Clear Sky Science · en
Efficient genome editing in a Mozambique tilapia cell line using CAS ribonucleoprotein complexes
Why this study matters for fish and food
Tilapia is a staple fish on dinner plates around the world, especially in regions where affordable, high quality protein is urgently needed. Yet tilapia farms are vulnerable to viral diseases such as Tilapia lake virus, which can wipe out stocks and threaten livelihoods. This study describes a lab based method to quickly and precisely edit tilapia genes in brain cells, laying the groundwork for future work to breed fish that can better withstand disease and support global food security.
Fish on the front line of food security
Aquaculture already provides about one sixth of the animal protein eaten globally, and tilapia are among the easiest and most common fish to farm. They grow fast, tolerate crowded ponds and are generally hardy. However, traditional selective breeding to improve traits like growth and disease resistance is slow and limited by the natural variation present in a stock. Genome editing offers a way to introduce useful genetic changes in a single generation, which could be especially valuable for traits like resistance to serious viral infections in farmed fish.
Building a useful cell model
The researchers focused on a cell line called OmB, derived from the brain of Mozambique tilapia. These cells are known to be infected by Tilapia lake virus, which makes them a practical stand in for studying how the virus interacts with its host. Before such cells can be widely used to test which genes affect infection or resistance, scientists need a reliable, efficient way to tweak specific genes in these cells and then measure what happens. This study set out to fine tune a step by step protocol to do exactly that using a popular gene editing tool known as CRISPR.
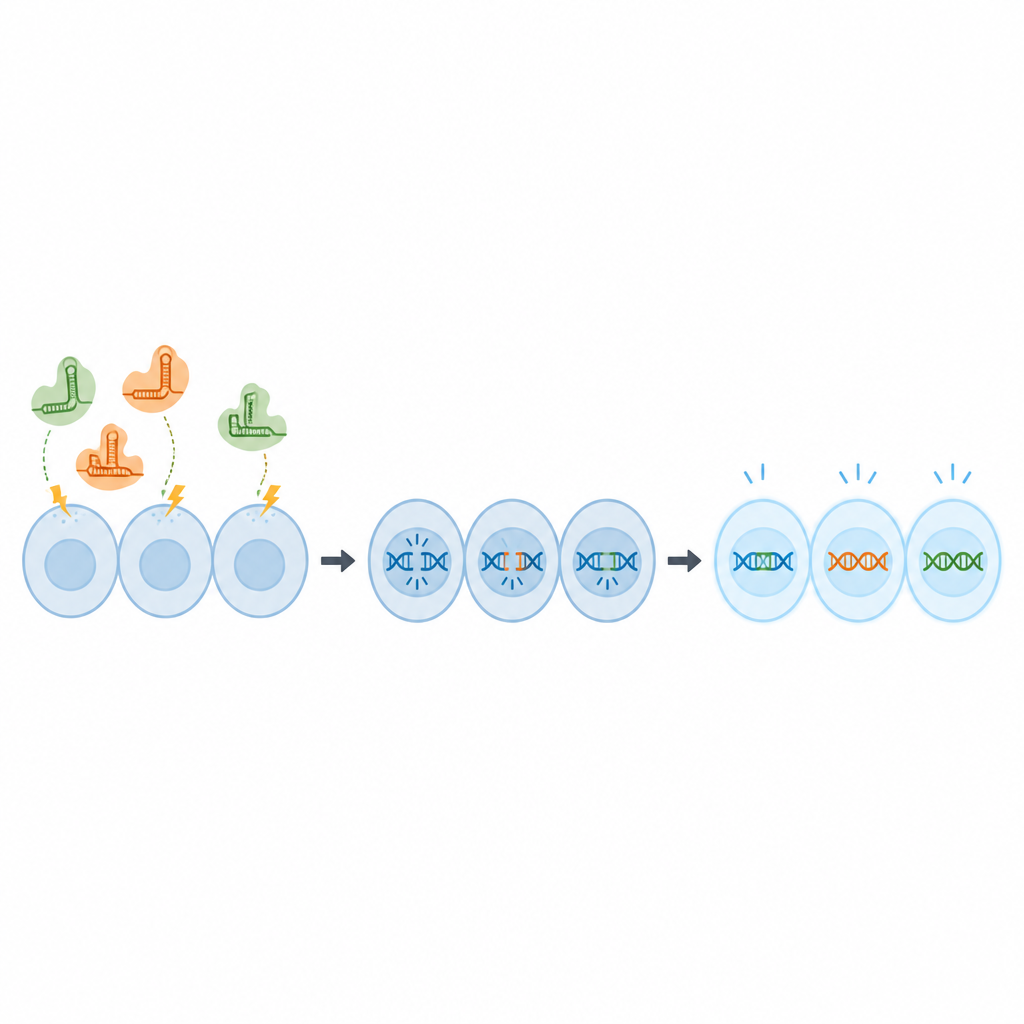
Fine tuning the editing tool
Instead of delivering CRISPR components as DNA, which can linger in cells and raise safety concerns, the team used ready made protein and RNA complexes known as ribonucleoproteins. These were pushed into OmB cells by short electrical pulses in a technique called electroporation. First, the scientists used a fluorescent tracer molecule to test different combinations of voltage, pulse length and concentration, looking for conditions that moved the tracer into nearly all cells without harming their survival. They found that a concentration of 2 micromolar tracer and a specific pulse setting gave close to complete uptake with healthy cells.
Testing and proving the method
Next, the team asked whether these conditions could actually change genes. They used a version of the OmB cell line that glows green and designed CRISPR molecules to cut the green protein gene. Under the optimized electrical settings, nearly 80 percent of gene copies were edited, and two weeks later only a small fraction of cells still glowed, showing that the gene had been effectively switched off. The scientists then moved on to two normal tilapia genes, chosen because changing them does not kill the cells but provides a clear readout. Using the same protocol, they achieved editing rates of around 67 to 70 percent and strong predicted knockout of gene function.
What this means for future fish health
The work delivers a practical recipe for high efficiency gene editing in a widely used tilapia brain cell line. By showing that CRISPR components can be delivered safely and that both marker and native genes can be disrupted in most cells, the study turns OmB cells into a powerful test bed. Researchers can now more easily switch candidate genes on or off to see how they influence viral infection, stress responses or other traits. While this study did not yet create disease resistant fish, it provides a key tool that will help scientists identify the genetic changes most likely to make farmed tilapia more resilient in the future.
Citation: Wang, J., Bobrik, M., Pankaew, N. et al. Efficient genome editing in a Mozambique tilapia cell line using CAS ribonucleoprotein complexes. Sci Rep 16, 15125 (2026). https://doi.org/10.1038/s41598-026-42702-w
Keywords: tilapia, aquaculture, CRISPR, fish disease, genome editing