Clear Sky Science · en
Human hippocampal neurogenesis in adulthood, ageing and Alzheimer’s disease
Why new brain cells in adults matter
For decades, scientists have argued over a simple but profound question: do adult humans grow new brain cells in the memory center of the brain, and if so, does this help protect us from memory loss and Alzheimer’s disease? This study tackles that debate head-on by combining cutting‑edge single‑cell tools and post‑mortem human brain tissue to show how newborn neurons in the hippocampus behave in healthy adults, in people on the path to Alzheimer’s, in patients with the disease, and in rare older adults with exceptional memory known as SuperAgers.
The brain’s memory nursery
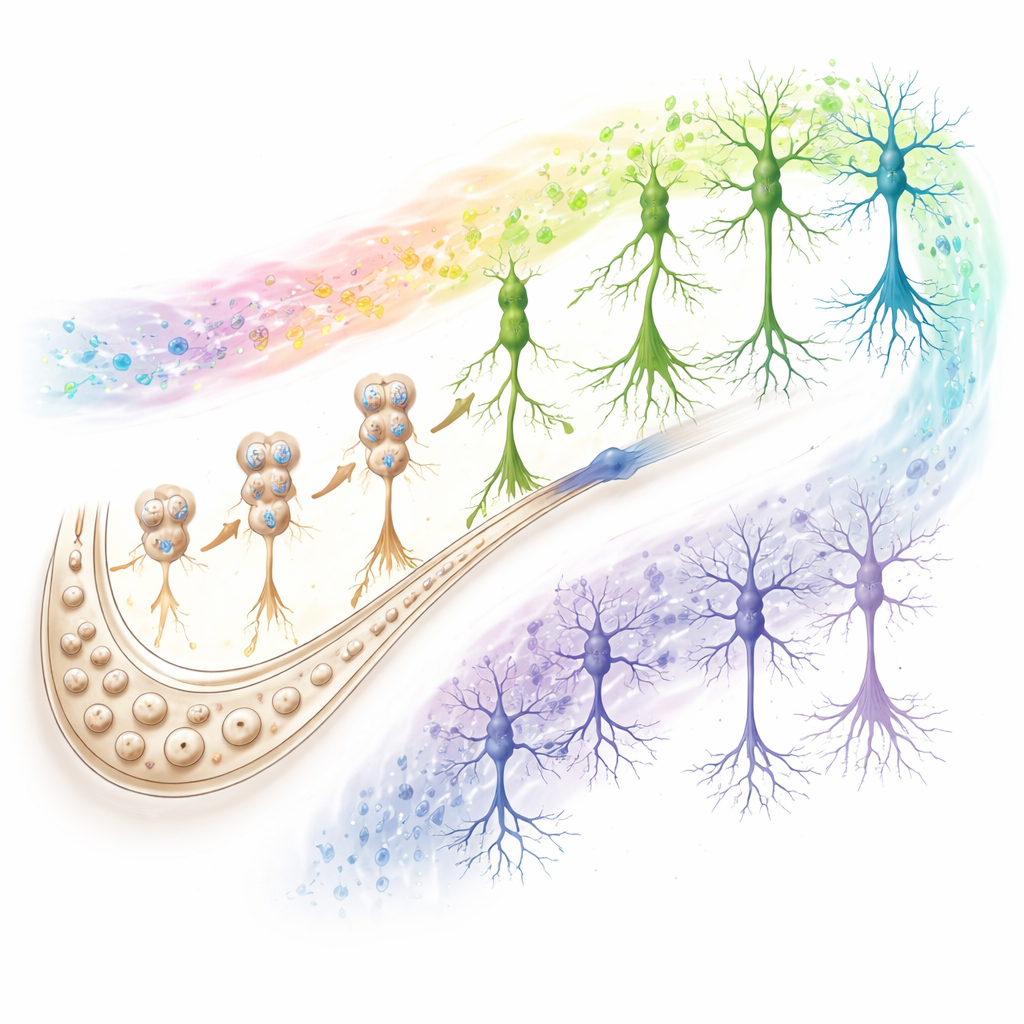
Deep in the brain’s hippocampus lies the dentate gyrus, a kind of memory “nursery” where new neurons can arise from resident stem cells. The authors dissected this region from the brains of 38 deceased donors, ranging from young adults to older adults with varying levels of memory performance and Alzheimer’s‑related changes. Using multiomic single‑cell sequencing—which reads both gene activity and how tightly DNA is packaged—they analyzed more than 350,000 individual cell nuclei. This allowed them to reliably distinguish neural stem cells, neuroblasts (early newborn cells) and immature granule neurons, and to map how these progress into fully mature neurons that join memory circuits. Their data firmly support the existence of a stepwise developmental trajectory of new neurons in the adult human hippocampus.
How age and Alzheimer’s bend the trajectory
Comparing young adults with older groups revealed that aging and Alzheimer’s do not simply turn neurogenesis on or off; instead, they reshape it. In people with preclinical intermediate pathology—individuals who seem to be transitioning from normal aging toward Alzheimer’s—the number of stem cells actually increases, but the later stages of neurogenesis begin to falter. In full Alzheimer’s disease, neuroblasts and immature neurons are markedly reduced, even though stem‑cell‑like cells are more abundant. Strikingly, the most consistent changes are not in the genes themselves, but in regions of DNA that become more or less accessible, like gates that open or close to control which genes can be used. These epigenetic shifts, especially in regions linked to synapses and neuronal structure, emerge early and intensify with disease, marking them as potential early warning signs and therapeutic targets.
The molecular fingerprints of resilience
SuperAgers—people in their 80s and beyond who remember as well as much younger adults—offered a window into resilience. Their hippocampi contained significantly more neuroblasts and immature neurons than those of Alzheimer’s patients, and often more than typical older adults. Rather than massive changes in stem cells, the key differences lay in thousands of DNA regions that were more open in neuroblasts and immature neurons, supporting robust growth and synaptic function. The team defined a “resilience score” capturing genes and DNA regions that remain stable in young adults, healthy agers and SuperAgers but collapse in Alzheimer’s. Many of these are tied to maintaining neuronal connections and efficient communication. SuperAgers also showed a distinct pattern of regulatory networks—combinations of control proteins and DNA switches—that resembles young adults in some ways but also includes unique activators that may push newborn neurons toward a particularly robust, long‑lasting state.
Circuits that mark successful versus failing aging
The study did not stop at newborn neurons. By examining all major cell types, the authors found that signals preserving or eroding cognition are concentrated in two key players: CA1 neurons (a core output station of the hippocampus) and astrocytes (support cells that manage the chemical environment and help regulate synapses). In successful agers and SuperAgers, genes in CA1 neurons support strong synaptic transmission, energy production and cellular clean‑up pathways, while astrocytes show chromatin patterns that favor protective responses. In people on the road to Alzheimer’s, these same pathways drift in the opposite direction. Analyses of cell‑to‑cell communication highlighted weakened adhesion and glutamate‑based signaling between CA1 neurons, astrocytes and neurogenic cells in preclinical and Alzheimer’s groups, suggesting that the breakdown of excitatory synapses is a shared hallmark of cognitive decline.
What this means for protecting memory
Put together, the results paint a detailed picture: adult humans do continue to generate new hippocampal neurons, but this process is exquisitely sensitive to age and disease. The earliest and most robust changes occur at the level of chromatin accessibility—the way DNA is packaged—long before many genes visibly change their activity. In resilient brains, such as those of SuperAgers, molecular networks keep neurogenesis and synaptic communication running in a “youthful” mode despite advanced age. In vulnerable brains, these same networks shift, leading to stalled development of new neurons and fraying of hippocampal circuits. By revealing these epigenetic and cellular signatures, the study points toward strategies that might one day preserve memory: therapies aimed not just at clearing plaques, but at keeping the brain’s memory nursery open, its DNA gates properly tuned, and its synapses resilient.
Citation: Disouky, A., Sanborn, M.A., Sabitha, K.R. et al. Human hippocampal neurogenesis in adulthood, ageing and Alzheimer’s disease. Nature 652, 1264–1273 (2026). https://doi.org/10.1038/s41586-026-10169-4
Keywords: hippocampal neurogenesis, cognitive resilience, SuperAgers, epigenetics, Alzheimer’s disease