Clear Sky Science · en
Type I interferon drives T cell responses to amyloid beta in the central nervous system
Why the Brain’s Immune Response to Plaques Matters
Alzheimer’s disease is often described as a problem of toxic protein buildup in the brain, but an equally important part of the story is how the immune system reacts to that damage. This study explores how a specific branch of the immune system, driven by Type I interferon signals, helps guide T cells toward amyloid beta plaques and reshapes brain inflammation over time. Understanding this shifting immune choreography may help explain why brain damage accelerates in later stages of disease and could point to better-timed treatments.
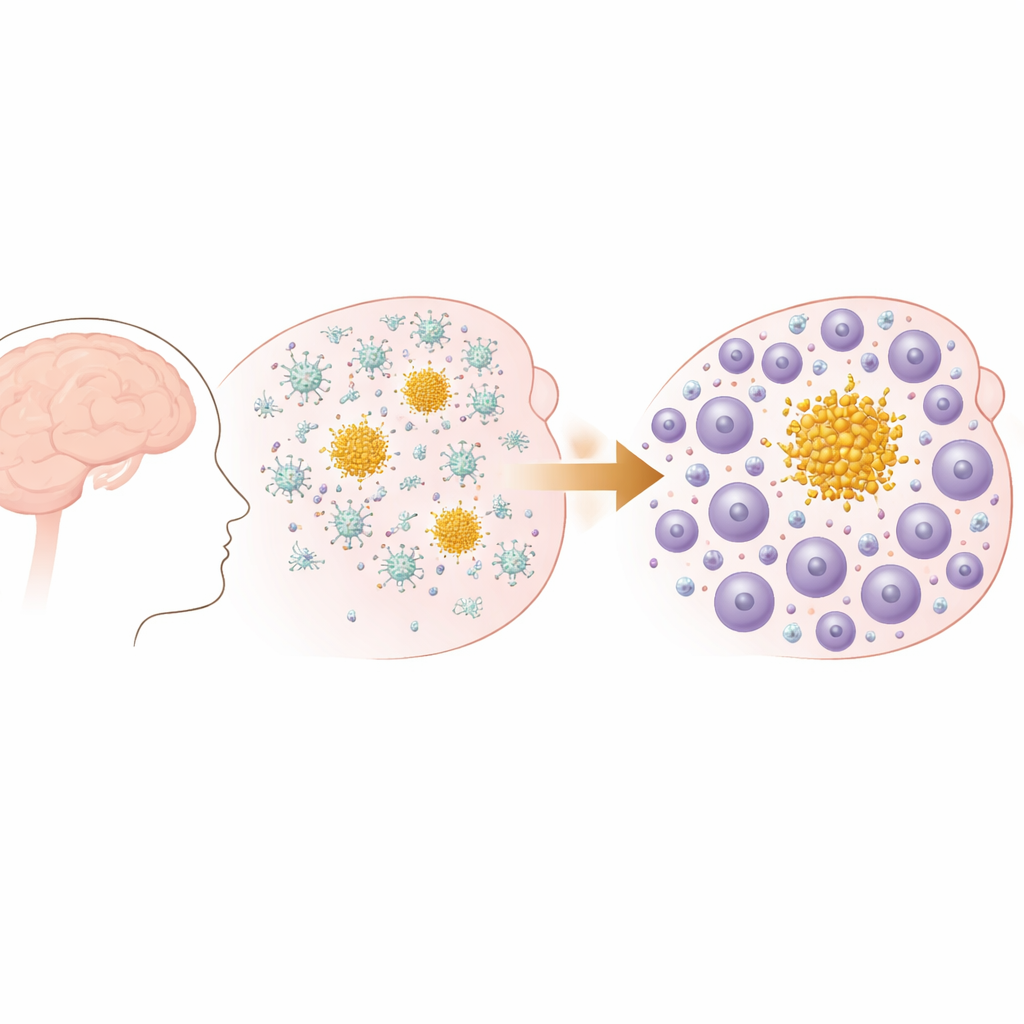
From Early Plaques to a Changing Immune Cast
The researchers used a mouse model that develops progressive amyloid beta plaques in the brain, similar to those seen in Alzheimer’s disease and cerebral amyloid angiopathy. Early in life, these mice form small deposits in the brain’s outer layers, which later spread and increase in number, especially in memory-related regions. Behavioral tests confirmed that as plaques accumulated with age, the mice developed measurable declines in everyday functions such as nesting, mirroring the kind of gradual functional loss seen in human dementia. Importantly, this model has amyloid plaques but no tangle pathology, allowing the team to focus on how the immune system responds specifically to amyloid buildup.
When Brain Guardians Give Way to Invading Foot Soldiers
At first, the immune response to plaques is dominated by microglia, the brain’s resident immune cells. Using single-cell RNA sequencing, the authors showed that in early disease these microglia adopt special disease-associated states and ramp up genes tied to engulfing debris and managing fats. But as the disease advances, the immune landscape shifts. The number of T cells in the brain, especially CD8 T cells, rises sharply, and pathways related to lymphocyte activation and movement become more prominent. Spatial analyses confirmed that this is not a random invasion: T cells cluster tightly around amyloid plaques in brain tissue, particularly around large “parenchymal” plaques rather than those in blood vessels, and their numbers increase in step with plaque burden.
A Specialized T Cell Subset that Calls in Reinforcements
Looking more closely at these T cells, the team identified a distinct subset of CD8 T cells that turns on a suite of genes normally triggered by Type I interferons, a family of antiviral and autoimmune signaling molecules. These “interferon-stimulated” T cells become especially abundant in late disease and are found near plaques. They produce the chemokine CXCL10, a small protein signal that binds to the receptor CXCR3 on other T cells. Spatial transcriptomics showed that both interferon-responsive genes and CXCL10 are strongly enriched in plaque neighborhoods, and that CXCL10-producing T cells are positioned to influence other nearby immune cells. Laboratory migration experiments confirmed that T cells move toward CXCL10 signals and that disabling CXCR3 sharply reduces their ability to follow these cues, supporting a model in which plaque-associated interferon-driven T cells help draw additional T cells into the brain.
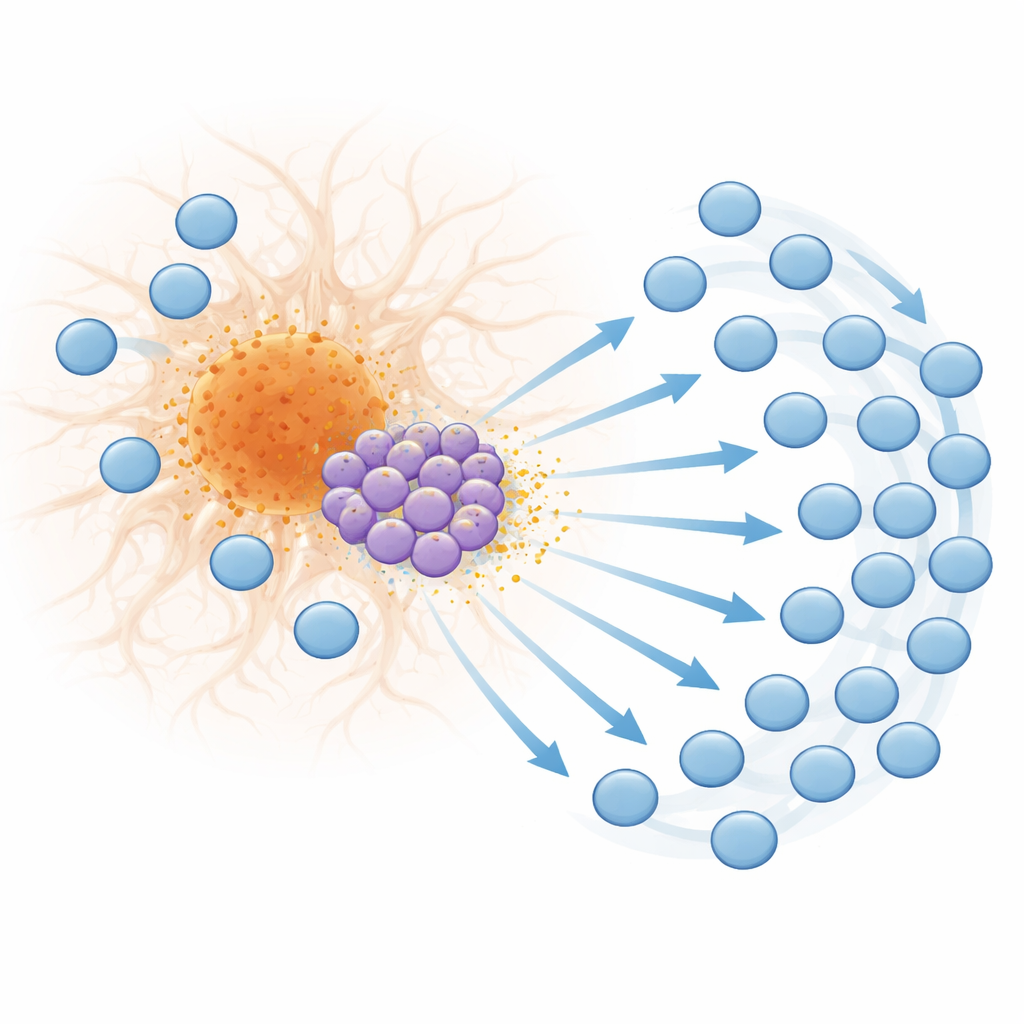
From Activation to Exhaustion at the Plaque Edge
The influx of T cells is not just numerical; it also changes in quality. In late-stage disease, many CD8 T cells around plaques show markers of high activation, cytotoxic potential, and clonal expansion—signs that they are repeatedly responding to specific triggers. At the same time, these cells begin to display hallmarks of exhaustion, a state in which chronically stimulated T cells lose effectiveness and express inhibitory molecules such as PD-1. Spatial mapping revealed that these exhausted T cells accumulate right at the border of plaques, suggesting that intense, ongoing stimulation by plaque-associated signals drives them into a worn-out state. Similar patterns of interferon-responsive genes, CXCL10, T cell activation markers, and PD-1 were found near plaques in human Alzheimer’s brain tissue, indicating that this mouse-derived mechanism is likely relevant to people.
What This Means for Future Alzheimer’s Treatments
Overall, the study paints a time-lapse picture of brain inflammation in amyloid disease: early on, microglia dominate the response, but as plaques grow and persist, a specialized subset of CD8 T cells takes over as the main driver of Type I interferon signaling and T cell recruitment. This evolving immune response may contribute to ongoing nerve cell damage and could interact with antibody-based treatments that remove plaques, potentially influencing side-effects linked to vascular inflammation. For a general reader, the key message is that in Alzheimer’s disease, it is not only the plaques themselves that matter, but also how—and when—the immune system responds to them. Targeting interferon pathways or the CXCL10–CXCR3 signaling axis at the right stage of disease could help calm harmful inflammation without completely silencing the brain’s defenses.
Citation: Michel, J.J., Sanghvi, K., Rosenbauer, J. et al. Type I interferon drives T cell responses to amyloid beta in the central nervous system. Nat Commun 17, 3737 (2026). https://doi.org/10.1038/s41467-026-72262-6
Keywords: Alzheimer’s disease, amyloid beta, T cells, neuroinflammation, interferon signaling