Clear Sky Science · en
Cancer-associated fibroblasts regulate DNA repair in pancreatic cancer through NDRG1-mediated R-loop processing
Why the Tumor’s Neighborhood Matters
Pancreatic cancer is notoriously difficult to treat, in part because the tumors sit inside a tough, fibrous neighborhood of support cells and proteins. This study reveals how that scar-like tissue does more than just block drugs from entering. It actively helps cancer cells repair the DNA damage caused by chemotherapy, allowing tumors to survive and return. Understanding this hidden protection system offers new ideas for making treatments work better.
A Tough Cancer in a Harsh Environment
Pancreatic ductal adenocarcinoma grows in a nutrient-poor setting packed with connective tissue known as stroma. Much of this stroma is made by cancer-associated fibroblasts, or CAFs, which secrete large amounts of extracellular matrix proteins such as collagen and laminin. Doctors have long known that stroma-rich tumors resist chemotherapy, but the exact reasons have been unclear. The researchers asked whether signals from these fibroblasts and their matrix directly change how cancer cells respond to DNA-damaging drugs.
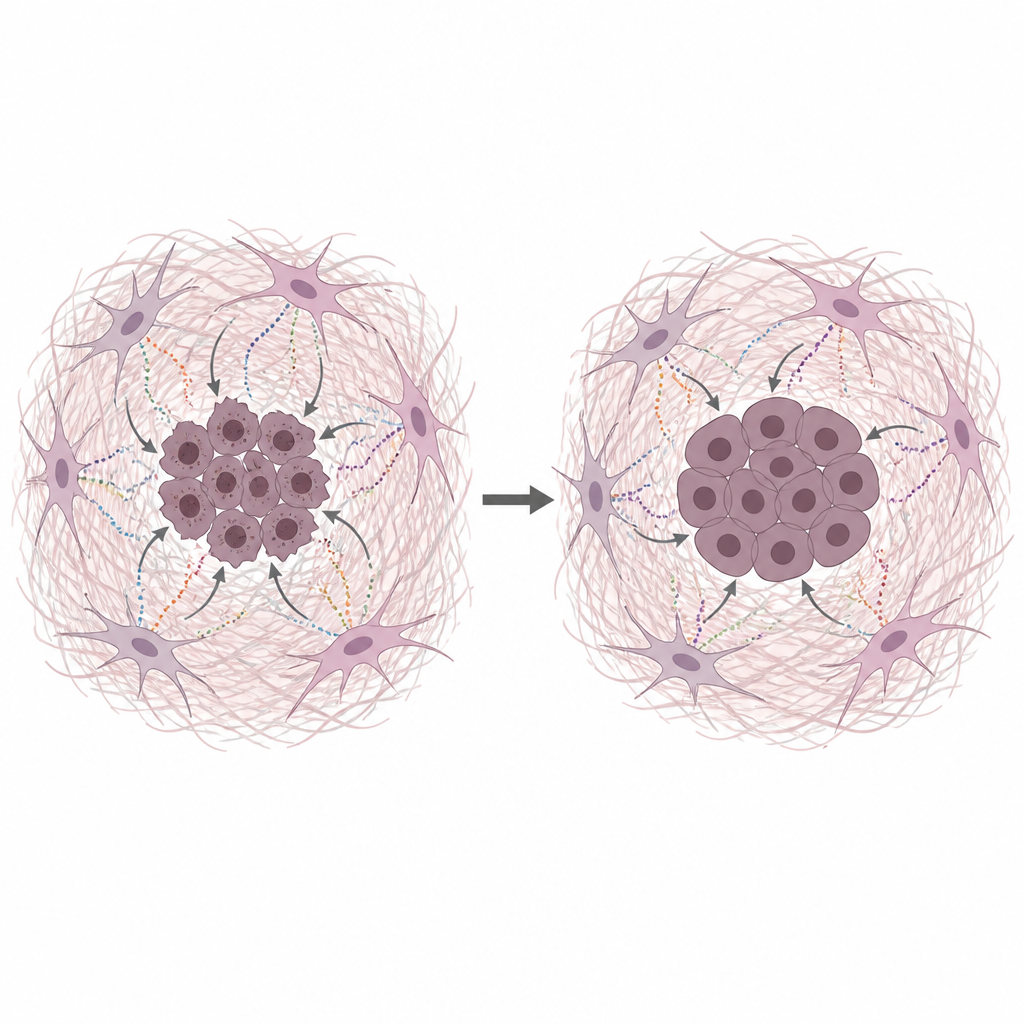
Fibroblast Secretions Shield Cancer DNA
The team grew human pancreatic cancer cells together with medium taken from starved CAFs, which mimics the nutrient-poor conditions in real tumors. This conditioned medium made cancer cells both grow more and withstand several common chemotherapies that work by damaging DNA or stalling its copying. Direct tests of DNA damage showed fewer breaks when CAF-derived medium or purified matrix proteins were present. Collagen, fibronectin and laminin each reduced DNA damage, pointing to the matrix itself, not just small nutrients, as a key protector.
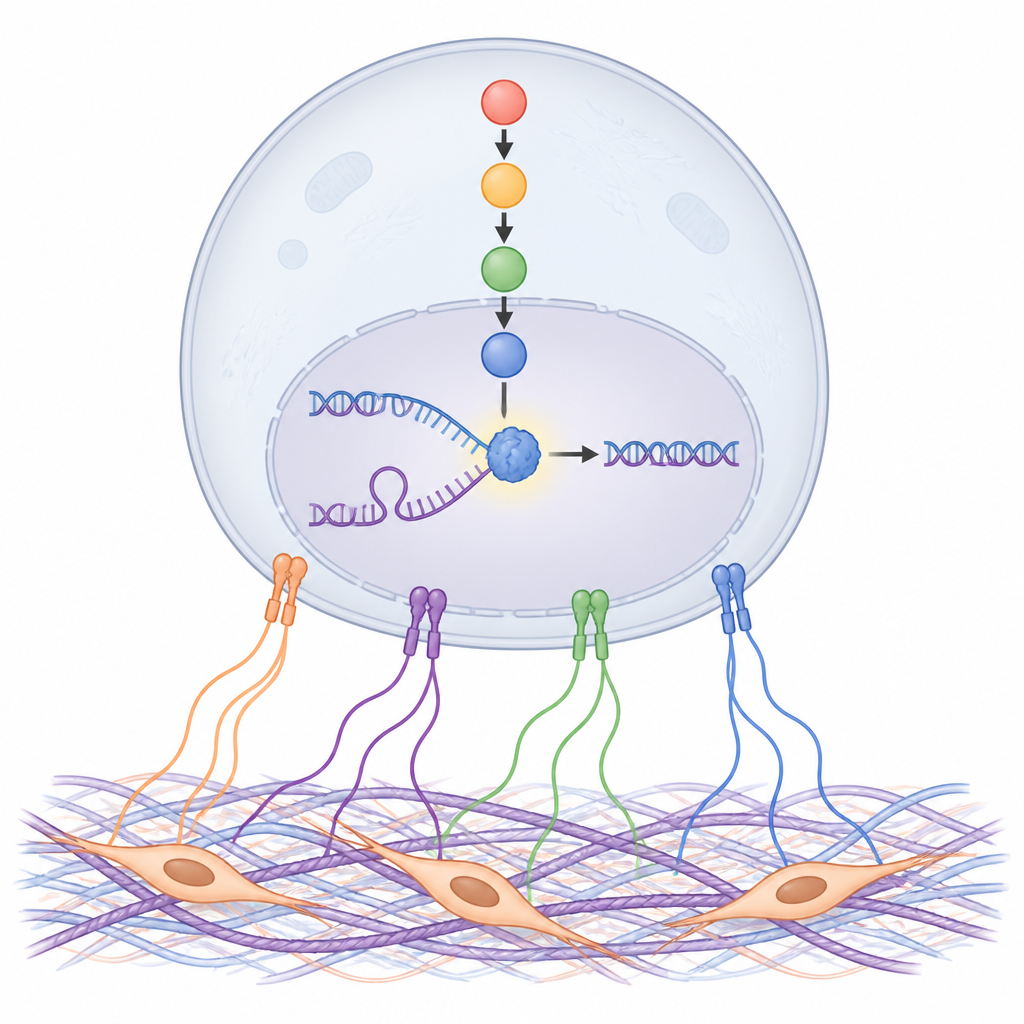
A Stress Sensor Links the Matrix to DNA Repair
To uncover how matrix signals are translated inside cancer cells, the authors surveyed hundreds of proteins. One stood out: a stress-response protein called NDRG1, especially in its switched-on, phosphorylated form. Matrix proteins and CAF-conditioned medium boosted NDRG1 activity through cell-surface adhesion receptors called integrins and a chain of signaling proteins inside the cell. Blocking this chain, particularly a kinase family called SGK, prevented NDRG1 activation. In tumor samples from patients and mouse models, high NDRG1 levels were most prominent near fibrotic stroma and were linked to faster tumor growth, higher cell division and poorer survival after chemotherapy.
NDRG1 Keeps DNA Copying on Track
The study then zoomed in on what NDRG1 actually does to DNA. Using sensitive methods to tag and pull out newly copied DNA, the researchers found NDRG1 directly at the sites where DNA is being replicated, especially when forks are stalled by drugs. Removing NDRG1, or blocking its activation by SGK, slowed DNA copying, made it harder for stalled forks to restart and increased DNA damage, both in dishes and in mice. A specific building block of NDRG1, the amino acid histidine at position 194, proved essential for this protective role, suggesting that NDRG1’s shape and possible enzyme-like activity are critical for stabilizing replication forks.
Clearing Harmful RNA–DNA Knots
One frequent cause of stalled DNA copying is the formation of R-loops, small knots where newly made RNA sticks back onto the DNA template. The authors showed that cells lacking NDRG1 or its activation accumulate these RNA–DNA hybrids and experience more collisions between the machines that copy DNA and those that read genes. When they added an enzyme that specifically removes R-loops, fork movement and DNA damage returned toward normal. This indicates that NDRG1 helps keep replication running smoothly by limiting R-loops and easing these transcription–replication conflicts.
What This Means for Future Treatments
In simple terms, the fibrotic tissue surrounding pancreatic tumors sends signals that switch on NDRG1 inside cancer cells, allowing them to patch up chemotherapy-induced DNA damage and keep dividing. Patients whose tumors express more NDRG1 tend to fare worse on standard DNA-damaging drugs. Targeting this matrix–NDRG1 pathway, or combining chemotherapy with drugs that weaken NDRG1’s repair function, could make cancer cells more vulnerable without increasing the dose of toxic treatments.
Citation: Kozlova, N., Cruz, K.A., Ruzette, A.A. et al. Cancer-associated fibroblasts regulate DNA repair in pancreatic cancer through NDRG1-mediated R-loop processing. Nat Cell Biol 28, 986–999 (2026). https://doi.org/10.1038/s41556-026-01938-4
Keywords: pancreatic cancer, tumor microenvironment, DNA repair, cancer-associated fibroblasts, R-loops