Clear Sky Science · en
SPNS2 exports sphingosine-1-phosphate and imports glucose
A Hidden Gatekeeper for Sugar and Cell Signals
Our bodies constantly juggle thousands of chemical signals while also keeping blood sugar in a safe range. This study uncovers an unexpected player that helps do both at once: a tiny protein called SPNS2 that sits in cell membranes. The researchers show that SPNS2 not only releases a signaling fat molecule important for immunity and blood vessels, but at the same time helps pull glucose—the body’s main fuel—into cells. This dual role links how cells talk to each other with how they feed themselves, with implications for diabetes, heart disease, and cancer.
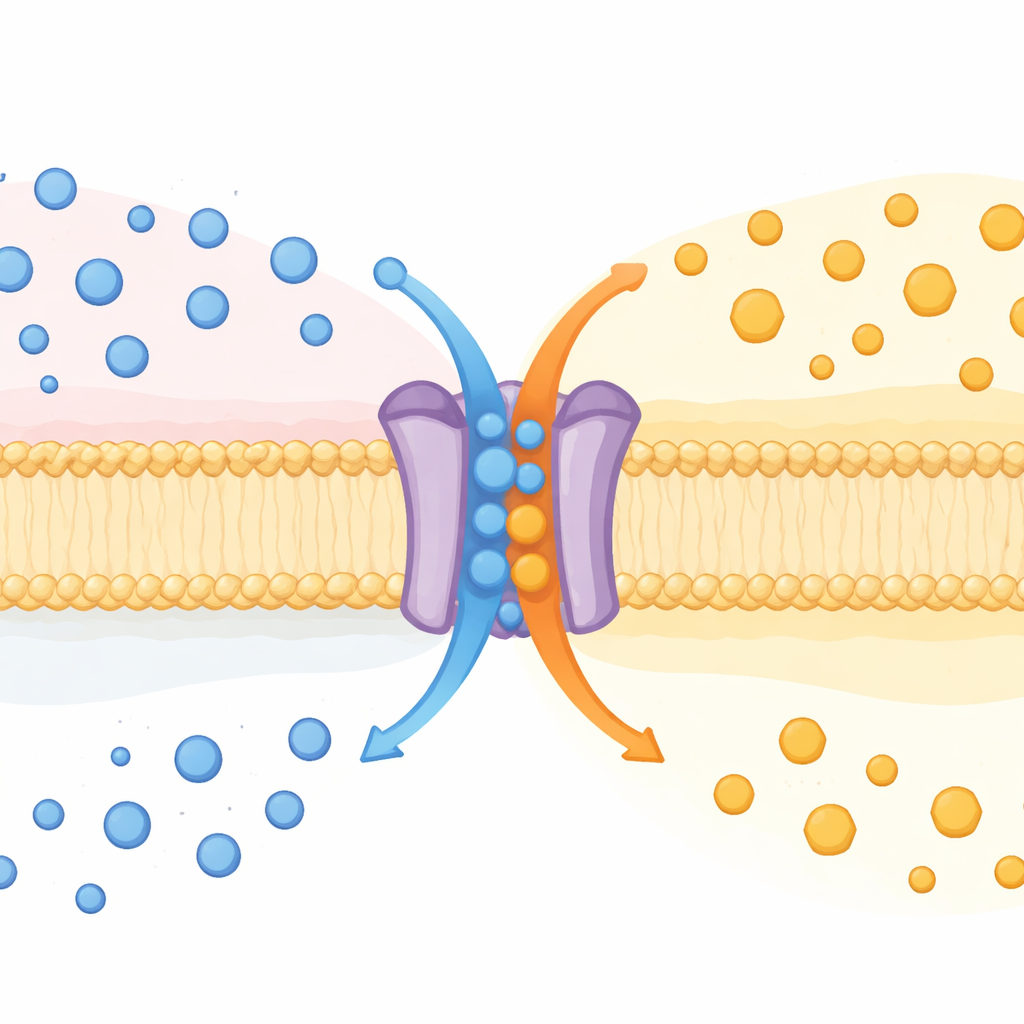
One Protein, Two Jobs
SPNS2 has long been known for moving a messenger molecule called sphingosine‑1‑phosphate (S1P) out of cells. S1P circulates in the blood and lymph, guiding immune cells, shaping blood vessels, and influencing inflammation. Until now, SPNS2 was thought of as a one‑way exporter. By studying mice lacking the Spns2 gene, along with many types of cultured cells, the authors discovered that SPNS2 actually behaves more like an exchange machine. It ships S1P out while bringing glucose in, in a manner similar to an “antiporter,” a class of transporters that swap one substance for another across membranes.
What Happens When SPNS2 Is Missing
In mice engineered to lack SPNS2, levels of S1P in blood and lymph dropped, while S1P built up in tissues such as liver, kidney, lung, and heart—exactly what you would expect if an exporter had been disabled. But their sugar balance was also disturbed. Blood glucose rose, and extra glucose appeared in urine and feces, while several organs showed lower internal glucose levels. Imaging with a radioactive glucose‑like tracer revealed that these mice absorbed sugar from the gut normally, but their tissues—and especially their kidneys—were poor at reclaiming glucose from the bloodstream and from filtered urine. Together, these findings pointed to a defect in getting glucose into cells and reusing it, despite normal insulin levels and hormone signals.
SPNS2 Pulls in Fuel as It Pushes Out Signals
To test whether SPNS2 itself moves glucose, the team manipulated its levels in blood vessel and other cell types. When they boosted SPNS2, cells released more S1P into the surrounding liquid and took up more glucose, producing more energy and showing stronger respiration and sugar burning. When they deleted or blocked SPNS2, cells kept S1P inside and took up less glucose. These effects persisted even when classic sugar transporters (like GLUT1 and GLUT3) were inhibited, and they did not depend on S1P receptors on the cell surface or on insulin. In carefully controlled artificial “proteoliposomes”—tiny lab‑made bubbles with purified SPNS2 inserted in their membranes—the protein could directly move fluorescent or radioactive glucose inward, but only efficiently when S1P was present on the inner side. This showed that SPNS2 itself can act as a glucose importer linked to S1P export.
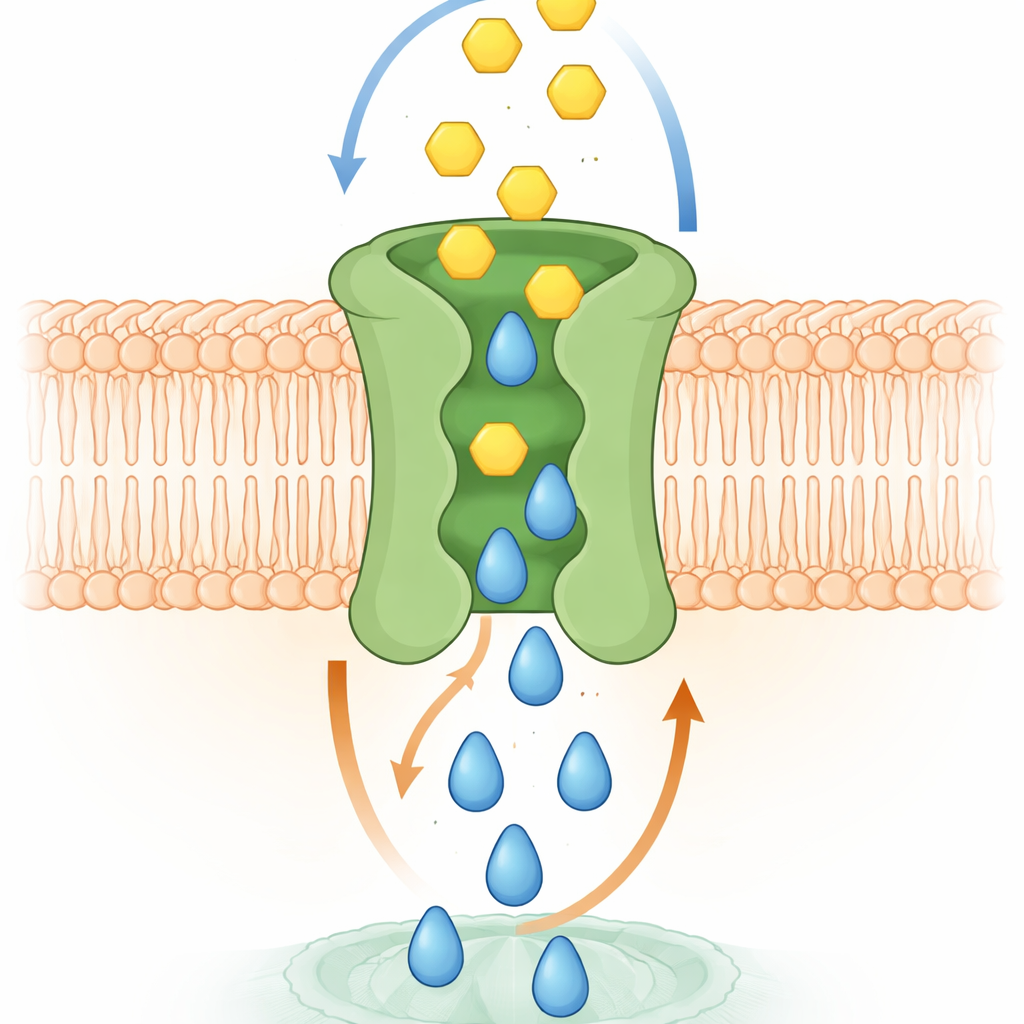
How the Molecular Swap Likely Works
Computer simulations and structural experiments helped explain the mechanism. High‑resolution models of SPNS2 in a membrane showed S1P sliding upward through a central cavity, triggering subtle shape changes that open temporary pockets for glucose on the outer side. As S1P moves toward the outside, glucose can dock in these pockets and then slip down through the same central pathway toward the cell interior. The researchers identified specific amino acids that contact glucose in these pockets. When these residues were mutated, glucose binding dropped and transport failed, even though the altered proteins still reached the cell surface. In living cells, losing this coupled transport weakened S1P‑driven processes such as cell growth, movement to close a wound‑like gap, and the ability of endothelial cell layers to form a tight barrier—functions that require both signal and energy.
Why This Matters for Health and Disease
By revealing that SPNS2 is a two‑way gate—sending out S1P while bringing in glucose—this work links a powerful signaling molecule to basic energy supply in a single step. When SPNS2 releases S1P to trigger immune or vascular responses, it simultaneously helps cells import extra fuel to power those actions. Disruptions in this coupling may contribute to high blood sugar, kidney sugar loss, and insulin‑independent forms of dysregulated glucose handling. Because SPNS2 and S1P signaling also shape cancer spread, inflammation, and blood vessel integrity, targeting this “signal‑and‑sugar” gatekeeper could open new avenues for treating metabolic diseases, autoimmune conditions, and tumors that rely on both heightened signaling and increased glucose uptake.
Citation: Weigel, C., Hossen, M.L., Brown, R.D.R. et al. SPNS2 exports sphingosine-1-phosphate and imports glucose. Nat Commun 17, 3901 (2026). https://doi.org/10.1038/s41467-026-71659-7
Keywords: glucose transport, sphingosine-1-phosphate, membrane transporter, endothelial biology, metabolic signaling