Clear Sky Science · en
Notch-1 in macrophages promoted the ischemia-reperfusion via modulating EZH2/HSF1/BRD4/SIRPα/SHP2 induced ROS and apoptosis in cardiomyocyte
Why heart repair can sometimes hurt the heart
When doctors reopen a blocked heart artery after a heart attack, blood rushes back into starved tissue. That rescue, called reperfusion, is life‑saving—but it can also injure the heart further. This study explores why the body’s own immune cells, meant to help with healing, can instead worsen this "reperfusion injury," and points to molecular switches that might be turned to protect the heart in future treatments.
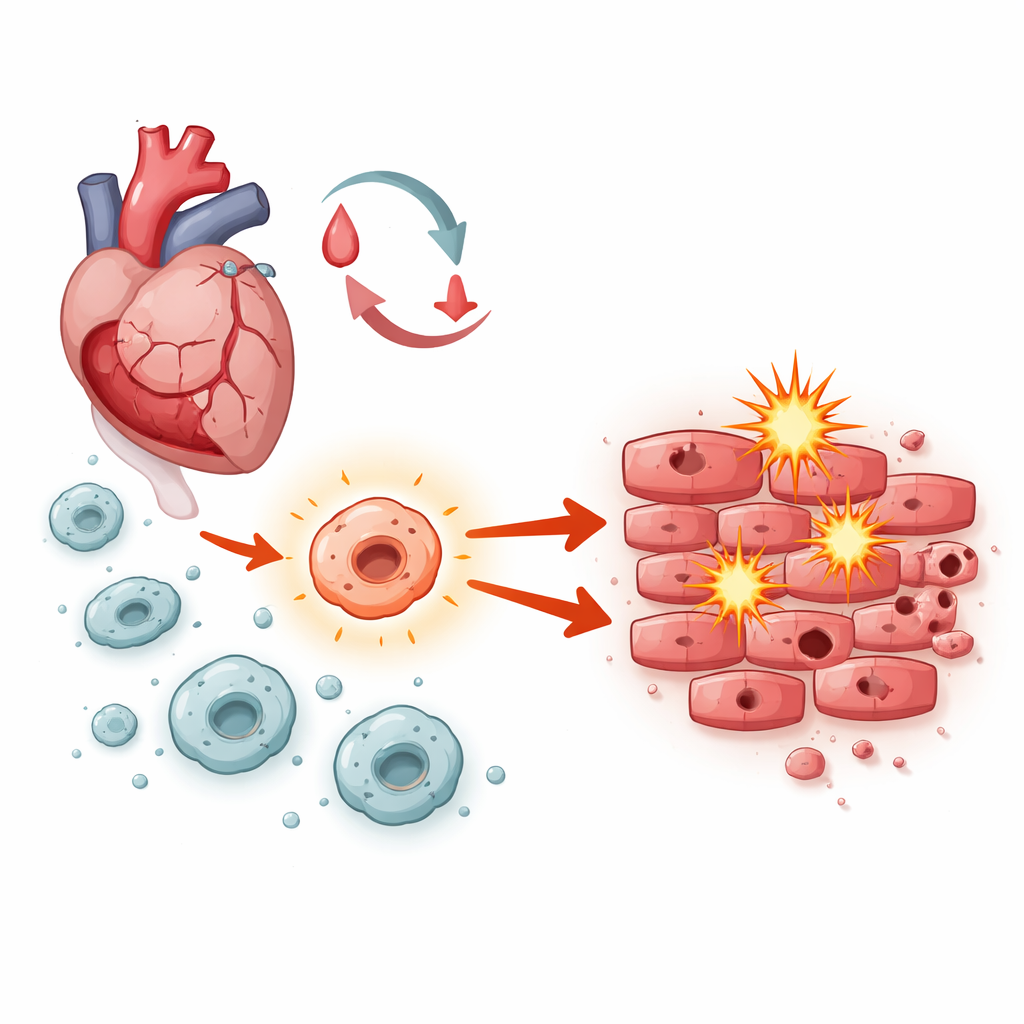
The tug-of-war inside a healing heart
After blood flow is restored, heart muscle cells face a storm of stress: sudden changes in oxygen, bursts of unstable molecules, and waves of inflammation. Macrophages—front‑line immune cells that rush into damaged tissue—play a central role in orchestrating what happens next. Depending on how they are activated, they can either help clear debris and support repair or drive swelling and cell death. The researchers focused on a signaling system inside macrophages known as Notch‑1, asking whether it tips this balance toward healing or harm during ischemia‑reperfusion injury.
Digging into genes, rats, and heart cells
The team first mined a public gene‑expression dataset from people who had suffered acute heart attacks. They identified more than 100 genes whose activity changed markedly, especially in pathways linked to inflammation and cell suicide. Notch‑related genes stood out, suggesting that this pathway was active during heart damage. The scientists then turned to rats, temporarily blocking and reopening a heart artery to mimic what happens in patients. They infused the animals with macrophages engineered to boost or alter Notch‑1 signaling, and examined heart pumping function with ultrasound, tissue structure under the microscope, and the extent of dead tissue and dying cells.
How one immune switch drives damage
Rats receiving macrophages with high Notch‑1 activity fared worse: their hearts pumped less efficiently, showed more structural disorganization, and contained larger zones of dead muscle and more dying cells. By carefully adding or blocking other molecules, the team mapped a chain of events downstream of Notch‑1 inside macrophages. In this chain, Notch‑1 dampened a gene regulator called EZH2, which in turn freed other proteins—including BRD4, HSF1, SIRPα, and SHP2—to become more active. Together, these molecules pushed macrophages into a highly inflammatory state that encouraged overproduction of reactive oxygen species, the chemically aggressive forms of oxygen that can punch holes in cell membranes and damage DNA.
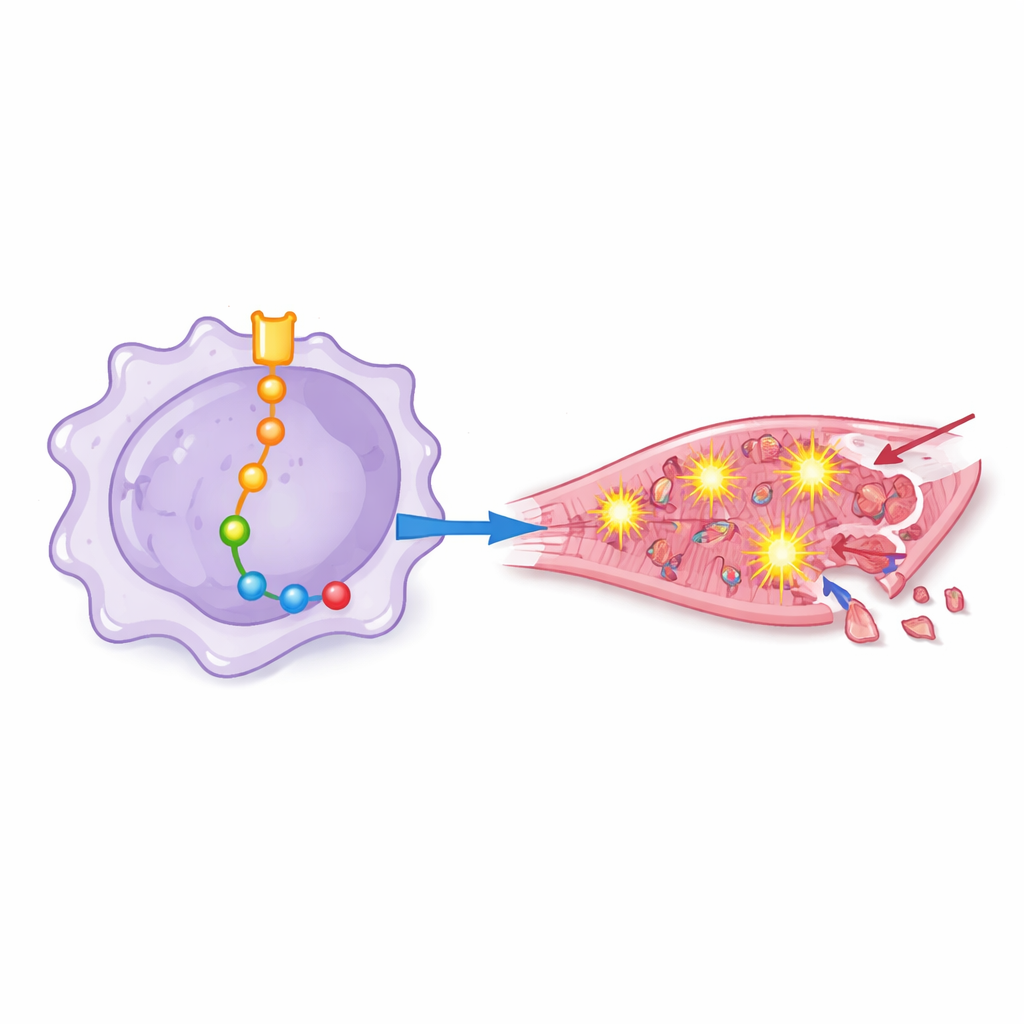
From chemical stress to heart cell death
To see how these "primed" macrophages affect human heart cells, the researchers bathed cultured cardiomyocytes in liquid taken from differently engineered macrophage cultures. Fluid from macrophages with strong Notch‑1 signaling triggered higher oxidative stress, more activation of an inflammatory protein complex known as the inflammasome, and a surge in markers of programmed cell death. When Notch‑1 or certain downstream proteins were blocked, heart cells showed fewer signs of oxidative damage, maintained better antioxidant defenses, and were less likely to die. This showed that the damaging effects are not just abstract molecular shifts but translate directly into heart cell loss.
What this means for future heart care
In simple terms, the study reveals that a signaling switch called Notch‑1 inside macrophages can push them toward a harmful mode during the critical window when blood flow returns to the heart. Through a multi‑step molecular relay involving EZH2, HSF1, BRD4, SIRPα, and SHP2, these cells flood nearby heart muscle with destructive oxygen molecules and signals that trigger cell suicide, enlarging the injury. While the work was done in cells and animals, it highlights new targets for drugs designed to calm overactive macrophages and limit collateral damage when a blocked artery is reopened—potentially helping future patients keep more of their heart muscle alive after a heart attack.
Citation: Tong, C., Zhang, J., Zuo, Y. et al. Notch-1 in macrophages promoted the ischemia-reperfusion via modulating EZH2/HSF1/BRD4/SIRPα/SHP2 induced ROS and apoptosis in cardiomyocyte. Sci Rep 16, 11020 (2026). https://doi.org/10.1038/s41598-026-40683-4
Keywords: myocardial ischemia-reperfusion, macrophages, Notch-1 signaling, oxidative stress, cardiomyocyte apoptosis