Clear Sky Science · en
Hetero-oligomerization drives structural plasticity of eukaryotic peroxiredoxins
Proteins that reshape themselves to protect our cells
Inside every cell, a quiet battle rages against reactive oxygen molecules that can damage DNA, proteins and fats. A family of enzymes called peroxiredoxins acts as a frontline defense, detoxifying these reactive species and helping cells sense when oxidation levels change. For years, scientists thought each peroxiredoxin worked alone in uniform clusters made of identical building blocks. This study overturns that view by showing that many peroxiredoxins actually mix and match with closely related siblings, forming hybrid assemblies that change shape and stability in ways that could fine‑tune how cells cope with stress.
Why protein teamwork matters
Peroxiredoxins are among the most abundant proteins in organisms ranging from yeast to humans and plants. They remove hydrogen peroxide, a reactive by-product of metabolism, and also act as molecular chaperones that help other proteins keep their shape. The classic textbook picture shows these enzymes assembling into either pairs (dimers) or doughnut-shaped rings of ten units (decamers), but always built from one isoform at a time. Yet many cells carry two nearly twin versions of the same peroxiredoxin in the same compartment. The authors asked a simple but fundamental question: do these isoforms really stay separate, or do they assemble into mixed complexes with new properties?
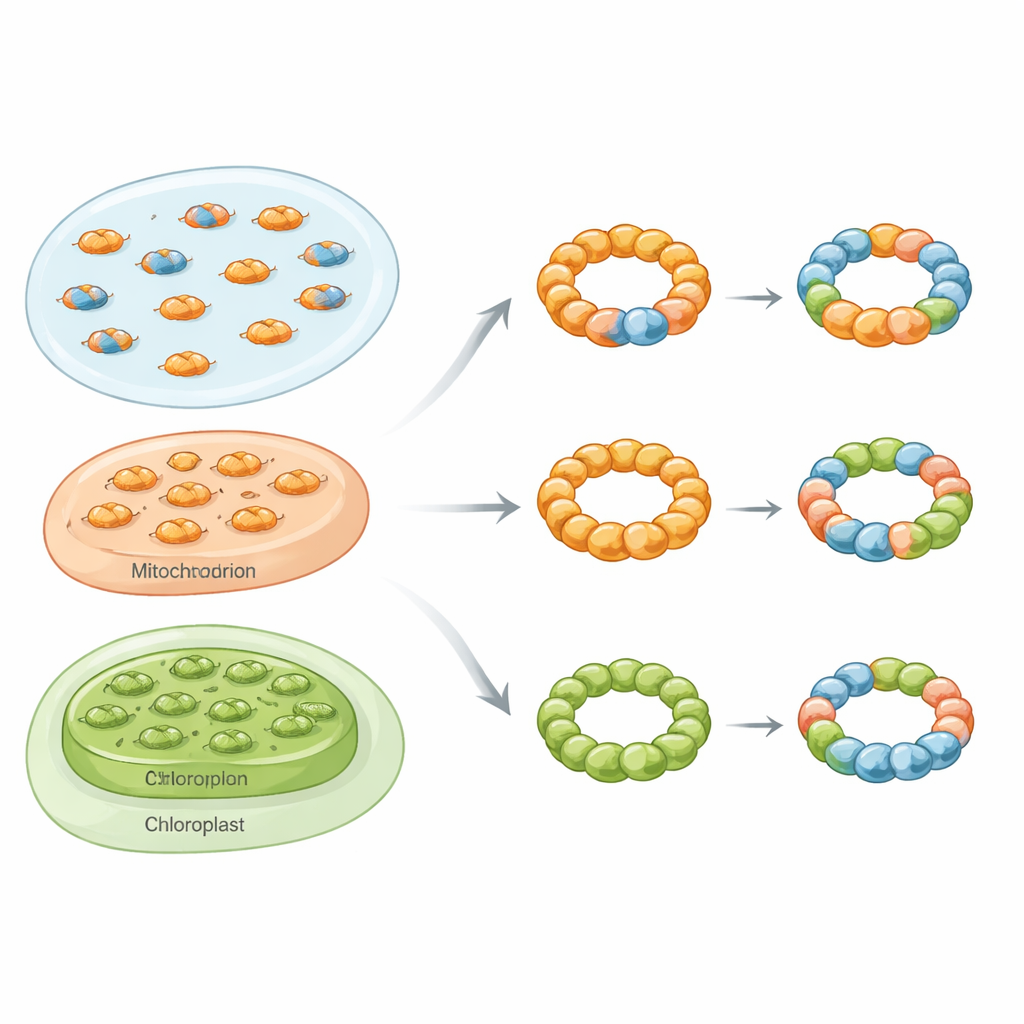
Mixed rings across kingdoms of life
Using a toolkit that included biochemical purification, native mass photometry, electron microscopy and live-cell fluorescence assays, the researchers show that mixed, or hetero‑oligomeric, peroxiredoxin complexes are not rare curiosities. In baker’s yeast, they focused on two cytosolic isoforms called Tsa1 and Tsa2. When the two proteins were produced together in bacteria, they formed both mixed dimers and mixed decamers with a spectrum of subunit ratios. Under the electron microscope these assemblies appeared as characteristic ring-shaped particles, confirming that Tsa1 and Tsa2 can share the same structural scaffold instead of forming only separate rings.
Stress-triggered reshaping in yeast cells
To test whether this mixing matters inside living cells, the team tagged Tsa1 and Tsa2 with a fluorescent sensor and followed their behavior in yeast. Under normal growth conditions, Tsa1 is plentiful whereas Tsa2 is scarce, and Tsa1 sits mostly in smaller forms. When the cells were exposed to hydrogen peroxide, production of Tsa2 shot up, and clear bands corresponding to larger ring-shaped assemblies appeared on native gels. Intriguingly, even the incorporation of just one or two Tsa2 units into a Tsa1 ring was enough to stabilize the ten-unit structure against falling apart when warmed. Enzyme kinetics showed that these hybrid rings remained fully active, with reaction rates similar to the pure forms but with reduced tendency to become over‑oxidized and inactivated.
A widespread strategy for tuning protein function
The story did not stop with yeast. In human kidney cells, the authors separated two closely related peroxiredoxins, PRDX1 and PRDX2, by ion-exchange chromatography. In cells lacking one isoform, each remaining protein eluted as a distinct species. In normal cells, however, both isoforms co‑eluted over a shared range, consistent with the presence of mixed complexes. Similar tests in yeast engineered to produce human, plant and parasite peroxiredoxins showed that pairs from all these organisms could form functional hybrids. In plant chloroplast enzymes and in a parasite that causes leishmaniasis, introducing a more “decamer-loving” isoform could pull a more dimeric partner into larger rings, or conversely destabilize rings depending on which subunit dominated. Bioinformatics analyses of more than 1,500 eukaryotic genomes revealed that over 80% of species likely harbor multiple Prx1-type peroxiredoxins in the same compartment, suggesting that such mixing is the rule rather than the exception.
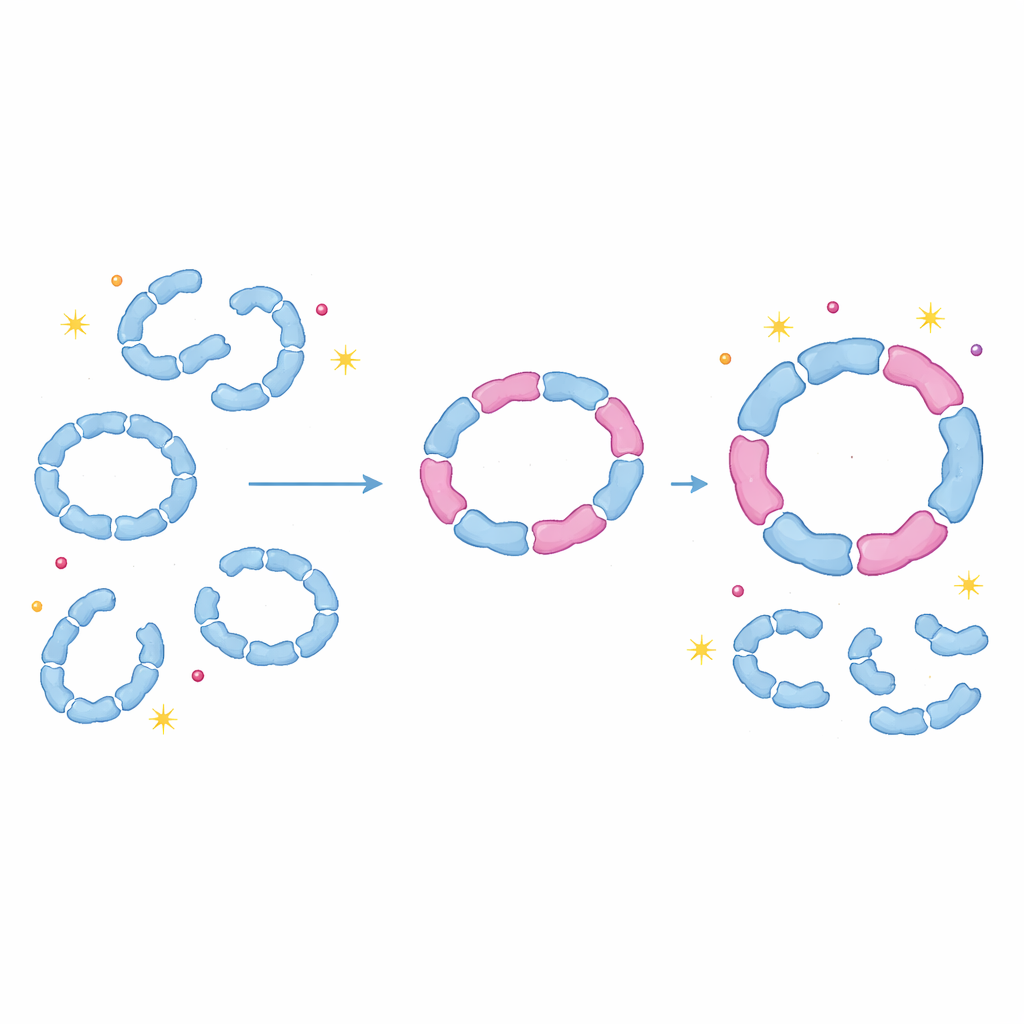
Proteins as adjustable shock absorbers
By showing that peroxiredoxins routinely build mixed rings and dimers, this work challenges the long-standing assumption that each isoform functions only as a pure assembly. Instead, cells appear to use hetero‑oligomerization as a flexible way to adjust how stable these rings are, how sensitive they are to damage and when they switch between detoxifying and chaperone roles. A small change in expression of one isoform, for example during oxidative stress, can ripple through the system by reshaping many mixed complexes at once. For non-specialists, the key message is that protein complexes behave less like rigid Lego models and more like adjustable shock absorbers: by swapping in different but compatible parts, cells can subtly tune their protection against oxidative stress, with implications for aging, cancer, infection and other conditions where redox balance goes awry.
Citation: Zimmermann, J., Lang, L., Malo Pueyo, J. et al. Hetero-oligomerization drives structural plasticity of eukaryotic peroxiredoxins. Nat Chem Biol 22, 580–592 (2026). https://doi.org/10.1038/s41589-026-02157-6
Keywords: peroxiredoxin, oxidative stress, protein complexes, redox signaling, cellular adaptation