Clear Sky Science · en
Spatial proteomics of breast ductal carcinoma in situ reveal distinct regional differences
Why early breast changes matter
Ductal carcinoma in situ (DCIS) is an early form of breast disease often found on routine mammograms. It has not yet broken out of the milk ducts into surrounding tissue, but in some women it eventually leads to invasive breast cancer. Right now, doctors cannot reliably tell which DCIS lesions are harmless passengers and which are future threats, so many patients receive surgery and radiation they might not actually need. This study takes a new, high‑resolution look at DCIS to see how the arrangement of proteins in and around these ducts might help predict which cases are more likely to become dangerous.
Looking at breast tissue like a map
Instead of grinding tissue into a single blended sample, the researchers used a technology called spatial proteomics to measure dozens of proteins in precisely chosen spots on the slide, almost like dropping pins on a map. They examined archived tissue from 103 women treated for DCIS, some of whom never had further breast problems, some who later developed DCIS again in the same breast, and some who eventually had invasive cancer in that breast. For each case, pathologists marked regions inside the DCIS ducts, nearby supporting tissue (stroma), normal‑looking ducts, and biopsy sites. From more than 1,200 such tiny regions, the team quantified 53 proteins linked to hormones, growth, structure, and immune activity, creating a detailed spatial portrait of each lesion. 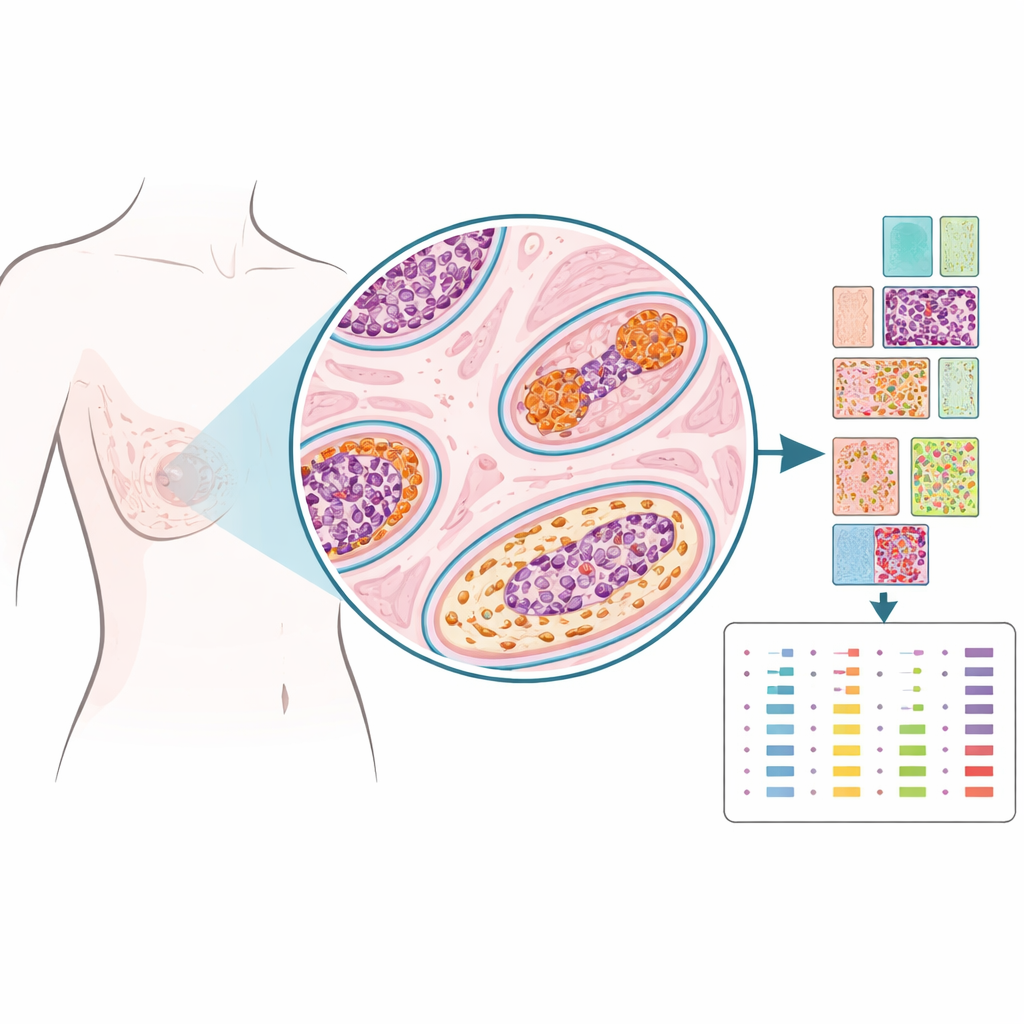
Two main types of early lesions
When the team grouped the data without telling the computer anything about clinical details, a striking pattern emerged. Most DCIS samples fell into one of two camps: those with high levels of estrogen receptor (ER) and low levels of the growth factor receptor HER2, and those with the opposite pattern, low ER and high HER2. This split resembled, but was not identical to, the way invasive breast cancers are categorized. Normal‑appearing ducts did not show the same sharp clustering, and the supporting tissue around DCIS showed its own distinct protein patterns. In the surrounding stroma, areas next to ER‑negative DCIS tended to be rich in immune cell markers, suggesting a more active immune neighborhood compared with regions around ER‑positive DCIS.
Hidden differences within each duct
Because the technique could separate regions within a single duct, the researchers also compared protein levels at the duct edge with those at the center. Proteins associated with the contractile outer layer of the duct and cell division were more abundant at the periphery, implying that cells hugging the duct wall are especially active. In contrast, a sugar transporter linked to energy supply was higher in the duct center, hinting at different metabolic demands in crowded inner cells. Notably, the overall hormone receptor levels did not change much between center and edge, but the fine‑scale balance of structural and growth‑related proteins did, revealing an internal organization that bulk tests would miss.
Clues to which DCIS may progress
The key question was whether any of these spatial protein patterns foretold future invasive cancer. Comparing women who went on to invasive disease with those who did not, the researchers saw suggestive but statistically modest differences in growth and structural proteins when looking at DCIS alone. Stronger signals appeared when they looked at how protein levels in DCIS compared with nearby normal‑looking ducts from the same patient. In lesions that later became invasive, the ratio of certain immune‑related proteins inside the ducts shifted: markers of helpful killer T cells and related receptors tended to be lower, while a protein that can dampen immune responses was relatively higher. This points to a local immune environment inside the ducts that may be less able to keep emerging cancer cells in check. 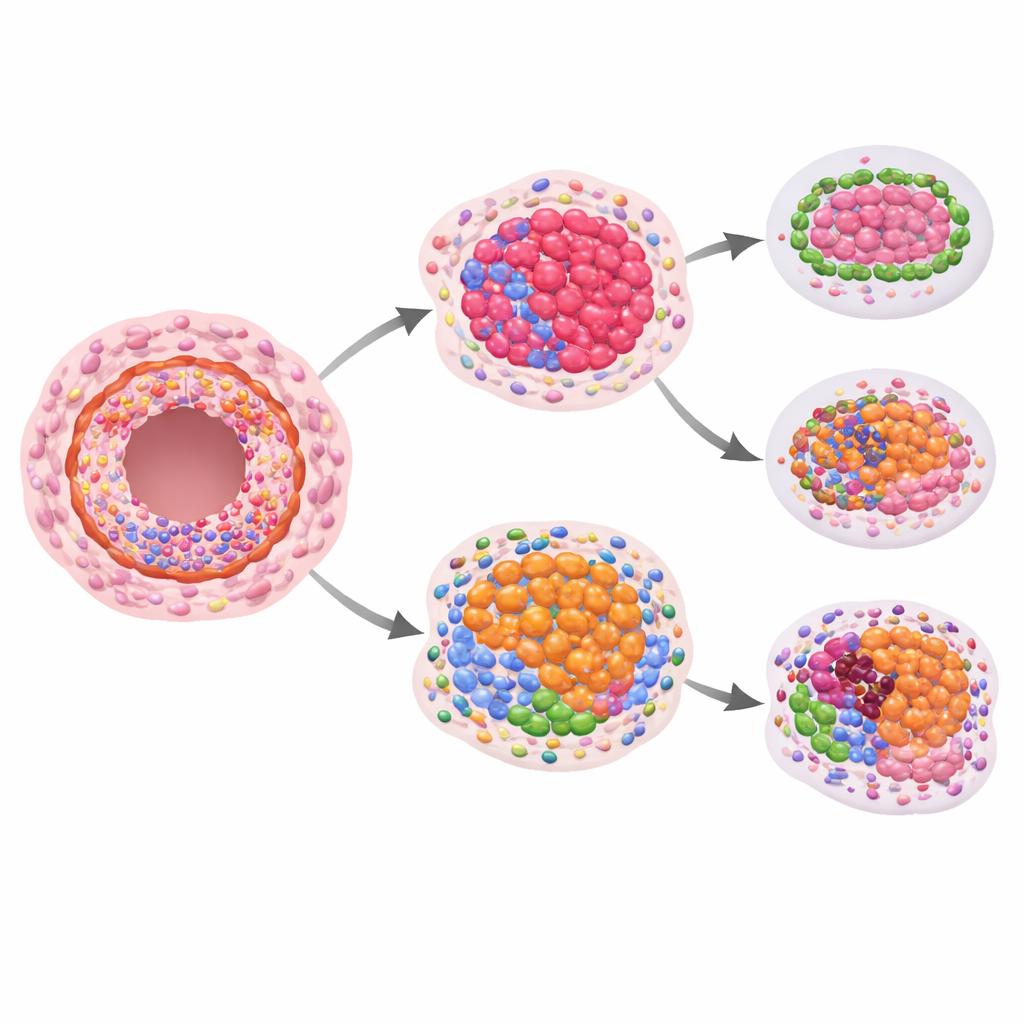
How treatment and time reshape tumors
For a subset of patients, the investigators also studied tissue from later events in the same breast—either another DCIS lesion or an invasive tumor—and compared them with the original DCIS. In many pairs, hormone and HER2 patterns were similar, but some showed major shifts, including cases where an ER‑high, HER2‑low DCIS was followed by a HER2‑high, ER‑low recurrence, or where HER2‑high DCIS was followed by an invasive cancer with much less HER2. Some of these changes likely reflect entirely new tumors arising independently rather than a straight‑line evolution of the original lesion. Among women who received hormone therapy, later cancers often had reduced levels of proteins driven by estrogen, suggesting that treatment can reshape the molecular profile of subsequent disease.
What this means for patients
The study shows that DCIS is not a uniform condition, but a patchwork of regions that differ by hormone signaling, immune presence, and cell behavior, and that these differences can matter for future risk. While no single protein emerged as a perfect crystal ball, the work highlights specific immune markers inside the ducts—as well as the contrast between DCIS and neighboring normal tissue—as promising ingredients for future risk scores. By adding this spatial layer to existing genetic and clinical information, researchers hope to move toward a future in which women with DCIS can be told, with greater confidence, whether they can safely choose careful monitoring or truly need aggressive treatment.
Citation: Marks, J.R., Dai, Y., King, L.M. et al. Spatial proteomics of breast ductal carcinoma in situ reveal distinct regional differences. Sci Rep 16, 13350 (2026). https://doi.org/10.1038/s41598-026-43486-9
Keywords: ductal carcinoma in situ, spatial proteomics, breast cancer risk, tumor microenvironment, immune markers