Clear Sky Science · en
A two-step mechanism for sugar translocation
How Cells Choose the Right Sugar
Every cell in your body depends on a steady supply of sugar for energy, especially the brain and rapidly growing tissues like tumors. That sugar has to cross a fatty cell membrane, and it does so through specialized “gates” called transporters. This article explores a deceptively simple question with big medical implications: when many similar sugars are present, how does a transporter decide which ones to let through and which to block? The answer turns out not to lie in the first touch between sugar and protein, but in a subtle two-step dance deep inside the membrane. 
Why Similar Sugars Behave So Differently
In humans there are 14 closely related glucose transporters that look strikingly alike where the sugar actually binds. Yet they move different sugars at different speeds and are linked to conditions ranging from diabetes to cancer. To untangle this puzzle, the authors turned to a bacterial cousin of human transporters, a protein called XylE. XylE carries the sugar xylose but binds glucose with almost the same strength, even though glucose cannot pass through and instead clogs the gate. That odd behavior makes XylE an ideal model to ask what truly distinguishes a transported sugar from one that simply jams the system.
Listening to Sugars with Magnetic Resonance
The team used a magnetic resonance method known as saturation transfer difference (STD) NMR as a kind of “stethoscope” for sugar–transporter encounters. They reconstituted purified transporters in tiny artificial membranes called liposomes, mimicking the natural environment. In STD NMR, only sugar molecules that linger close to the protein long enough pick up a detectable signal. The surprising finding was that XylE produced strong signals with xylose, its natural cargo, but almost none with glucose, even though both sugars bind. When the researchers genetically locked XylE so it could no longer change shape and transport, the xylose signal virtually disappeared. Conversely, when they engineered XylE to gain the ability to move glucose, strong glucose signals appeared. In other words, only sugar–transporter pairs that actually complete the journey across the membrane light up in this experiment. 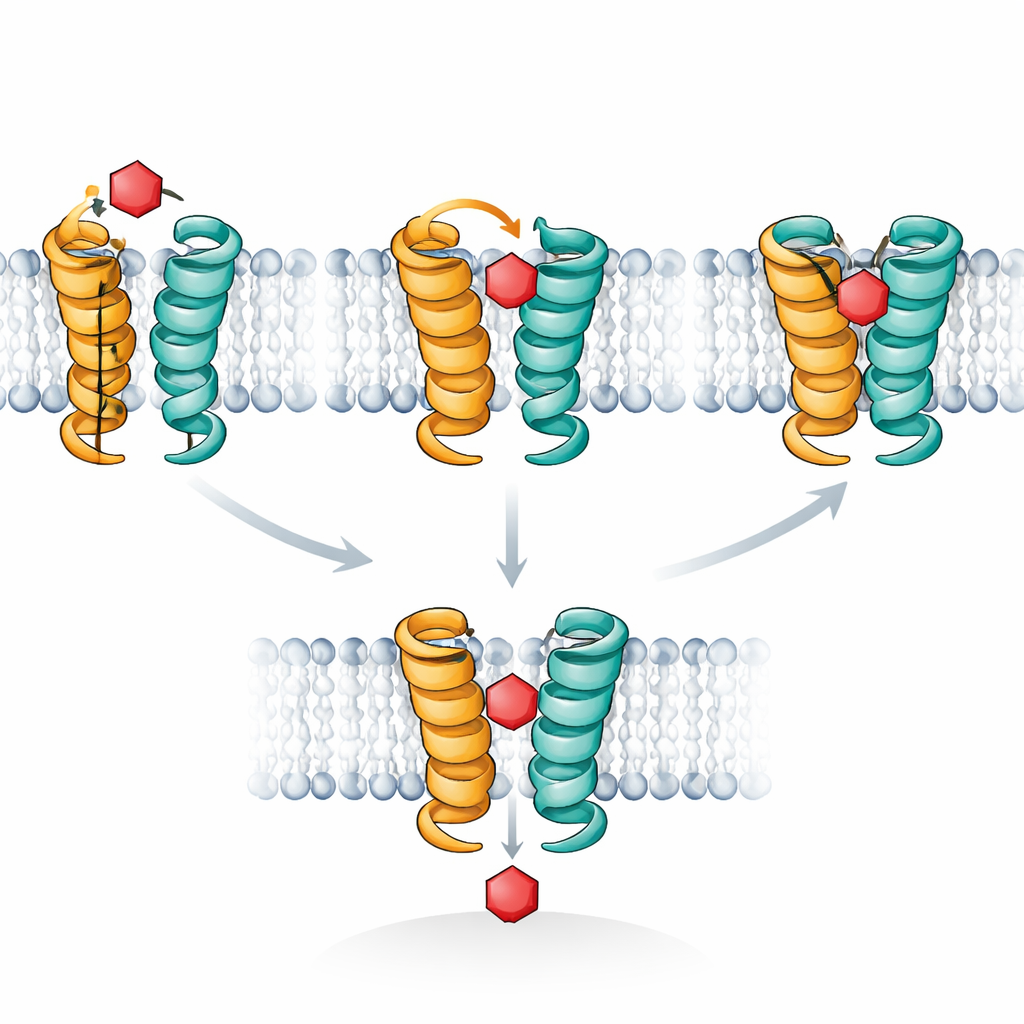
A Hidden Intermediate Step in the Journey
Computer simulations provided a molecular movie to explain these observations. XylE, like human glucose transporters, works by cycling through shapes that alternately open to the outside and inside of the cell, with an in-between “occluded” state where the sugar is sealed off from both sides. The authors focused on a short helical segment, called TM7b, that acts as an outer gate. Their simulations showed that when xylose sits in just the right pose, TM7b can swing shut fully, stabilizing the deeply occluded state where the sugar is tightly embraced. Glucose, in contrast, tends to wobble and never consistently drives TM7b to complete this closure; it remains stuck in a shallower, outward-occluded pose that blocks the pathway without finishing transport. The STD NMR signals track exactly this difference in how long sugars dwell in the fully occluded, high-contact configuration.
Lessons from a Malaria Transporter and a Fructose Specialist
To see whether this principle holds beyond XylE, the researchers turned to PfHT1, a sugar transporter from the malaria parasite that can move both glucose and fructose, and to mammalian GLUT5, a fructose specialist in the intestine. Using STD NMR, crystal structures, cryo-electron microscopy and further simulations, they found that these transporters also rely on TM7b to coordinate sugars during the occluded step. A single asparagine amino acid in TM7b plays a central role, contacting key hydroxyl groups on the sugar ring as the gate closes. In PfHT1, a high-resolution cryo-EM snapshot captured a “halfway” inward-occluded pose where the sugar is only partly coordinated; a previously determined crystal structure caught the fully occluded pose. Comparing the two showed the sugar shifting by only about two billionths of a meter, yet picking up a network of additional contacts that make the transition state-like occluded form far more tightly bound.
Transporters as Tiny Enzymes
Putting all the evidence together, the authors propose that sugar porters operate much like enzymes, with specificity governed not by the first binding step but by how well a sugar fits a fleeting transition state. Transporters spontaneously flicker into outward- and inward-occluded shapes even without sugar. A compatible sugar first “chooses” one of these pre-existing poses (conformational selection), then triggers a final tightening of the gate into a fully occluded state (induced fit) that commits the protein to flip and release the sugar on the other side. Incompatible sugars can still bind, sometimes strongly, but fail to drive this last tightening step, so they act as inhibitors rather than cargo. Recognizing that the occluded state is the true decision point suggests new ways to design drugs that freeze transporters in a transition-like pose, potentially starving cancer cells or parasites by blocking their sugar supply.
Citation: Ahn, DH., Alleva, C., Reichenbach, T. et al. A two-step mechanism for sugar translocation. Nat Struct Mol Biol 33, 652–663 (2026). https://doi.org/10.1038/s41594-026-01784-w
Keywords: glucose transporters, sugar translocation, membrane proteins, STD NMR, transition state specificity