Clear Sky Science · en
Synergistic induction of apoptosis by the epigenetic modulator decitabine and metformin in gastric cancer cells highlights the potential for combination therapy
Why this study matters
Stomach cancer is one of the deadliest cancers worldwide, in part because tumors often resist standard chemotherapy. This study explores whether pairing two already approved drugs—one for blood disorders and one for diabetes—can work together to push stomach cancer cells toward self-destruction, potentially pointing to safer and more effective treatment combinations in the future.
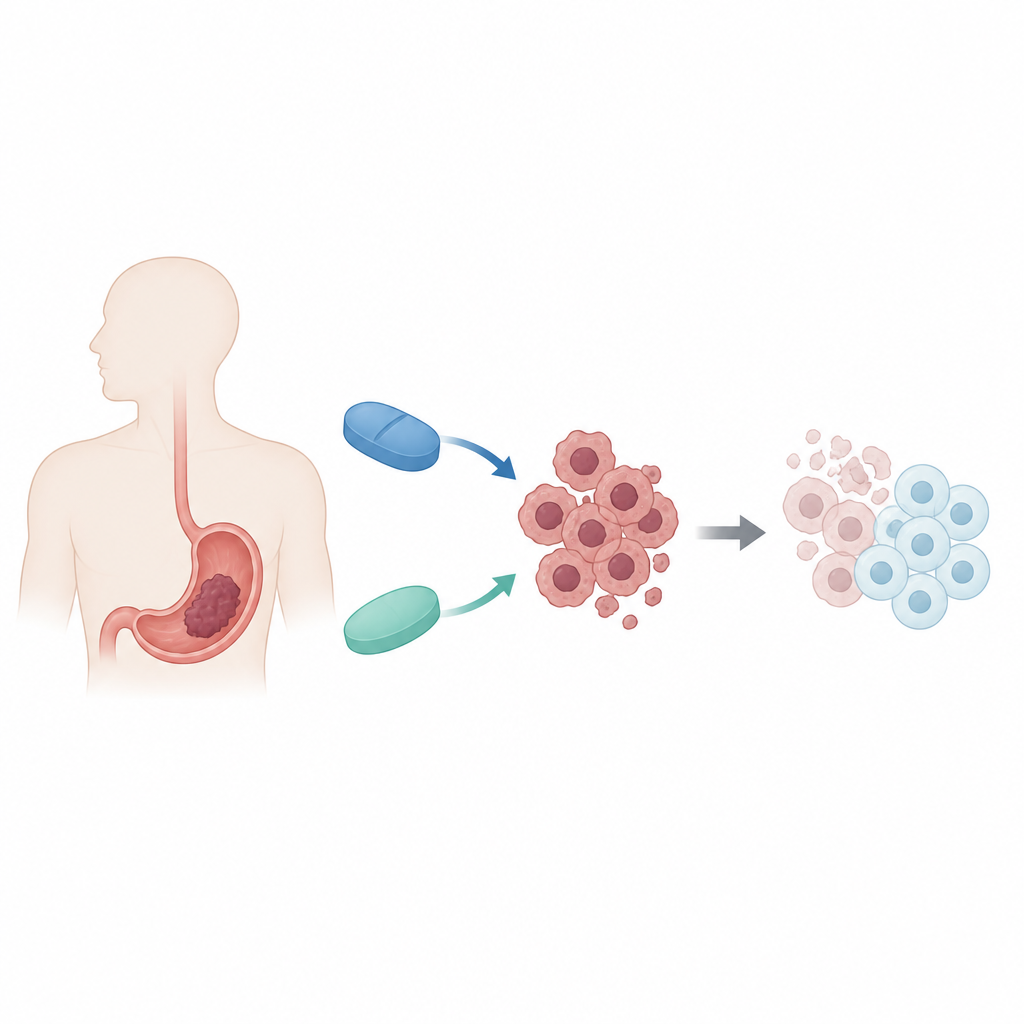
Two familiar drugs, a new partnership
The researchers focused on two medicines with very different primary jobs. Decitabine is used to treat certain bone marrow cancers by loosening chemical tags on DNA that silence protective genes. Metformin is a common diabetes drug that alters how cells handle energy and activates a cellular “fuel gauge.” Both have shown anticancer activity on their own, but only modest effects in solid tumors like gastric cancer. The team asked whether using them together in human gastric cancer cells might exploit the tumor’s weak points in both gene control and metabolism at the same time.
Testing the impact on cancer cell survival
Using a human stomach cancer cell line called MKN45, the scientists first exposed cells to a range of doses of each drug alone over several days, then to fixed-dose pairs of the two drugs together. They measured how many cells remained alive using a standard color-based laboratory test. A mathematical method that evaluates how drugs interact showed that the combination reduced the dose needed to slow cell growth and that the two drugs behaved synergistically at low to moderate killing levels. In other words, together they were more effective than expected from simply adding their individual effects, while allowing lower doses, especially of metformin.
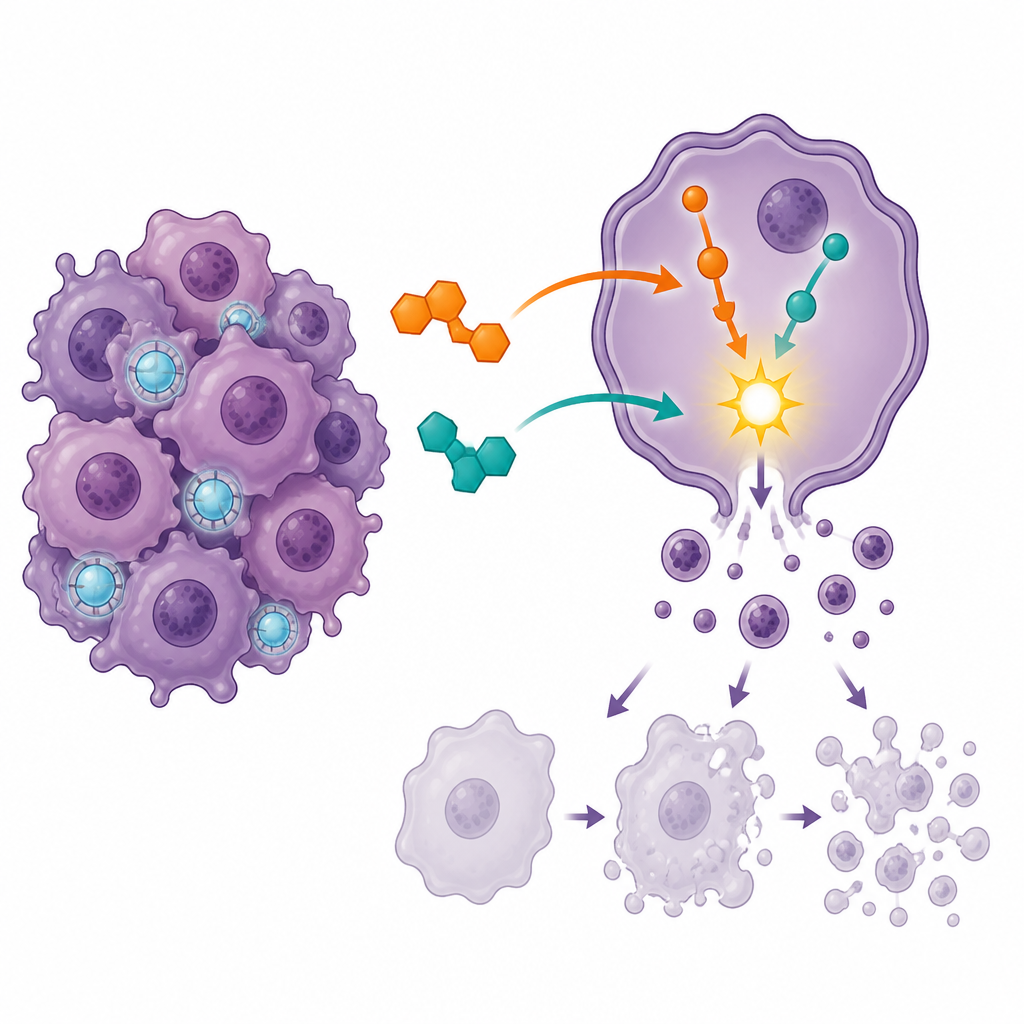
Triggering cell suicide and halting cell division
To see how the drugs were affecting the cancer cells, the team used flow cytometry, a technique that can distinguish living cells from those in different stages of programmed cell death, or apoptosis. Each drug alone nudged more cells toward death, but the combination caused a much larger jump in late-stage apoptosis and reduced the fraction of fully viable cells. At the same time, most cells were forced to pause at a key checkpoint of the cell cycle known as G2/M, where cells normally prepare to divide. This strong traffic jam at the division stage suggests that the combined treatment traps cells at a vulnerable moment, after which they are more likely to undergo controlled self-destruction rather than continue multiplying.
Switching on death programs inside the cell
The scientists then looked at activity levels of several genes that act as internal switches for survival and death. With the drug combination, genes that promote apoptosis, such as BAX and two caspases that cut up cellular components, were turned up, while BCL2, a gene that normally helps cells resist death, was turned down. The balance between BAX and BCL2 shifted sharply in favor of cell suicide. Another gene, ATG7, which helps manage a recycling process called autophagy, was also selectively increased, hinting that controlled breakdown and cleanup inside the cell may cooperate with apoptosis under treatment. Computer-based network and pathway analyses confirmed that the main biological themes affected were apoptosis pathways, stress signals linked to the p53 protein, and routes tied to resistance against standard platinum-based chemotherapy drugs.
What this could mean for future treatments
In plain terms, this work shows that decitabine and metformin, when used together, make stomach cancer cells more likely to stop dividing and to activate their built-in self-destruct machinery than when either drug is used alone. By jointly targeting faulty gene control and abnormal energy use, the pair appears to press on multiple “off” switches that the cancer relies on to survive and resist treatment. Although the findings so far come from one type of cancer cell grown in the lab, they suggest that combining epigenetic and metabolic drugs could help overcome treatment resistance in aggressive gastric cancers, and they provide a strong rationale for testing this strategy in additional cell models and, eventually, in animal studies.
Citation: AlAli, M., Latifi-Navid, S. & Khakzad, M.R. Synergistic induction of apoptosis by the epigenetic modulator decitabine and metformin in gastric cancer cells highlights the potential for combination therapy. Sci Rep 16, 14951 (2026). https://doi.org/10.1038/s41598-026-42417-y
Keywords: gastric cancer, decitabine, metformin, apoptosis, combination therapy