Clear Sky Science · en
Gamma-aminobutyric acid transaminase mediates tumor suppression in renal cell carcinoma through the cGAS-STING–interferon-β axis
Turning a Nerve Chemical into a Cancer Weakness
Kidney cancer, especially its most common form called clear cell renal cell carcinoma, is notoriously hard to treat once it spreads, often resisting both chemotherapy and radiation. This study reveals that an overlooked enzyme best known from brain chemistry—gamma‑aminobutyric acid transaminase, or ABAT—can act as a hidden ally against kidney tumors. By boosting this enzyme inside cancer cells, the researchers were able to wake up a powerful built‑in immune alarm system that slows tumor growth and reshapes the tumor’s immune environment.
A Missing Defender in Kidney Tumors
The team began by examining large patient databases and tumor samples to see how ABAT behaves in kidney cancer. In healthy tissue, ABAT helps break down the calming brain messenger GABA, keeping its levels in balance. In tumor samples, however, ABAT levels were consistently much lower than in nearby normal kidney tissue, and patients with the lowest amounts of ABAT tended to fare worse. This pattern suggested that tumors might silence ABAT to gain an advantage. 
Waking Up the Cell’s Intruder Alarm
To understand how a metabolic enzyme could have such a strong anti‑tumor effect, the researchers compared gene activity in cancer cells with and without extra ABAT. They found a striking rise in genes usually switched on by antiviral defense signals, especially those controlled by a molecule called interferon‑β. This pointed to activation of an innate immune “intruder alarm” normally used to sense viral or DNA damage. By using selective drugs that block different sensing routes, they traced the effect to the cGAS‑STING pathway—a cellular system that responds when DNA appears in the wrong place inside the cell. When this pathway was chemically shut down, ABAT could no longer drive high interferon‑β levels or suppress tumors as effectively, showing that its cancer‑fighting power depends on this internal alarm system.
Stress Inside the Cell’s Power Plants
ABAT usually lives inside mitochondria, the cell’s energy factories. The authors discovered that raising ABAT levels disturbs mitochondrial health: the membrane potential that powers them dropped, their shapes became abnormal under electron microscopy, and fragments of mitochondrial DNA leaked into the surrounding fluid within the cell. These escaped DNA pieces are exactly the kind of signal that switches on cGAS‑STING. The team also found that ABAT binds to another enzyme, PRMT5, which has been linked to cancer growth and to damping down immune alarms. When PRMT5 was reduced, STING and interferon‑β activity increased, hinting that ABAT may both damage mitochondria and counteract PRMT5 to lift the brakes on the alarm pathway, though the fine details remain to be worked out. 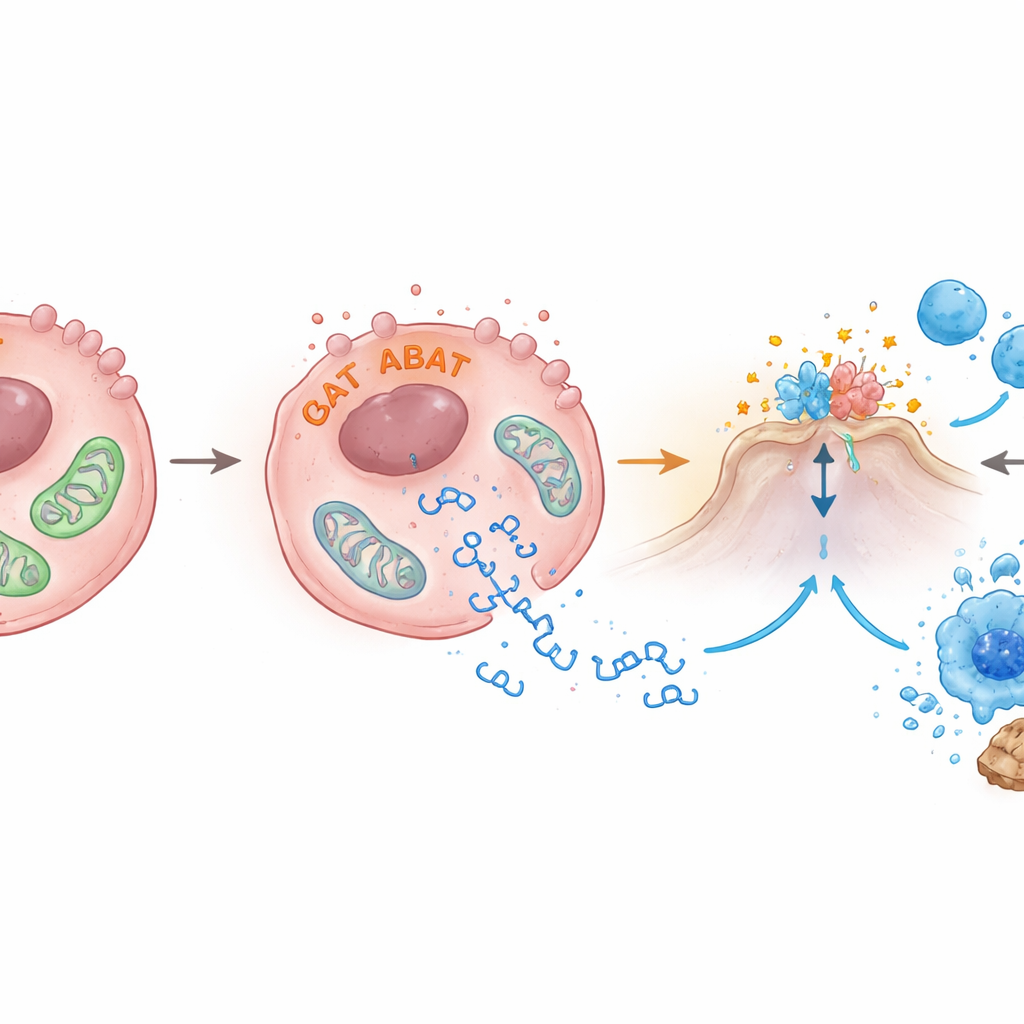
Shifting the Immune Balance Around the Tumor
Cancer does not grow in isolation; it lives within a complicated neighborhood of immune cells that can either attack or protect it. In immune‑competent mice, tumors made from ABAT‑rich kidney cancer cells not only grew more slowly but also showed a different immune makeup. The proportion of regulatory T cells—immune cells that normally calm responses and are often co‑opted by tumors to shield themselves—was noticeably lower, while the balance shifted in favor of killer T cells. This suggests that ABAT, by triggering interferon and damage signals, can tip the local immune environment away from suppression and toward anti‑tumor activity, potentially making other treatments like checkpoint inhibitors more effective.
What This Could Mean for Future Treatments
Altogether, the study portrays ABAT as more than a metabolic housekeeping enzyme: in kidney cancer it behaves like a built‑in tumor suppressor that, when restored, inflicts stress on cancer cell power plants, releases danger signals, and flips on an internal alarm that calls the immune system into action. Because ABAT is frequently reduced in patient tumors, strategies that boost its activity—or mimic the mitochondrial and immune‑activating effects it causes—could open new therapeutic avenues for advanced kidney cancer, especially in combination with existing targeted and immunotherapies.
Citation: Feng, Y., Cao, S., Cai, T. et al. Gamma-aminobutyric acid transaminase mediates tumor suppression in renal cell carcinoma through the cGAS-STING–interferon-β axis. Sci Rep 16, 13082 (2026). https://doi.org/10.1038/s41598-026-42861-w
Keywords: kidney cancer, innate immunity, tumor microenvironment, mitochondria, interferon signaling