These stalled cells accumulated damage and senescence markers and began to secrete higher levels of inflammatory factors, the so‑called senescence-associated secretory phenotype, which can further weaken tissue and fuel chronic inflammation. 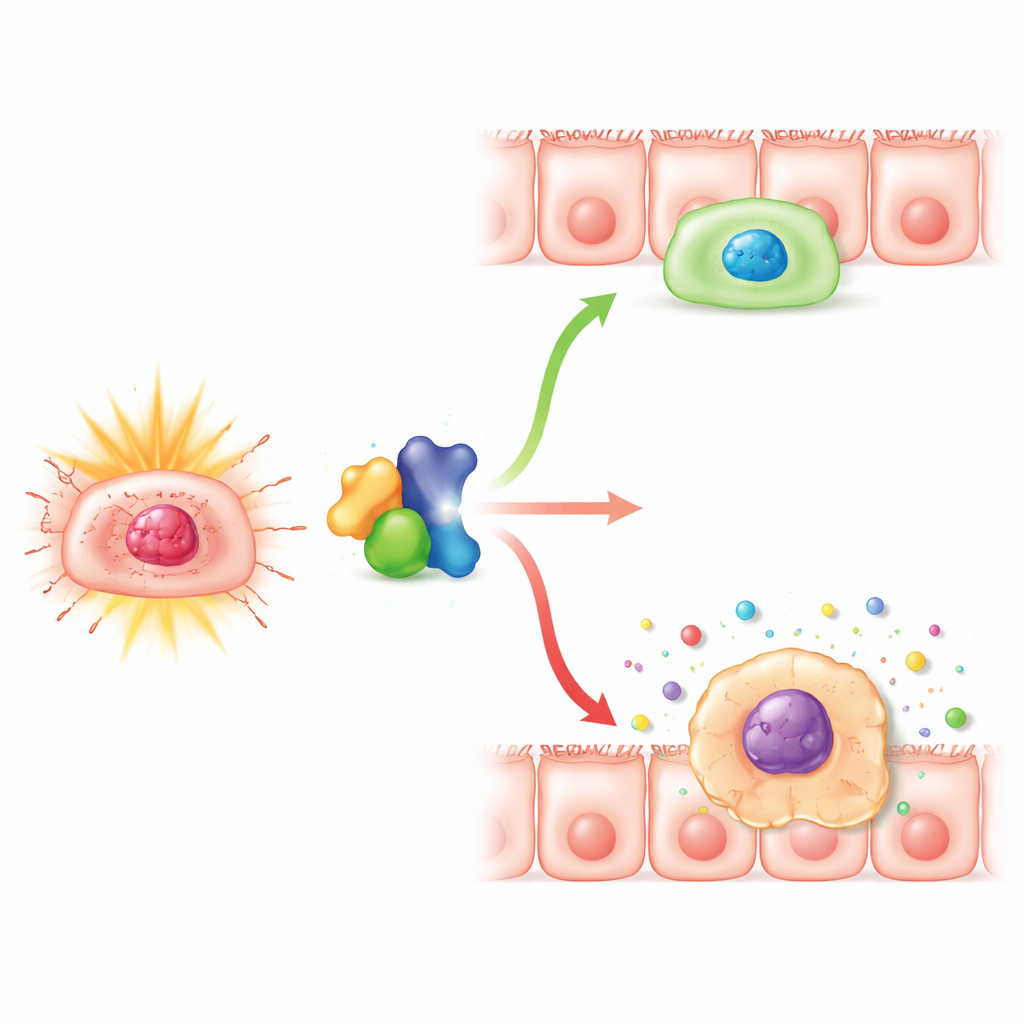
A Molecular Partner That Tips the Balance
The researchers then turned to PLK1, a protein that helps cells move through the final steps of division and recover from checkpoint pauses after stress. Earlier work had hinted that Numb and PLK1 interact. Here, the team confirmed that the two proteins physically associate in intestinal cells and that lowering Numb also reduced PLK1 levels. Blocking PLK1 alone made radiation-treated cells more prone to senescence, while boosting PLK1 could partly rescue the senescent, growth-arrested state caused by Numb loss. Overexpressing PLK1 decreased senescence markers, restored cell growth, reduced the fraction of cells stuck before division, and dampened the release of inflammatory molecules. These findings suggest that Numb helps keep PLK1 activity at a level that allows cells to repair damage and re-enter the cell cycle instead of sliding into a permanent, inflammatory standstill.
What This Means for Patients
The study proposes that Numb, working through PLK1, acts as a guardian of the intestinal barrier during radiation exposure by limiting premature cellular aging in the gut lining. Rather than simply preventing cell death, this pathway seems to steer damaged cells toward repair and controlled recovery, reducing the buildup of senescent, inflammation-driving cells that can undermine the barrier over time. While many details remain to be worked out, especially in living animals and human patients, the work points to the Numb–PLK1 axis as a promising target for treatments designed to protect the gut during radiotherapy—potentially allowing doctors to deliver effective cancer doses while sparing patients from chronic bowel problems.
Citation: Yang, Y., Hu, X., Pan, Y. et al. Numb mitigates intestinal epithelial cell senescence induced by radiation through a PLK1-dependent pathway. Sci Rep 16, 10876 (2026). https://doi.org/10.1038/s41598-026-44793-x
Keywords: radiation enteritis, intestinal barrier, cellular senescence, Numb protein, PLK1