Clear Sky Science · en
Targeted degradation of USP7 in solid cancer cells reveals distinct effects of deubiquitinase degraders and inhibitors
Why breaking down one protein matters for cancer
Cancer treatments often aim to block the activity of harmful proteins, but an emerging idea is to remove those proteins from cells altogether. This study looks at a protein called USP7, which helps other proteins avoid natural breakdown, and asks a simple question with big consequences: is it better to switch USP7 off temporarily, or to get rid of it entirely? By comparing these two strategies in solid tumor cells from pancreatic cancer and melanoma, the researchers uncover important differences that matter for designing safer and more precise drugs. 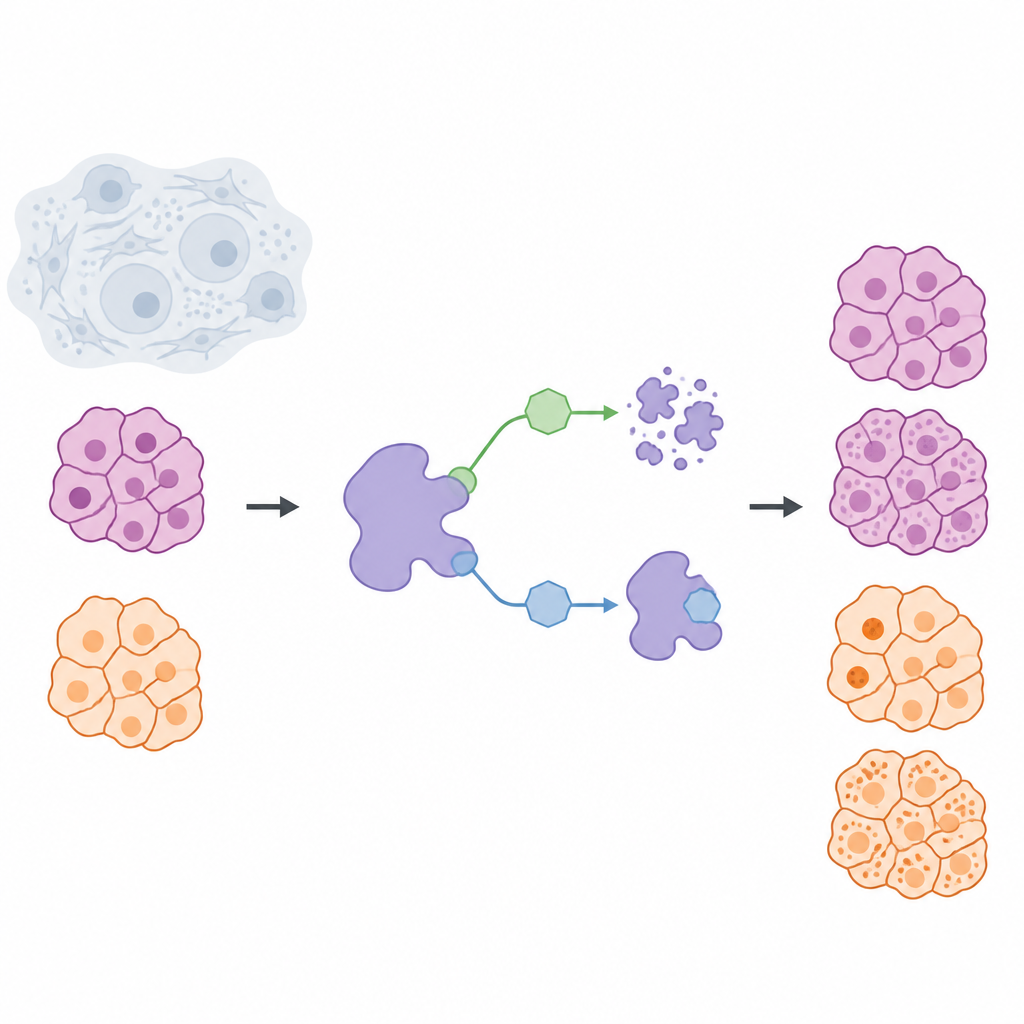
Two different tools to control the same target
The team built a chemical toolbox to control USP7 in living cells in two distinct ways. One set of molecules, called inhibitors, latch onto USP7 and stop its activity while the protein itself remains in place. The second set, called degraders, act more like guided removal tags: they bring USP7 into contact with the cell’s waste-disposal machinery so the protein is broken down and cleared. To make this comparison fair, the scientists first designed a highly selective inhibitor, NK192, that binds USP7 strongly and rarely touches other proteins. From this scaffold they then engineered a series of degrader molecules that can both recognize USP7 and recruit a cellular disposal system known as VHL, allowing them to strip USP7 out of cancer cells efficiently.
Custom-made removers for different cancer cells
When the researchers tested their degrader library in pancreatic and melanoma cell lines, they discovered that linker shape and rigidity between the USP7-binding part and the VHL-recruiting part strongly influenced performance. After several rounds of design and testing, they arrived at two standout degraders: NK250, which works best in pancreatic cancer cells, and NK266, which is more effective in melanoma cells. These molecules could almost completely remove USP7 at very low doses, and follow-up experiments confirmed that the effect depended on the intended waste-disposal route. Using sensitive assays, the team showed that NK250 in particular forms a stable three-part complex with USP7 and VHL, explaining its rapid action.
Zooming in on what changes inside the cell
To see what happens to the rest of the cell when USP7 is blocked or removed, the scientists measured thousands of proteins in treated cancer cells using advanced mass spectrometry. Degraders produced a surprisingly focused response: USP7 vanished, and a limited set of known partner proteins, especially components of a gene-silencing machine called the Polycomb complex, decreased in both cancer types. These changes loosened the brakes on certain genes that are normally kept quiet, confirming that USP7 supports specific gene-silencing programs. Importantly, many previously suggested USP7 partners did not change in these solid cancer cells, highlighting that the role of USP7 varies strongly between cell types. 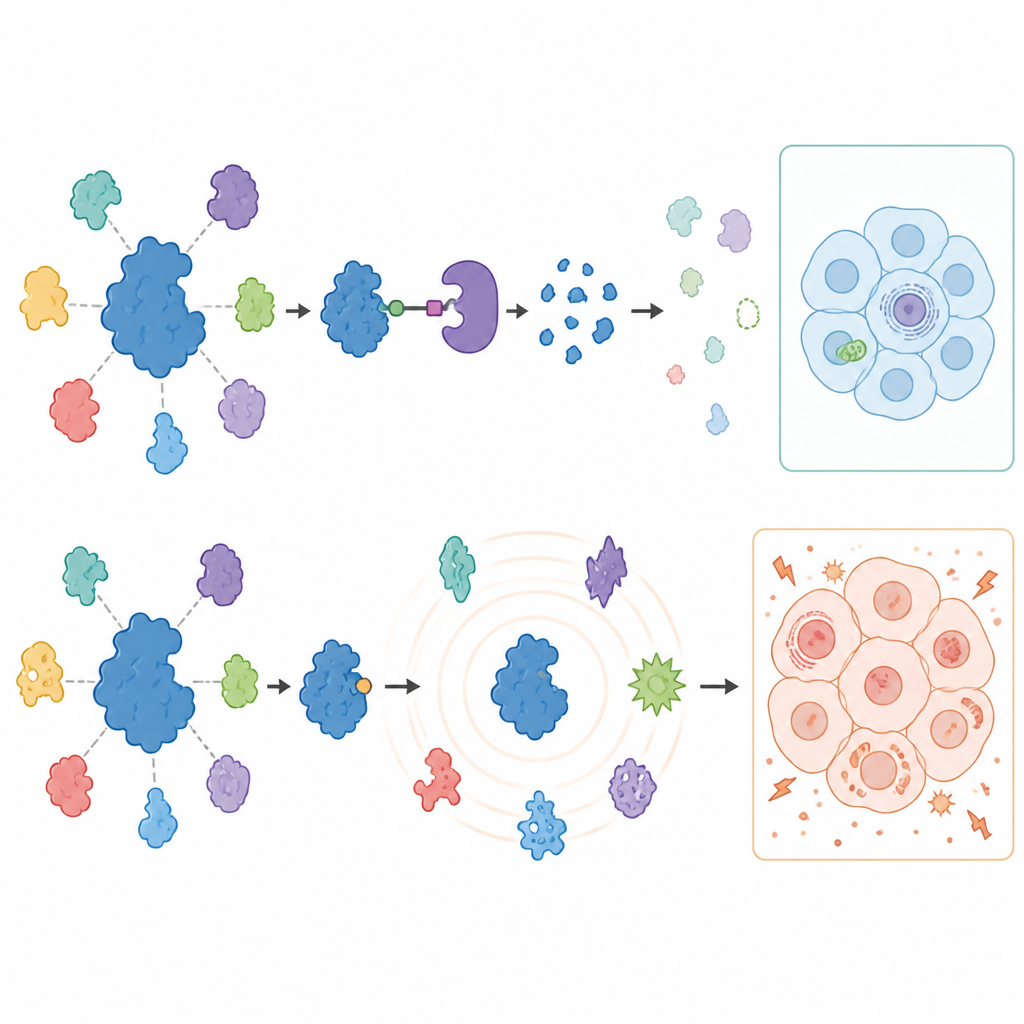
When blocking goes too far beyond the target
Inhibitors told a very different story over longer times. For the first day, the pattern of protein changes largely mirrored what was seen with degraders, pointing to genuine USP7 functions. But after three days of treatment, the inhibitors caused a wave of broad and sometimes strange protein shifts, including the appearance of proteins not usually found in these cancers. The cells also began to rely heavily on sugar, showed reduced ability to generate energy in their mitochondria, and became prone to cell death when glucose was limited. Crucially, these metabolic disturbances still occurred in melanoma cells where USP7 had been removed by gene editing, proving that they were caused by off-target actions of the inhibitors rather than by USP7 itself.
What this means for future cancer drugs
This work shows that carefully designed degraders can remove USP7 with high precision, revealing a focused set of true downstream effects on gene control in solid cancers. In contrast, even well-characterized inhibitors that look clean in early tests can trigger widespread, USP7-independent changes when used for longer periods. For scientists and drug developers, the message is clear: degraders are powerful tools to cleanly dissect what a single protein really does in different cancers, while inhibitors are best used for short-term tests of activity. Together, these complementary tools open a clearer path to understanding and eventually exploiting USP7 and related proteins in cancer therapy.
Citation: Klink, N., Urban, S., Seier, J.A. et al. Targeted degradation of USP7 in solid cancer cells reveals distinct effects of deubiquitinase degraders and inhibitors. Nat Commun 17, 4331 (2026). https://doi.org/10.1038/s41467-026-72295-x
Keywords: USP7, PROTAC degrader, cancer cells, protein inhibition, cell metabolism