Clear Sky Science · en
Integrated wet lab and in silico discovery and characterization of bacteriophages with antibiotic synergy against multidrug resistant Acinetobacter baumannii
Old Germ Foes Gain New Allies
Drug resistant infections are making once routine hospital treatments far riskier, especially when the culprit is Acinetobacter baumannii, a hard to kill bacterium that shrugs off many antibiotics. This study explores how viruses that prey on bacteria, called bacteriophages or phages, can be found in everyday wastewater and then trained and tested in the lab and on computers to work together with modern antibiotics against these stubborn hospital germs. 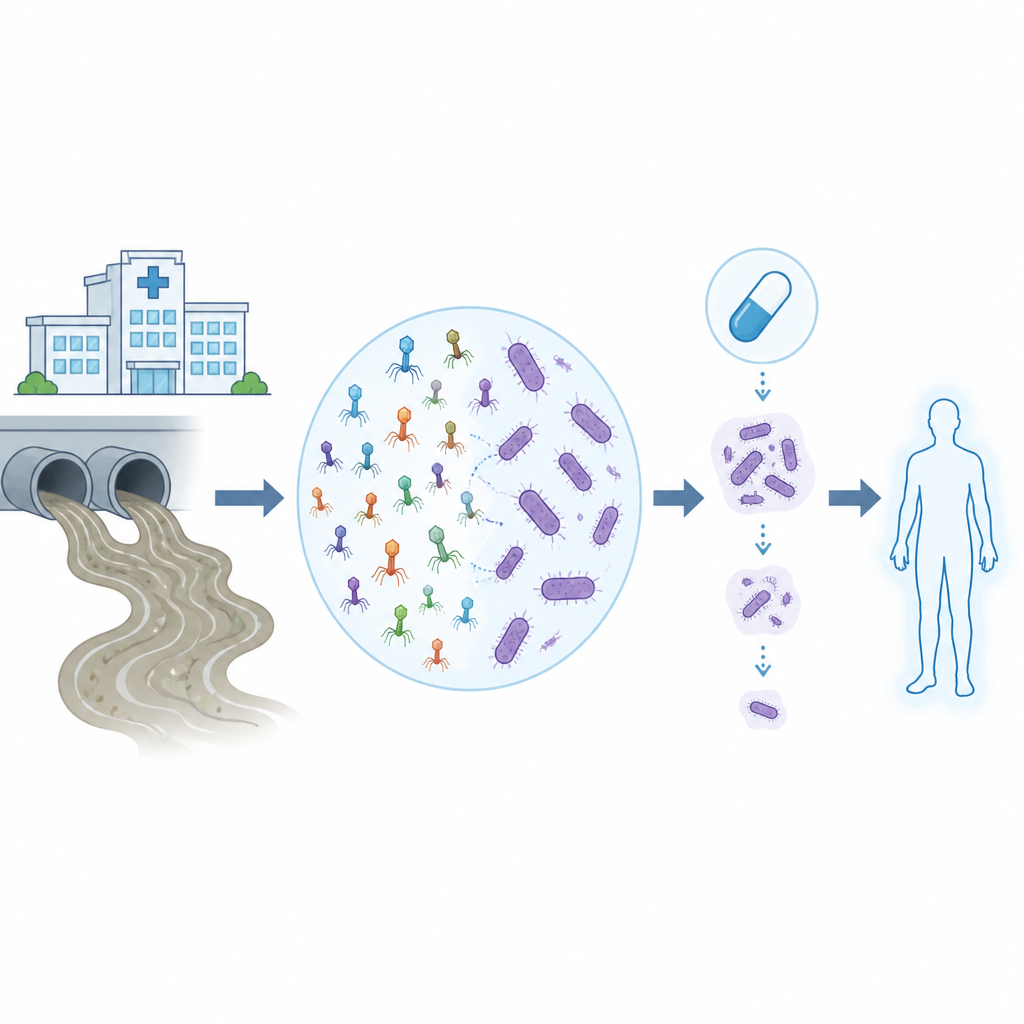
Hunting Useful Viruses in Wastewater
The researchers began by assembling a panel of twenty multidrug resistant A. baumannii strains collected from hospitals across Italy. Using genetic fingerprints, they chose two representative strains as bait for phages and exposed them to untreated wastewater, a rich natural reservoir of these viruses. Only one strain, called Abau1, yielded phages that formed clear spots of dead bacteria on lab plates. Electron microscopy showed four related phages, all from the same broad viral group, but with two distinct body shapes hinting at different lifestyles and behavior.
Sorting Good Killers from Quiet Hitchhikers
Not every phage is useful for therapy, because some tend to hide quietly inside bacterial DNA instead of destroying their host. The team sequenced the phage genomes and compared them with the host bacterium’s own genetic material. Two phages turned out to be nearly identical to pieces already embedded in the Abau1 chromosome, meaning they were host derived hitchhikers that could switch to a quiet, temperate lifestyle. The other two, named ΦAb1-SL1 and ΦAb1-SL2, showed no overlap with host DNA and carried no known genes for toxin production or antibiotic resistance, marking them as purely lytic phages that reliably burst and kill their targets.
Testing Strength, Reach, and Teamwork with Drugs
The scientists then asked how rugged and versatile the two lytic phages were. Both remained infectious across a wide range of temperatures and acidity, including conditions similar to those found in parts of the human body, and they multiplied efficiently once inside bacteria. However, they could infect only a narrow set of A. baumannii strains that shared a specific outer capsule type, underscoring how phages often behave like lock and key systems. Most strikingly, one phage, ΦAb1-SL1, showed strong synergy with several cephalosporin antibiotics, especially cefiderocol. In lab tests, combining this phage with low doses of cefiderocol cut bacterial growth far more than either treatment alone and lowered the antibiotic dose needed to move the strain out of the resistant range, while such help was not seen with the carbapenem drug meropenem. 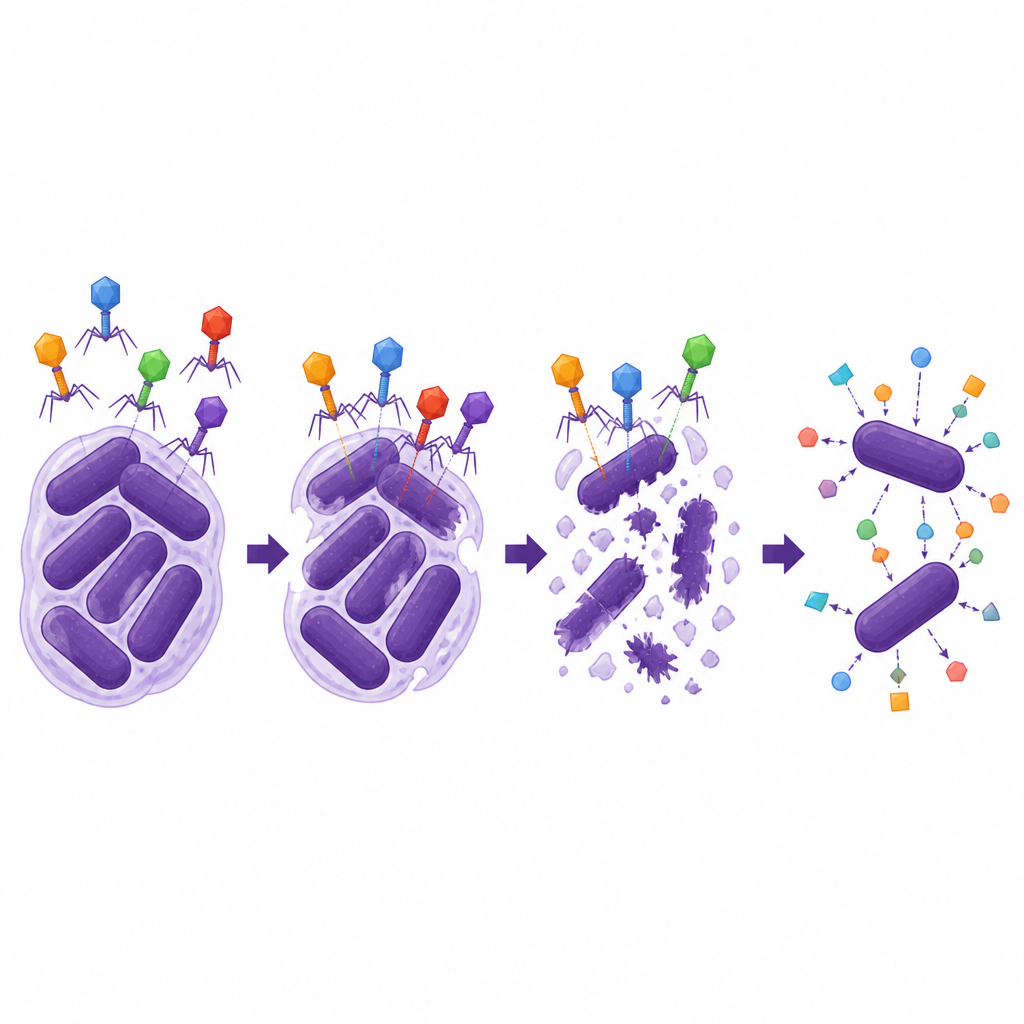
Training a Virus to Recognize New Targets
Because the narrow reach of a single phage limits its medical usefulness, the team tried to push ΦAb1-SL1 and ΦAb1-SL2 to infect a second, initially resistant strain named Abau2. By repeatedly growing phages with this new host, they eventually obtained an adapted variant, ΦAb2-SL1, that could attack both the original and the new strain, although it multiplied less efficiently on Abau2. Genome sequencing revealed only small genetic tweaks, mainly in tail proteins that likely touch the bacterial surface, yet these subtle changes expanded the phage’s host range while preserving its basic growth features and stability.
What This Means for Future Treatments
In simple terms, this work shows that useful phages against a high risk hospital germ can be fished from wastewater, screened and cleaned using modern genetic tools, and then trained to recognize additional bacterial targets. One of these phages worked especially well when paired with the advanced antibiotic cefiderocol, hinting that such combinations could revive drug activity against stubborn infections rather than replacing antibiotics outright. Although the experiments were done only in test tubes and the phages still infect a limited set of strains, the step by step pipeline outlined here points toward building regional phage banks that clinicians could draw from when treating multidrug resistant A. baumannii and, eventually, other tough bacterial foes.
Citation: Nicolosi, A., Bonanno Ferraro, G., Brandtner, D. et al. Integrated wet lab and in silico discovery and characterization of bacteriophages with antibiotic synergy against multidrug resistant Acinetobacter baumannii. Sci Rep 16, 15091 (2026). https://doi.org/10.1038/s41598-026-44506-4
Keywords: bacteriophage therapy, Acinetobacter baumannii, antibiotic synergy, cefiderocol, multidrug resistance