Clear Sky Science · en
Signed, sealed, delivered: a generalizable model for living biotherapeutic dosing and metabolism
Turning Helpful Germs into Medicine
Many people know that the microbes in our gut can affect health, but it is far less obvious how to turn those tiny passengers into precise, reliable medicine. This study tackles a very practical question: if we swallow a capsule of engineered “good bacteria” meant to clean up a harmful chemical in the gut, how much should we take and when should we take it? The authors build a mathematical framework that connects what happens to food as it moves through the stomach and intestines, what our resident microbes do to it, and how an added probiotic strain can step in to protect the body.
A Body Odor Disorder as a Test Case
The researchers focus on trimethylaminuria, sometimes called “fish odor syndrome.” In this condition, mutations in a liver enzyme mean that a strong-smelling compound, trimethylamine (TMA), is not properly neutralized and instead builds up in sweat, breath, and urine. TMA is mostly made in the gut when bacteria break down nutrients such as choline from common foods like eggs and meat. Current treatments try to starve the system of these nutrients or kill off gut bacteria with antibiotics, strategies that are hard to sustain and may cause side effects. The authors explore a different idea: add a live biotherapeutic product—an engineered bacterium that carries an enzyme capable of converting TMA into a harmless, odorless form directly inside the gut before it can enter the bloodstream.
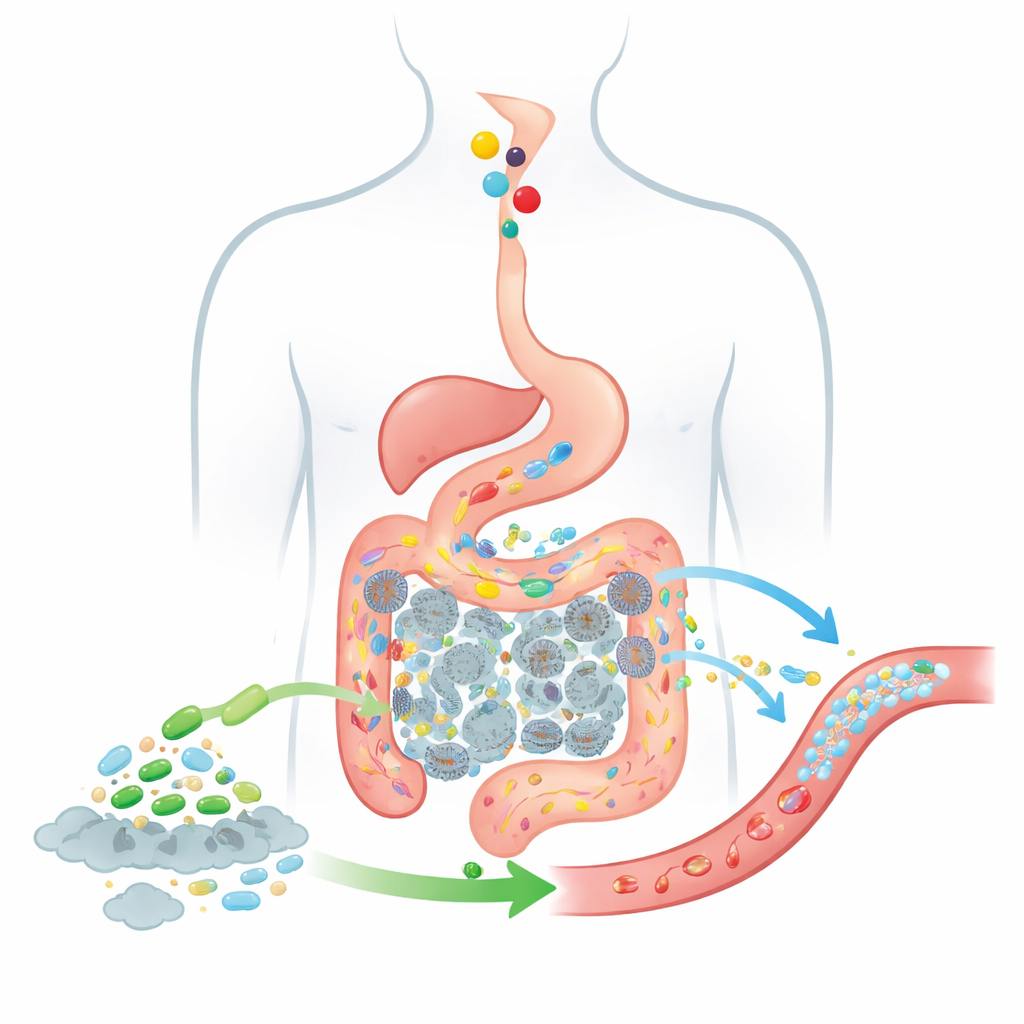
Following Food and Bacteria Through the Gut
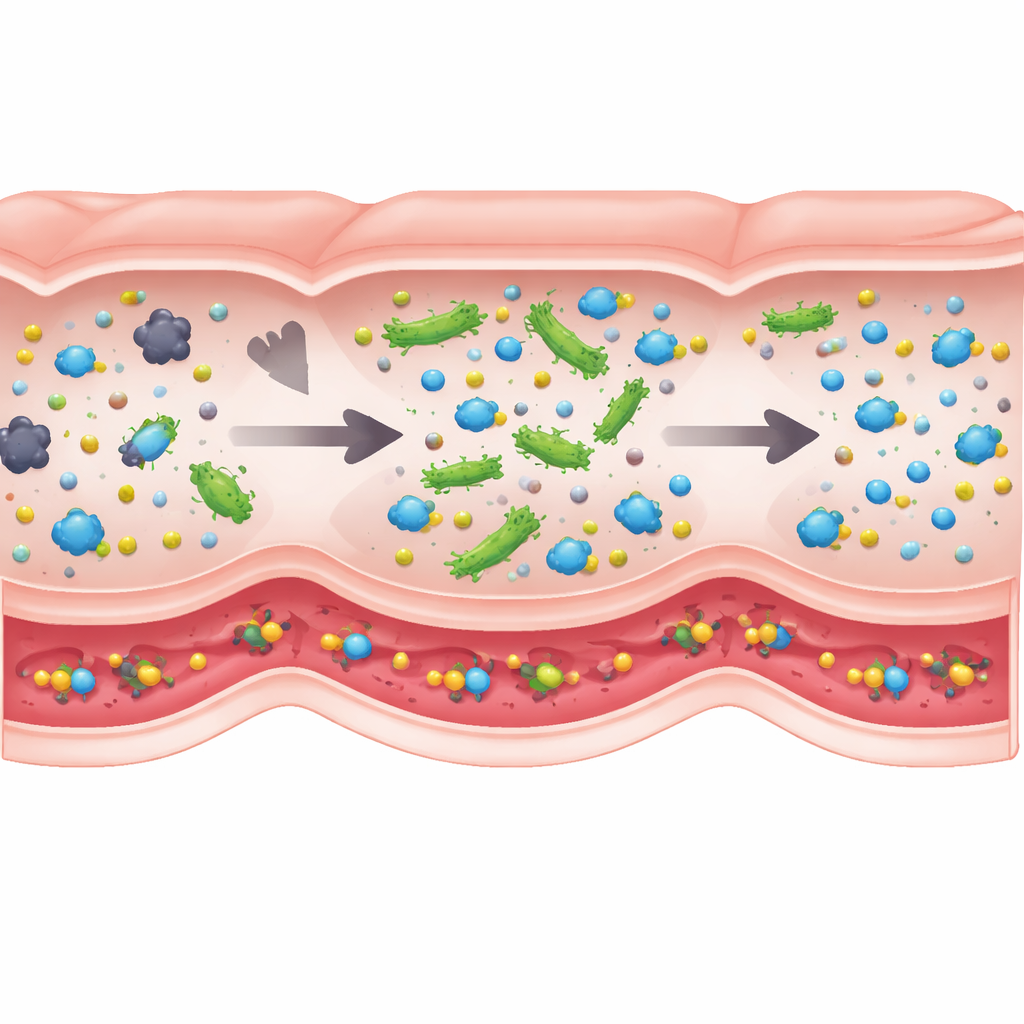
To predict how well such a living therapy might work, the authors extend a classic drug-absorption model known as the CAT model, which tracks how swallowed substances move from the stomach through segments of the small intestine and into the colon. Their updated BCAT model splits the gut into multiple connected “boxes,” each representing a stretch of intestine or colon with its own fluid volume, typical bacterial numbers, and absorption behavior. As food passes from one box to the next, the model keeps track of how much choline is present, how much TMA gut bacteria produce, how much of that TMA crosses into the blood, and how added probiotic cells travel and act along the same route. Unlike earlier models, this one treats the probiotic population as a moving, changing cloud of cells rather than a fixed background, and it explicitly includes the colon, where the vast majority of TMA is generated.
Checking the Model Against Real-World Data
A model is only useful if it matches reality, so the team tested BCAT against human data. First, they asked whether the model could reproduce classic nutrition studies from the 1950s through the 1990s, where volunteers ate measured doses of choline and researchers later collected and analyzed the TMA and its neutralized form in urine. Across several dose levels and independent studies, the fraction of choline turned into trimethylamines hovered around two-thirds; BCAT successfully captured this pattern. Next, they applied the framework to a different engineered probiotic, SYNB1618, designed for the inherited disease phenylketonuria. Using clinical trial data on how much of a diagnostic breakdown product appeared in subjects’ urine, BCAT predictions differed by only about 5 percent, far outperforming simpler, low-resolution models.
How Much Probiotic and When to Take It?
With this validation in hand, the authors turned BCAT loose on trimethylaminuria. They asked: What is the smallest probiotic dose that can cut total TMA exposure in the blood by 95 percent, roughly matching the natural cleanup power of a healthy liver enzyme? When probiotic and meal are taken together, the model suggests a dose around ten billion cells, similar to what fits in a typical over-the-counter probiotic capsule. But timing matters. Because resident gut bacteria are already established throughout the colon while swallowed probiotics must travel there from the stomach, there is a delay before the therapy is in the right place. Simulations show that taking the probiotic about three to four hours before a choline-rich meal allows it to “arrive early,” spread through the colon, and intercept TMA more effectively. Under this schedule, the required dose drops by roughly a factor of four, which could lower safety concerns about very high microbial loads.
What Matters Most Under the Hood
Not every biological detail influences the outcome equally, so the authors used a type of global sensitivity analysis to see which ingredients of the model actually drive success. The clear winner was the amount of active enzyme each probiotic cell can carry. Changes in gut transit times, absorption rates, or even overall bacterial density had comparatively small effects within realistic ranges. This means that when designing such live biotherapeutics, engineering cells that make more of the key enzyme may be far more important than fine-tuning how fast they move through the gut. It also suggests that dosing recommendations from BCAT should be fairly robust across different people, even though their digestion speeds and microbiomes vary.
From One Disorder to Many
In the end, this work does not just offer guidance for a rare and socially distressing odor disorder. It introduces a general, adaptable framework for planning and optimizing live microbial therapies for any condition where the gut microbiome transforms dietary or body-derived compounds into harmful byproducts. By knitting together gut transit, native microbial chemistry, and engineered enzymatic cleanup into one quantitative picture, the BCAT model can suggest realistic dosing targets and timing strategies long before a strain reaches clinical trials. For patients and clinicians, this brings the promise that future probiotic medicines will be not only friendly and natural-sounding, but also predictable, tunable, and grounded in the same kind of rigorous planning used for conventional drugs.
Citation: DeVito, V.L., Karamched, B.R. Signed, sealed, delivered: a generalizable model for living biotherapeutic dosing and metabolism. npj Syst Biol Appl 12, 48 (2026). https://doi.org/10.1038/s41540-026-00685-4
Keywords: gut microbiome, probiotic therapy, trimethylaminuria, mathematical modeling, live biotherapeutics