Clear Sky Science · en
TREM1–NLRP3–driven pyroptosis in sepsis-associated acute kidney injury (AKI) with parallel autophagy changes
Why this kidney story matters
Sepsis, a life-threatening reaction to infection, often strikes the kidneys first, leading to sudden kidney failure in millions of people each year. Once the kidneys falter, the risk of death rises sharply, yet doctors still lack precise early warning signals and targeted treatments. This study explores a newly uncovered chain reaction inside kidney cells during sepsis—one that can turn protective immune defenses into a self-destructive storm—and asks whether interrupting this chain could both flag danger earlier and protect the kidneys from severe damage.
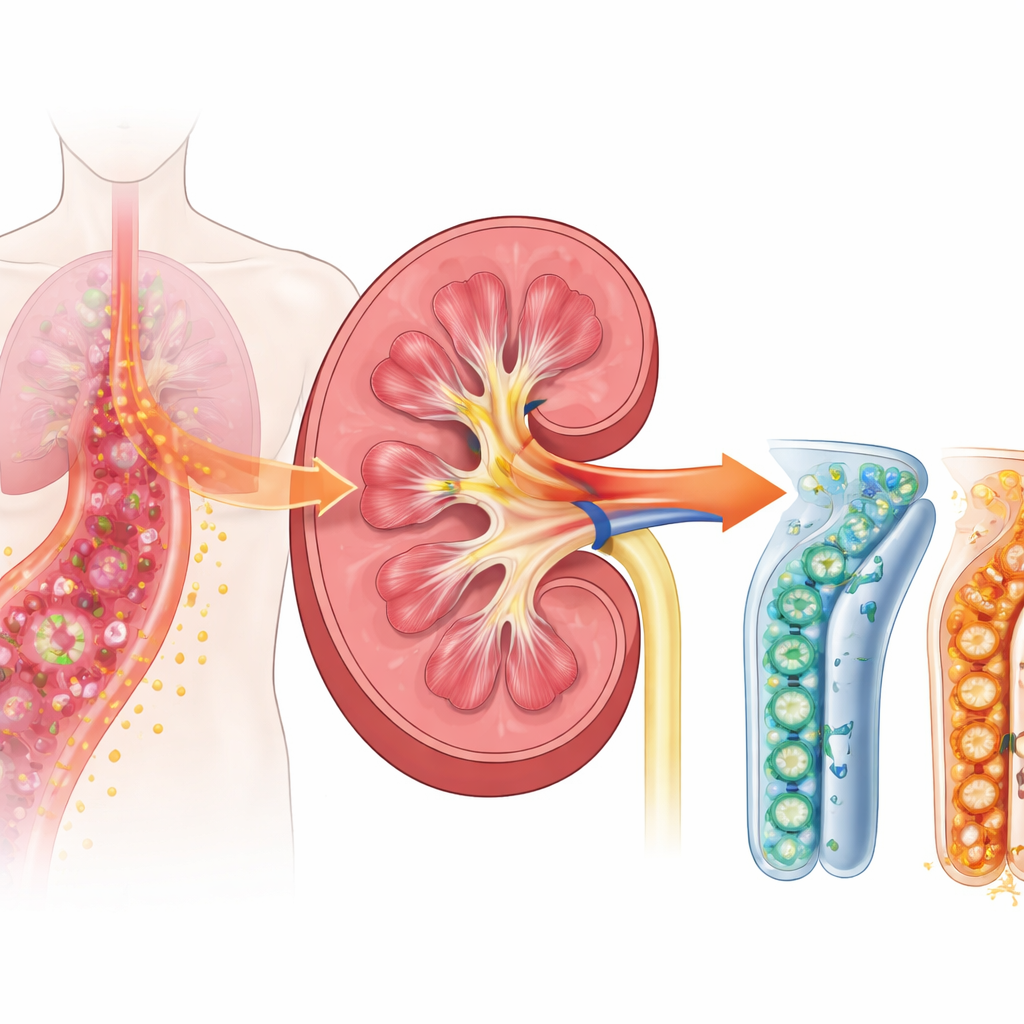
A dangerous chain reaction in sepsis
When sepsis develops, the body’s immune system goes into overdrive to fight invading microbes. The kidneys are not just filters; their tubular cells also help sense and respond to infection. The researchers focused on a surface receptor called TREM-1, which sits on immune cells and, as newer work suggests, on kidney tubule cells as well. TREM-1 acts like a volume knob for inflammation: once turned up, it can greatly amplify the alarm signals that drive swelling, fever, and tissue injury. Previous patient studies showed that a soluble form of this receptor, shed into blood and urine, rises early in sepsis and is linked to kidney problems, hinting that TREM-1 may be an important upstream trigger of damage.
From warning signal to cell explosion
Inside kidney tubule cells, the team examined how TREM-1 connects to a molecular machine called the inflammasome, which governs a dramatic type of cell death sometimes described as cellular explosion. In mice subjected to a standard sepsis model, the authors found that sepsis sharply increased TREM-1 levels in kidneys and in the bloodstream, along with activation of an inflammasome built around a protein called NLRP3. Once switched on, this complex activates enzymes that punch holes in cell membranes and release potent inflammatory molecules, killing the cell in the process. This fiery death program, known as pyroptosis, can help to clear infection but, when unchecked, can also destroy large swaths of fragile kidney tissue.
Turning down the volume knob and testing the pathway
To see how crucial TREM-1 is in this chain reaction, the researchers used mice genetically engineered to lack the Trem1 gene. Compared with normal mice subjected to the same septic insult, these knockout animals had less structural damage in their kidney tubules, lower blood levels of waste products such as creatinine and urea, and fewer inflammatory molecules circulating in their blood. Their kidneys also showed reduced signs of NLRP3 inflammasome activity and pyroptotic cell death. When the team directly blocked NLRP3 with a drug, kidney injury improved even in normal mice, whereas forcing NLRP3 into overdrive worsened damage even when Trem1 was missing. Together, these experiments place TREM-1 clearly upstream of NLRP3 in the injury cascade.
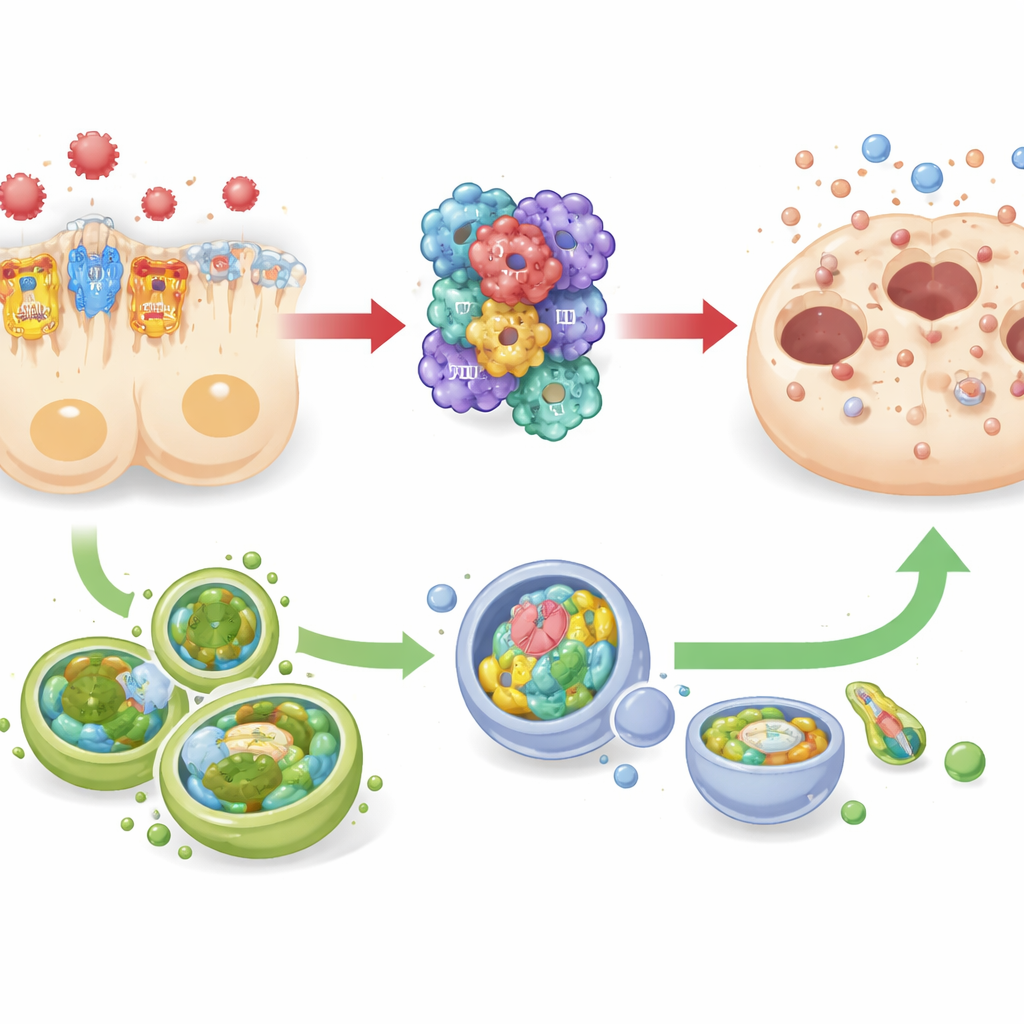
A tug-of-war with the cell’s cleanup crew
The study also tracked changes in autophagy—a built-in cleanup and recycling system that helps cells survive stress by removing damaged components. In both septic mice and human kidney tubule cells grown in the lab, dialing down TREM-1 was linked to higher levels of autophagy-related markers, while stronger TREM-1 signaling coincided with reduced autophagy. At the same time, inflammasome activity and cell death markers moved in the opposite direction. This pattern suggests a push–pull relationship: TREM-1 and NLRP3 acting together as a “gas pedal” for destructive inflammation, and autophagy behaving more like a “brake” that may counterbalance this drive. However, because the researchers did not directly switch autophagy on or off, they caution that its protective role, while strongly suspected, remains to be proven.
What this could mean for patients
For people with sepsis, the findings have two key implications. First, the soluble form of TREM-1 in blood or urine may serve as an early warning marker indicating that this damaging pathway is active in the kidneys before full-blown failure appears. Second, drugs that dampen TREM-1 or the NLRP3 inflammasome might one day protect kidneys from sepsis-related injury, particularly in patients whose lab tests show strong activation of this pathway. The work paints a picture of sepsis-associated kidney injury as a battle between an inflammatory accelerator and an internal cleanup system, and it identifies TREM-1 as a promising handle for both spotting high-risk patients and designing more precise treatments.
Citation: Liu, X., Chen, Q., Chen, X. et al. TREM1–NLRP3–driven pyroptosis in sepsis-associated acute kidney injury (AKI) with parallel autophagy changes. Sci Rep 16, 14239 (2026). https://doi.org/10.1038/s41598-026-40893-w
Keywords: sepsis-associated acute kidney injury, TREM-1, NLRP3 inflammasome, pyroptosis, autophagy