Clear Sky Science · en
Exclusion of Notch from the contact site during efferocytosis restricts anticancer immunity
When Cleanup Crews Silence Cancer Fighters
Our bodies constantly clear away dying cells to keep tissues healthy and quiet. But inside tumors, this same clean‑up process can backfire by calming the immune system right when we want it most: during a fight against cancer. This study uncovers a hidden switch inside immune cells that decides whether cell clean‑up will lull the immune response to sleep or help wake it up to attack tumors.
The Body’s Janitors and Their Quiet Work
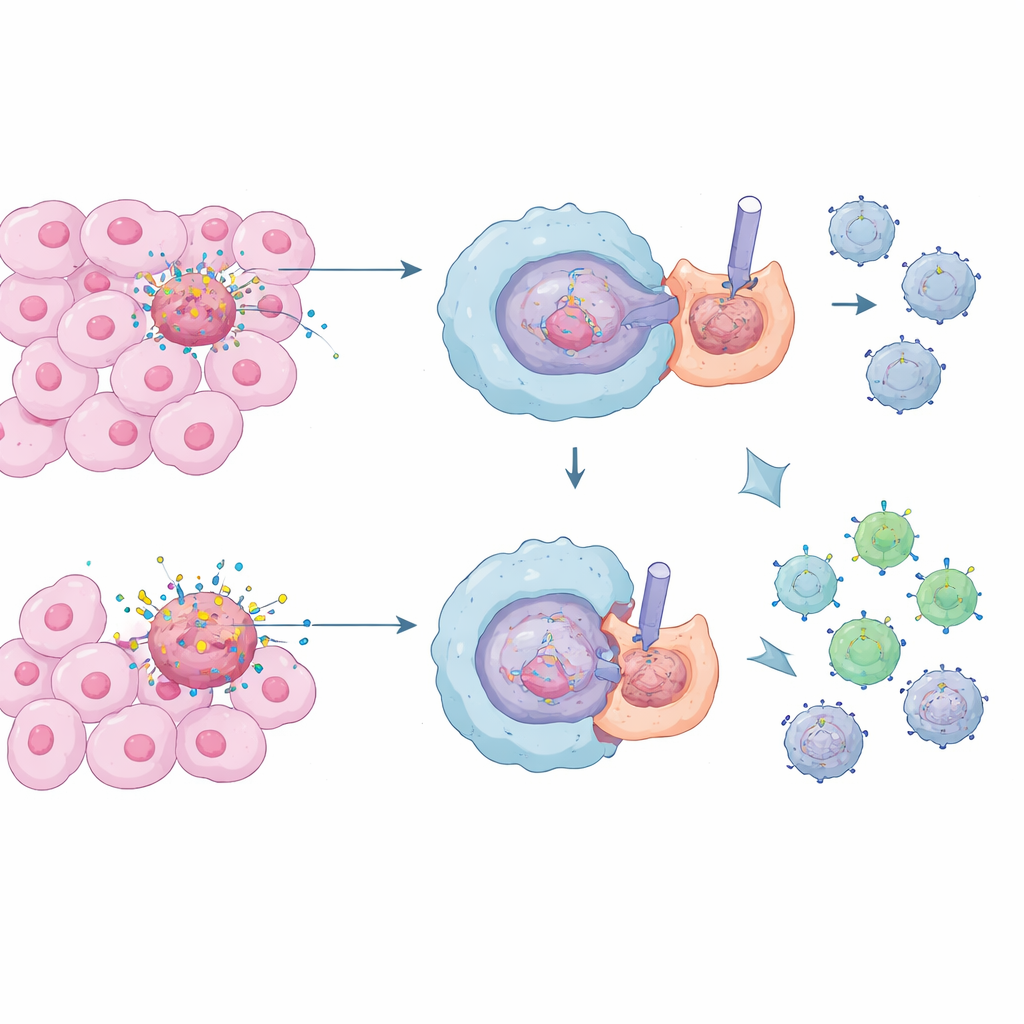
Special immune cells called macrophages act as the body’s janitors, swallowing and digesting dead and dying cells in a process known as efferocytosis. Under normal conditions this is vital: it prevents spills of toxic cell contents and keeps inflammation in check, protecting us from chronic tissue damage and autoimmune disease. Inside tumors, however, there is a constant storm of cell death. Macrophages that quietly clear these corpses can end up creating a calm, suppressive neighborhood around the cancer, making it harder for killer T cells to recognize and destroy malignant cells.
A Hidden Signal Gate on Macrophages
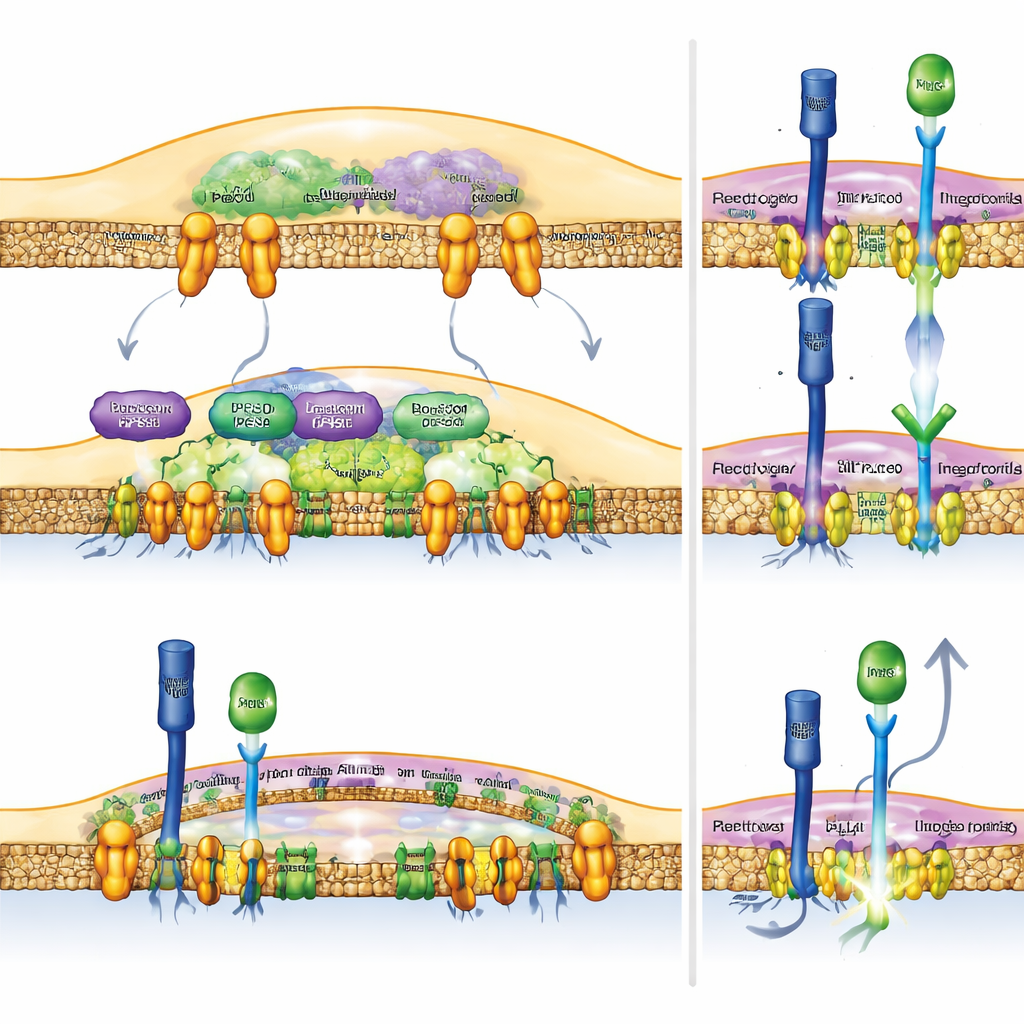
The researchers focused on a communication system called Notch, which helps cells decide how to behave based on signals from their neighbors. In macrophages, Notch activity tends to push them toward a more inflammatory, tumor‑fighting state. Surprisingly, the team found that when healthy macrophages engulf dead cells, they actively shut down Notch signaling right at the point of contact. They discovered that a protein complex built around a molecule called Rubicon (RUBCN) teams up with another enzyme, VPS34, to set up a physical “barrier” in the macrophage’s outer membrane. This barrier is made from activated integrins—gripping proteins that rearrange into a tight ring—literally pushing the bulky Notch receptors away from the patch where the dead cell touches the macrophage, so Notch cannot be switched on.
Breaking the Barrier to Boost Immune Attack
When the scientists removed Rubicon from macrophages, or blocked a downstream enzyme called phospholipase D (PLD), this integrin ring no longer formed properly. Without the barrier, Notch receptors could move into the contact zone and engage their matching ligands on the dead cell surface. In mice, tumors growing in animals that lacked Rubicon in myeloid cells—or that were treated with PLD‑blocking drugs—showed stronger Notch activity inside macrophages, more inflammatory gene programs, and a surge of cancer‑killing CD8 T cells and natural killer cells infiltrating the tumors. As a result, tumors grew more slowly.
Notch as a Lever for Cancer Immunity
To test whether turning up Notch directly in macrophages is enough to help the immune system, the team engineered mice whose myeloid cells continuously produce the active part of the Notch receptor. These mice mounted more powerful antitumor responses: their tumors were smaller, and T cells inside the tumors carried more of the molecular weapons needed to destroy cancer cells. Importantly, when the researchers disabled RBPJ—a key partner that Notch needs to turn on its target genes—the benefits of Rubicon loss or PLD inhibition disappeared. This shows that the antitumor effects of these interventions depend on intact Notch signaling in macrophages, rather than on unrelated side effects.
Clues from Human Tumors and Future Directions
Looking at data from thousands of human tumors, the authors found that patients whose cancers expressed high levels of Notch‑activating ligands but low levels of Rubicon tended to survive longer than those with high Rubicon levels. While this analysis cannot prove cause and effect, it fits the idea that blocking the Notch‑excluding barrier in the tumor microenvironment can tilt the balance toward more effective immune surveillance. The work suggests that drugs targeting Rubicon‑dependent efferocytosis pathways or PLD might one day be combined with existing immunotherapies to help macrophages flip from quiet trash collectors into active allies against cancer.
What This Means for Patients
Put simply, the study reveals that macrophages carry a built‑in gate that normally keeps a key pro‑immune signal, Notch, away from sites where they chew up dead cells. This protects healthy tissues from unnecessary inflammation but can unintentionally shield tumors. By disrupting the gate—through genetic changes or drug treatment—the researchers could let Notch signals through, reprogramming macrophages to rally T cells and other defenders against cancer. Although much work remains before these insights translate into therapies, they point to a new strategy: rather than only pushing T cells harder, we might also rewire the cleanup crews so they stop muffling, and start amplifying, the body’s own anticancer alarm.
Citation: Li, Z., Xu, B., Sharma, P. et al. Exclusion of Notch from the contact site during efferocytosis restricts anticancer immunity. Nat Immunol 27, 750–761 (2026). https://doi.org/10.1038/s41590-026-02452-3
Keywords: efferocytosis, Notch signaling, macrophages, tumor microenvironment, cancer immunotherapy