Clear Sky Science · en
Spontaneous switching in a protein signalling array reveals near-critical cooperativity
How Bacteria Make Split-Second Decisions
Bacteria may be tiny, but they constantly make life-or-death decisions: swim toward food, flee from toxins, or keep going straight. This study looks inside the molecular "antenna" that lets the gut bacterium Escherichia coli sense chemicals and steer. The authors show that these antennae operate right on the knife edge between order and disorder—a physical regime known as criticality—allowing bacteria to respond strongly to faint signals without becoming sluggish.
A Molecular Control Room on the Cell Surface
Instead of using single, isolated receptors, E. coli gathers thousands of sensing proteins into a flat, crystalline patch at its cell surface. These patches, called chemosensory arrays, detect molecules in the environment and control the motors that spin the bacterium’s flagella. When attractant molecules bind to the receptors, an internal enzyme network turns down a kinase that normally promotes tumbling, nudging the cell into longer, straighter runs toward better conditions. The big question has been how such large assemblies coordinate their many parts to produce precise, reliable signals.
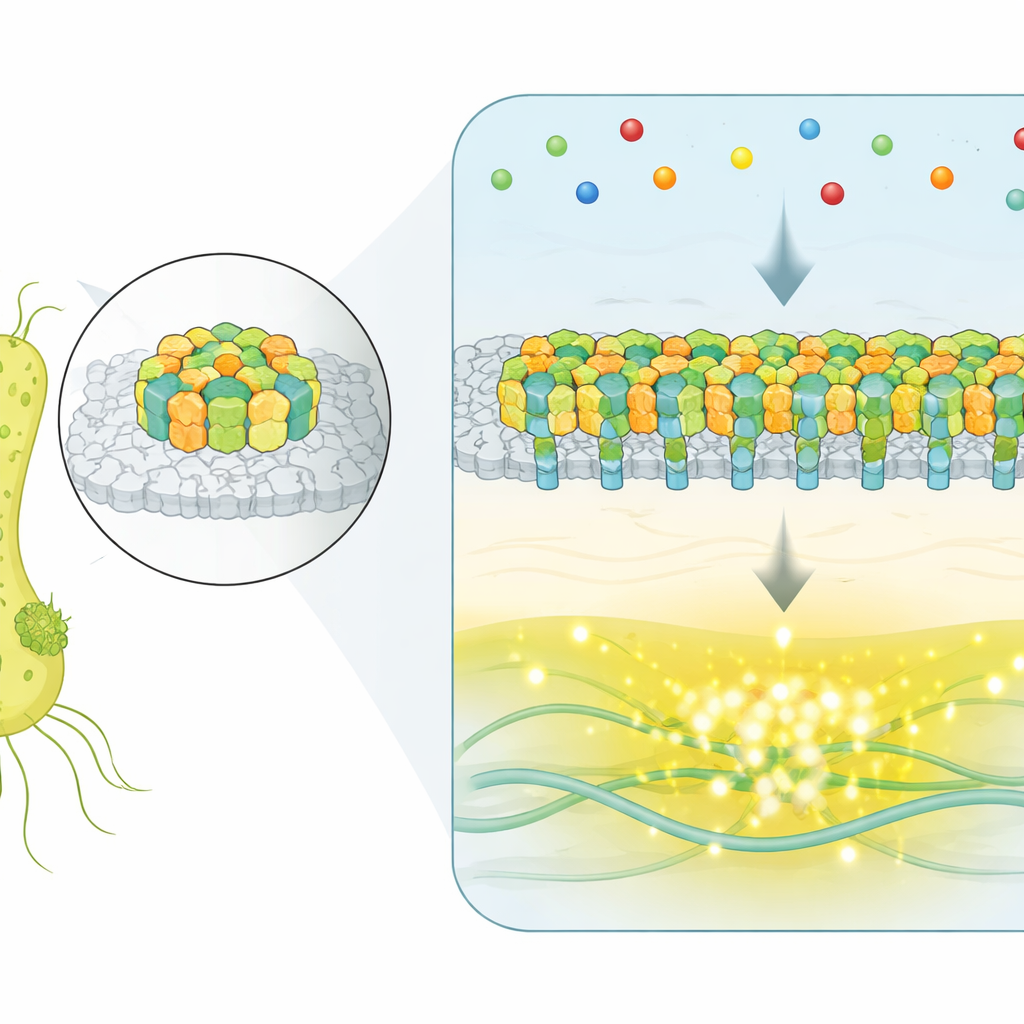
Catching Whole-Array Flips in Real Time
To watch the arrays in action inside living cells, the researchers used a fluorescence technique that reports the activity of a key signaling enzyme. They engineered strains in which a single, dominant array controls the whole cell, and then monitored thousands of individual bacteria over time, even when the external chemical environment was held constant. In a substantial fraction of cells, the output did not just flutter randomly—it snapped back and forth between two well-defined levels: an all-ON and an all-OFF state. This spontaneous two-level switching is a hallmark of cooperative systems, where many components act together rather than independently.
A Physics Model for Protein Decisions
To understand these abrupt flips, the authors turned to a classic model from physics, the Ising model, originally developed to describe how spins align in a magnet. In their adaptation, each receptor unit in the array can be active or inactive, and neighboring units prefer to match each other. Computer simulations showed that when the coupling between neighbors is weak, activity just jitters around a middle value. But when the coupling is tuned very close to a critical threshold, the whole lattice occasionally jumps between nearly all-active and nearly all-inactive states, with statistics that closely matched the experimental data. By comparing switching times and durations across many cells to these simulations, the team inferred that real bacterial arrays sit within a few percent of this critical point.
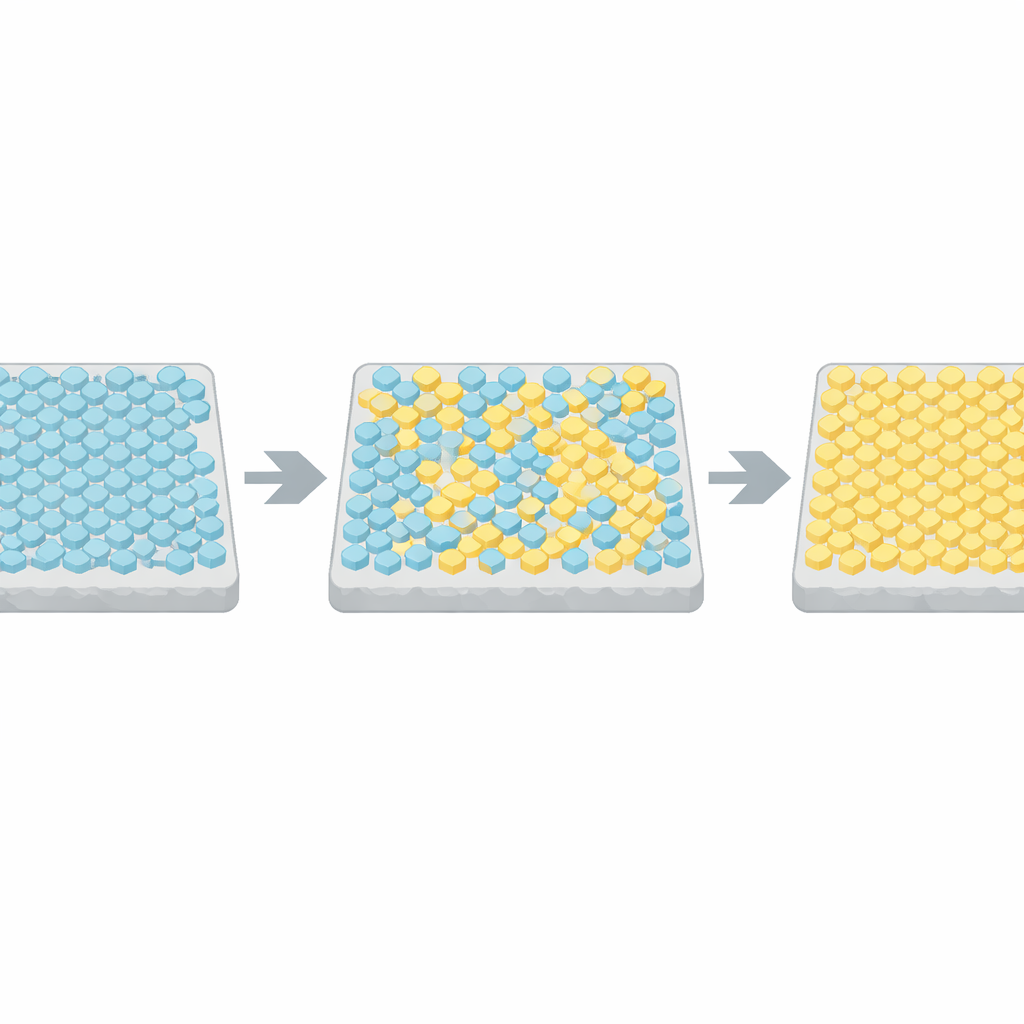
Balancing Speed and Strength of Response
Operating near criticality has a price. Strong coupling between receptors greatly amplifies small inputs, but it also slows the system’s ability to change state—a phenomenon known as critical slowing down. Simulations predicted that near-critical arrays could take tens of seconds to respond to a new stimulus, too slow for bacteria that reorient roughly once per second. The authors tested this directly by imposing rapid chemical steps using microfluidic devices. Cells engineered to have highly uniform arrays, without their normal adaptation machinery, indeed responded very slowly, confirming the trade-off between response size and response speed.
Using Disorder as a Design Feature
Wild-type bacteria, however, are not stuck with such sluggish behavior. Their arrays usually mix different receptor species, and they are constantly modified by enzymes that adjust sensitivity through chemical tags. Both effects introduce a kind of spatial and temporal "disorder" into the otherwise orderly lattice. The study’s simulations and experiments showed that this disorder effectively softens the cooperativity just enough to prevent severe slowing, while still preserving strong amplification and substantial natural fluctuations in the signaling output. These fluctuations, or noise, are thought to help E. coli explore its surroundings more effectively by varying its swimming patterns.
Life at the Edge of Order and Chaos
The authors conclude that near-critical cooperativity is a core design principle of the bacterial chemosensory array. The array is tuned to sit close to a physical tipping point where small chemical cues can trigger large, coordinated responses, but is tempered by built-in disorder so that decisions remain fast and flexible. This work links abstract ideas from statistical physics to tangible behavior in living cells, suggesting that many protein assemblies—and perhaps synthetic molecular devices of the future—may be engineered to operate at this delicate balance between sensitivity and speed.
Citation: Keegstra, J.M., Avgidis, F., Usher, E. et al. Spontaneous switching in a protein signalling array reveals near-critical cooperativity. Nat. Phys. 22, 452–460 (2026). https://doi.org/10.1038/s41567-025-03158-3
Keywords: bacterial chemotaxis, protein signaling arrays, criticality in biology, cooperative receptors, cellular decision-making