Clear Sky Science · en
Quercetin-4’-O-β-D-glucopyranoside inhibits ferroptosis though SIRT5-mediated desuccinylation of TFR1 in diabetic nephropathy
Why this matters for people with diabetes
Kidney disease is one of the most serious long-term complications of diabetes and a leading reason people need dialysis or transplants. This study explores a natural plant-derived compound, quercetin-4'-O-β-D-glucopyranoside (QODG), and how it might shield the kidneys from diabetes-related damage by blocking a specific form of cell death driven by iron and oxidative stress. Understanding this process could open the door to new, safer treatments that complement standard blood sugar and blood pressure control.
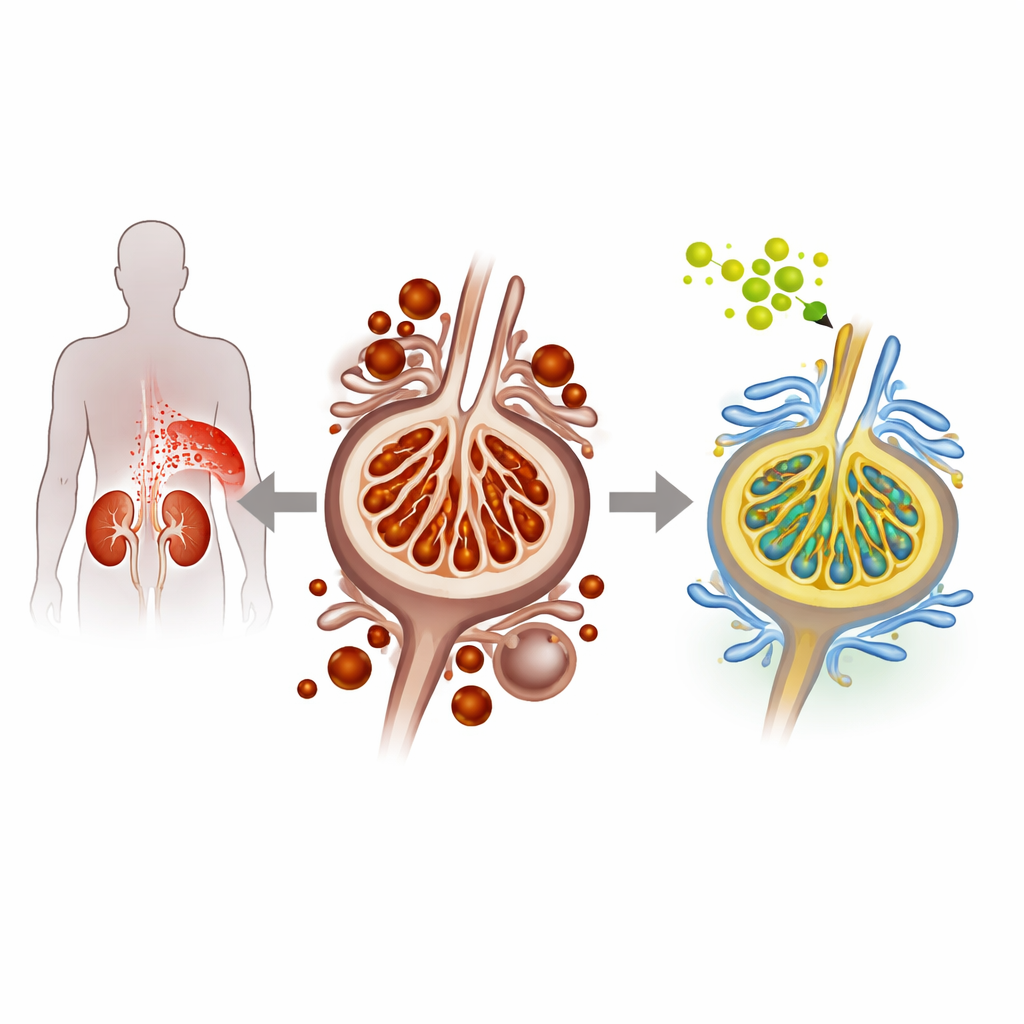
When sugar harms the kidney’s filter cells
In diabetic nephropathy, high blood sugar gradually injures podocytes, the delicate cells that line the kidney’s filtering units and prevent protein leakage into the urine. The researchers used both cultured mouse podocyte cells and a diabetic mouse model to mimic this condition. Under high-glucose conditions, the podocytes showed signs of a particular type of cell death called ferroptosis, which is fueled by excess iron and the buildup of damaged, oxidized fats in cell membranes. The team observed more iron inside cells, greater lipid peroxidation, and reduced levels of natural protective proteins, all pointing to heightened ferroptosis and worsening kidney stress.
A plant molecule that calms iron-driven damage
QODG is a flavonoid glycoside related to quercetin, a compound found in many fruits and medicinal plants. When the researchers treated high-glucose podocytes with QODG, cell survival improved in a clear dose-dependent manner. Markers of oxidative damage, such as lipid peroxidation and malondialdehyde, dropped, while antioxidant defenses like superoxide dismutase and catalase rose. At the same time, protein levels of GPX4 and SLC7A11, two key guardians against ferroptosis, increased, and levels of transferrin receptor 1 (TFR1), a major iron-import gate on the cell surface, fell. In diabetic mice, QODG treatment reduced structural kidney injury, lowered blood and urine markers of kidney dysfunction, and restored a healthier balance between damaging oxidative forces and protective antioxidant systems.
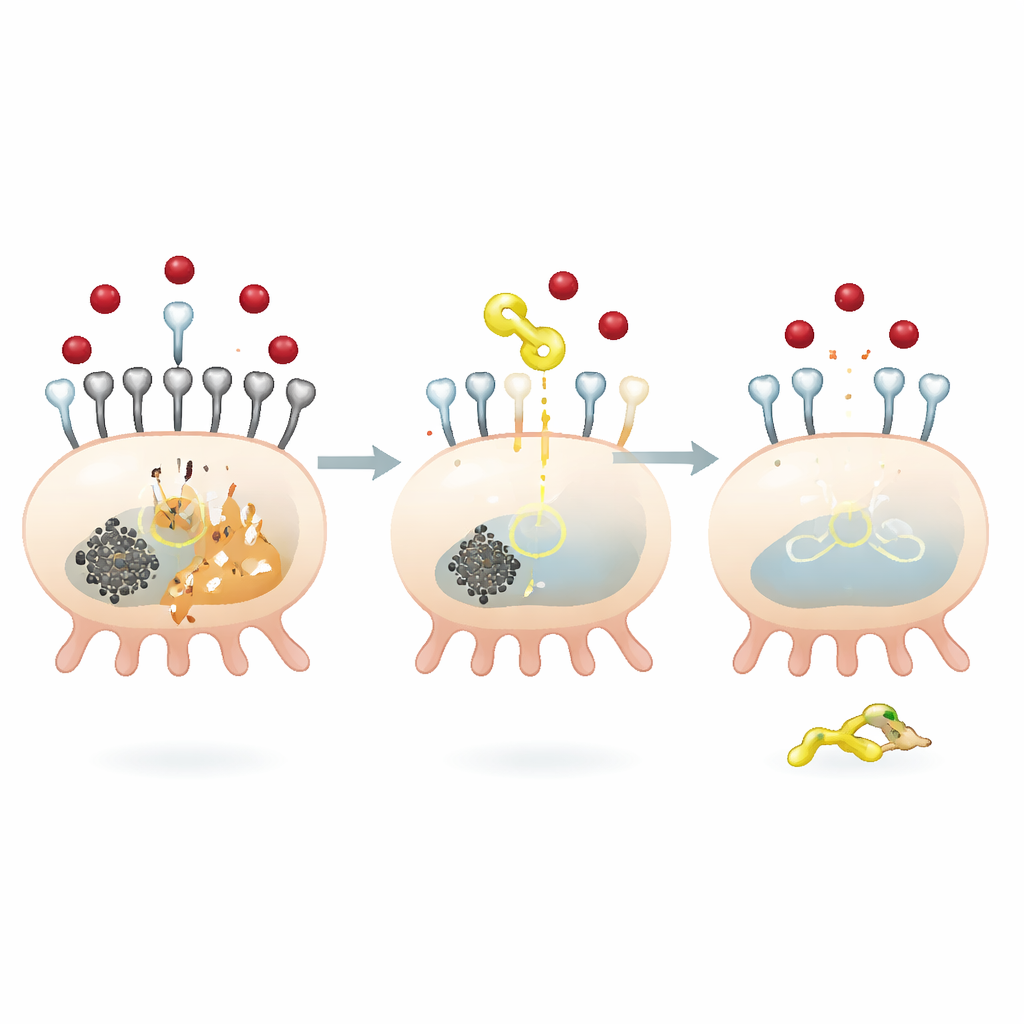
A molecular brake on iron entry into kidney cells
To understand how QODG exerts these protective effects, the team focused on a protein called SIRT5, an enzyme that removes a small chemical tag known as a succinyl group from other proteins. High glucose increased global succinylation inside podocytes and reduced SIRT5 levels. QODG reversed this: it boosted SIRT5 while bringing overall succinylation back down, without changing the supply of the succinyl donor molecule. The researchers found that SIRT5 specifically controls the succinylation of TFR1 at one site, an amino acid position called lysine 626. When SIRT5 was silenced, TFR1 became more heavily succinylated, more stable, and more abundant, leading to increased iron uptake and stronger ferroptosis signals. Conversely, restoring SIRT5 reduced TFR1 stability and expression, curbing iron influx and cell death.
Proving the link between the regulator and the iron gateway
The team used several complementary techniques to confirm that SIRT5 physically binds TFR1 and adjusts its modification state. Mutating different potential succinylation sites on TFR1 showed that only changes at lysine 626 altered both succinylation and total TFR1 protein levels, pinpointing this as the critical control point. Biochemical pull-down experiments and a proximity-based imaging assay confirmed direct interaction between SIRT5 and TFR1. Functionally, overexpressing SIRT5 alone made podocytes more resistant to ferroptosis, but forcing TFR1 levels back up wiped out this protection, restoring iron accumulation and oxidative damage. These findings place SIRT5 and TFR1 in a single pathway that determines how vulnerable kidney cells are to iron-driven injury under diabetic conditions.
What this could mean for future treatments
Overall, the study shows that QODG can ease diabetic kidney damage in cells and mice by turning up SIRT5, which then strips a chemical tag from TFR1, destabilizing this iron-import gateway and dampening ferroptosis. For a lay reader, the key idea is that a plant-based compound appears to help kidney filter cells by closing the “iron door” that feeds a destructive form of cell death. While more work is needed in larger animal studies and eventually in humans, these results highlight a promising strategy: targeting iron handling and oxidative cell death with natural molecules to slow or prevent diabetic kidney disease.
Citation: Wu, M., Ye, W. & Ye, X. Quercetin-4’-O-β-D-glucopyranoside inhibits ferroptosis though SIRT5-mediated desuccinylation of TFR1 in diabetic nephropathy. Sci Rep 16, 12384 (2026). https://doi.org/10.1038/s41598-026-41148-4
Keywords: diabetic nephropathy, kidney protection, ferroptosis, flavonoids, iron metabolism