Clear Sky Science · en
Nonsense-mediated mRNA decay safeguards telomeres in pluripotent stem cells
How cells keep their chromosome caps safe
Every time a cell divides, it must copy and protect its DNA. The ends of our chromosomes are capped by special structures called telomeres that act like plastic tips on shoelaces, preventing fraying and mistaken repair. This study explores how early mouse stem cells use an unexpected backup system—an RNA "quality-control" pathway—to keep these caps intact when a major protective protein is missing, revealing a surprising link between RNA surveillance and chromosome safety.
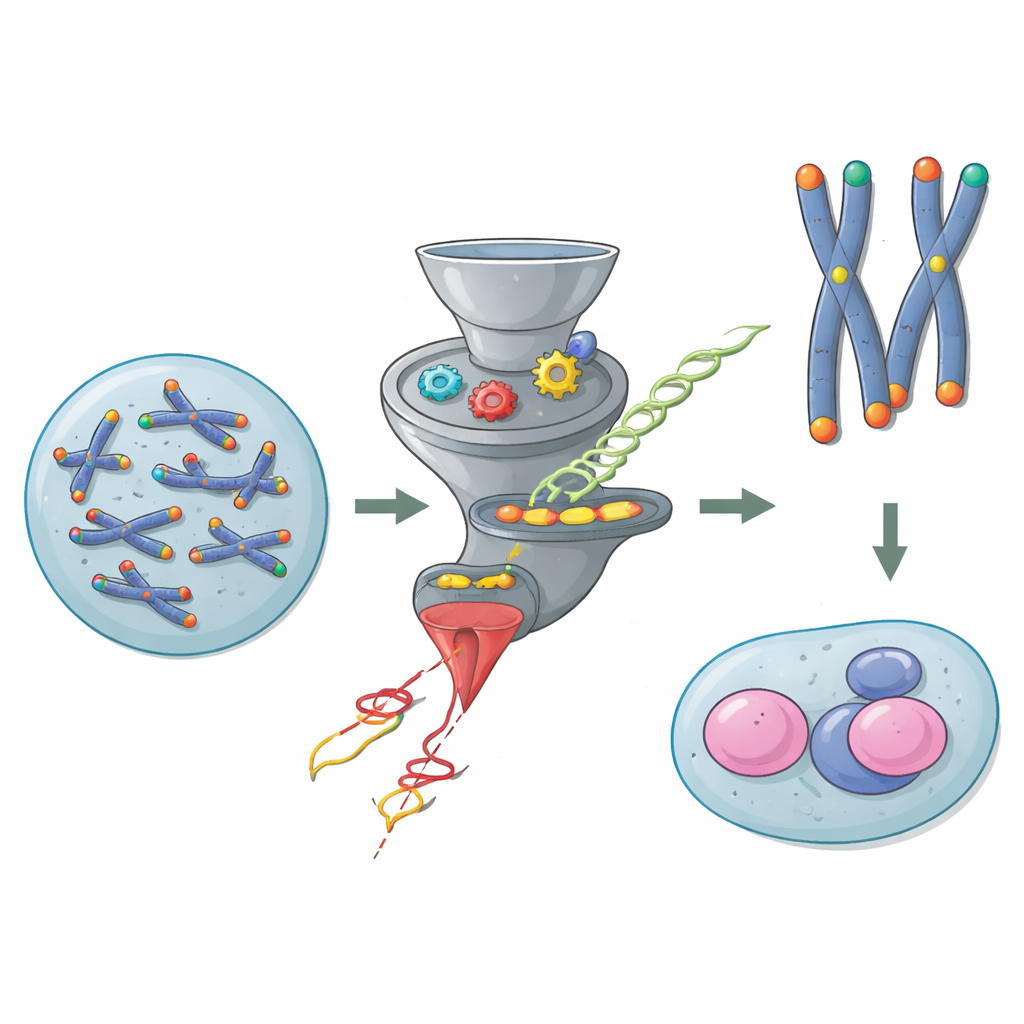
The protective caps at the ends of chromosomes
Chromosomes are long DNA molecules that need protection at their tips so they are not confused with broken DNA. Telomeres provide this protection, helped by a group of proteins known as Shelterin that coat the ends and block dangerous repair reactions. In most body cells, one Shelterin protein called TRF2 is crucial: without it, chromosome ends fuse together, DNA damage responses flare up, and genomes become unstable. Yet mouse embryonic stem cells break this rule. Previous work showed that these young cells can survive and divide even when TRF2 is removed, hinting that some hidden pathway steps in to guard their telomeres.
A genome-wide hunt for a hidden helper
To uncover this hidden safeguard, the researchers used a powerful genetic screen. They disabled the Trf2 gene in mouse embryonic stem cells and then used CRISPR–Cas9 to disrupt thousands of other genes one by one. If losing a second gene suddenly killed cells that were already missing TRF2, that second gene was flagged as a partner in "synthetic lethality"—a sign that it helps compensate for TRF2 loss. The screen recovered expected telomere factors but also highlighted several components of a seemingly unrelated pathway: nonsense-mediated mRNA decay (NMD), a system best known for eliminating faulty RNA messages that contain early stop signals.
When RNA cleanup fails, chromosome ends break
The team then tested what happens when NMD factors are removed in TRF2-deficient stem cells. Either TRF2 loss alone or NMD loss alone caused little telomere damage: chromosome ends looked mostly normal, and cells kept growing. But when TRF2 and NMD were disrupted together, the picture changed dramatically. Markers of DNA damage piled up specifically at telomeres, and chromosomes frequently fused end-to-end. These fused chromosomes coincided with strong growth defects, increased cell death, and blocked cell-cycle progression, even though the cells still retained core stem-cell features. Acute chemical or degron-based shutdown of NMD produced the same telomere-fusion phenotype, ruling out long-term adaptation and confirming that ongoing NMD activity is directly required to keep TRF2-free telomeres safe.
A harmful shortened protein undermines telomere guards
Diving deeper, the researchers examined which RNA messages are controlled by NMD in these stem cells. They found that the Trf1 gene, which encodes another telomere-binding protein, TRF1, produces an abnormal splice variant when NMD is impaired. This altered RNA skips an internal segment and carries an early stop signal, leading to production of a shortened TRF1 protein that lacks its DNA-binding tip but can still pair with normal TRF1. In NMD-deficient cells, this truncated version accumulates and pulls full-length TRF1 away from telomeres. Experiments showed that TRF1 occupancy at chromosome ends drops sharply under these conditions, and simply forcing cells to make the truncated protein was enough to trigger telomere fusions, especially when TRF2 was also missing. Conversely, boosting levels of full-length TRF1 in NMD-defective cells largely restored telomere protection, underscoring that loss of effective TRF1 at telomeres is the key problem.
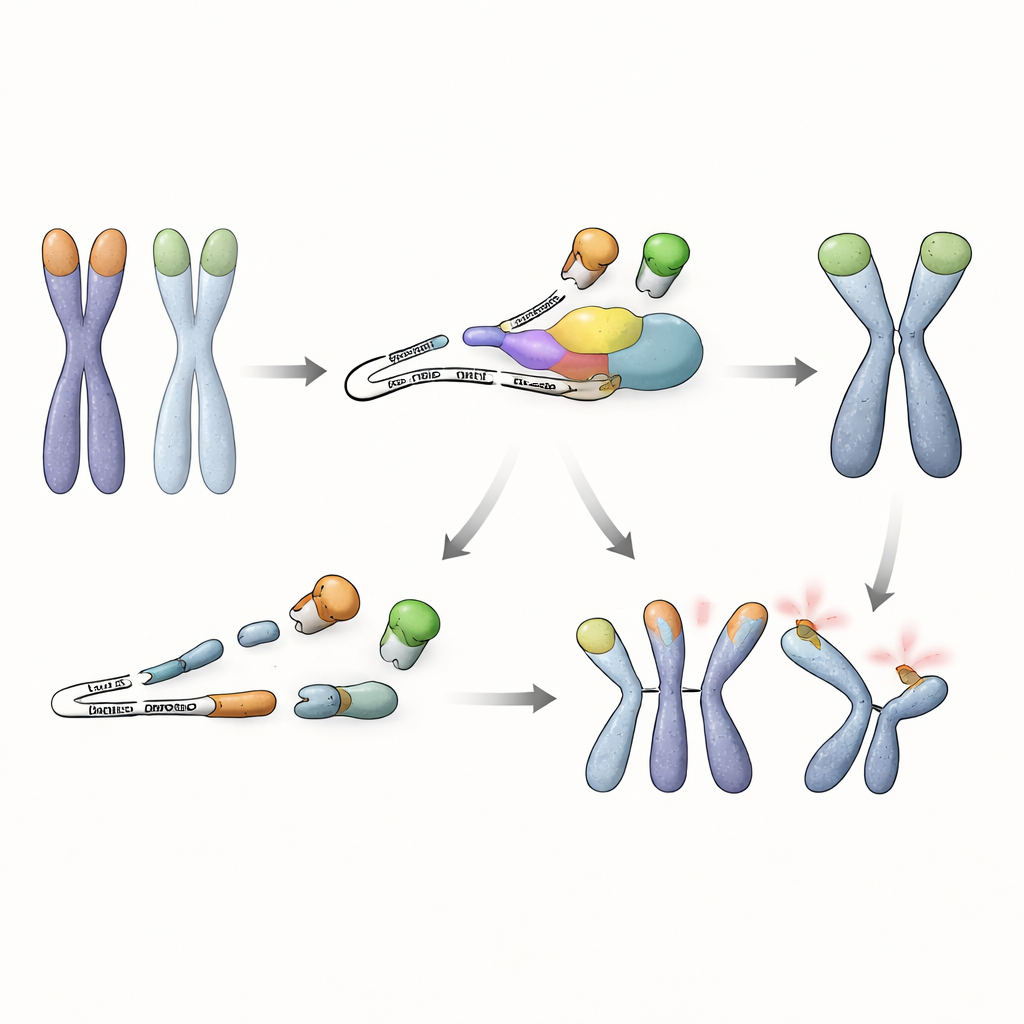
A new link between RNA surveillance and genome safety
Taken together, the work reveals that embryonic stem cells rely on more than just DNA-end–binding proteins to guard their chromosomes. They also need an RNA quality-control pathway to prevent buildup of a harmful, shortened TRF1 protein that displaces its protective full-length counterpart. As long as NMD is active, this faulty RNA is rapidly destroyed, TRF1 remains at telomeres, and stem cells can tolerate loss of TRF2 without catastrophic chromosome fusions. This discovery uncovers a previously unrecognized strategy by which early stem cells tie RNA surveillance to telomere protection, highlighting how multiple layers of control cooperate to preserve genome stability during the earliest stages of life.
Citation: Markiewicz-Potoczny, M., Lee, S.Y., Chatterjee, S. et al. Nonsense-mediated mRNA decay safeguards telomeres in pluripotent stem cells. Nat Cell Biol 28, 674–683 (2026). https://doi.org/10.1038/s41556-026-01912-0
Keywords: telomeres, pluripotent stem cells, RNA quality control, nonsense-mediated decay, genome stability