Clear Sky Science · en
Engineering a compact high-fidelity Staphylococcus aureus Cas9 variant with broader targeting range and mechanistic insights into its activation
Smaller Scissors for Fixing Faulty Genes
Gene-editing tools like CRISPR-Cas9 promise treatments for inherited diseases, but the most popular versions are bulky and can accidentally snip the wrong piece of DNA. This study introduces a compact, highly accurate DNA “scissors” based on an enzyme from Staphylococcus aureus, redesigned to reach many more spots in the genome while still cutting precisely. Because it is small enough to fit into a common gene therapy virus and works well in both cells and mice, it could bring safer DNA repair closer to real-world medical use.
Why Size and Accuracy Matter
The original workhorse of genome editing, SpCas9, comes from Streptococcus pyogenes. It is powerful but large, which makes it difficult to package into adeno-associated virus (AAV) vectors that are widely used to deliver gene therapies. It also only recognizes short DNA tags, called PAMs, of the form NGG. These limitations leave many disease-causing mutations out of reach. A smaller cousin, SaCas9 from Staphylococcus aureus, fits comfortably inside AAV and cuts DNA efficiently, but demands a longer and rarer PAM sequence, sharply narrowing the number of editable sites. Attempts to loosen these PAM rules in various Cas9 versions often created a trade-off: more targets but more off-target damage.
Engineering a Broader, Safer Gene Editor
Using detailed structural knowledge of SaCas9, the authors systematically replaced amino acids that make direct, letter-specific contacts with the PAM sequence. They then introduced new positively charged residues that grip the DNA backbone more generally. This produced a variant named SaCas9-NNG, which recognizes simple NNG PAMs instead of the stricter NNGRRT pattern, greatly enlarging the number of potential editing sites. Further fine-tuning removed a subtle clash that had weakened activity at some sequences. In test-tube reactions and human cells, SaCas9-NNG efficiently cut or rewrote DNA at a wide panel of NNG sites, especially those ending in C or T that the original enzyme could not touch. Across many targets, its performance rivaled or exceeded other “PAM-relaxed” editors such as SpRY, SpG, and iGeoCas9.
Making the Edits More Trustworthy
Expanding where Cas9 can cut raises a critical question: can it still avoid the wrong places? To address this, the team engineered a high-fidelity version by weakening certain contacts that help the protein tolerate mismatches between its guide RNA and the DNA. Two key substitutions in a region that stabilizes the far end of the RNA–DNA helix made the enzyme less forgiving of mismatches, particularly those distant from the PAM that are a common source of off-target cuts. This variant, called eSaCas9-NNG, kept strong activity on correct targets but sharply reduced cutting at imperfect ones. Comparisons in human cells showed that it matched or outperformed an established high-fidelity SaCas9 and had fewer spurious cuts than its lower-fidelity relatives.
From Test Tube to Treating Bleeding Disorders
To test whether the compact editor could work inside a living organism, the researchers packaged SaCas9-NNG and its guide RNA into a single AAV8 vector and injected it into mice. Targeting the human clotting factor 9 (F9) gene, important in hemophilia B, they saw efficient introduction of small DNA changes (indels) at NNG sites in the liver, including sites that standard SaCas9 could not reach. They also created base-editing versions that swap a single DNA letter without cutting the strand. In mouse models carrying human hemophilia B mutations, the adenine base editor built from SaCas9-NNG corrected disease-causing changes at both a classic PAM and a previously inaccessible one. Treated animals showed substantial increases in clotting factor activity in their blood, indicating functional rescue.
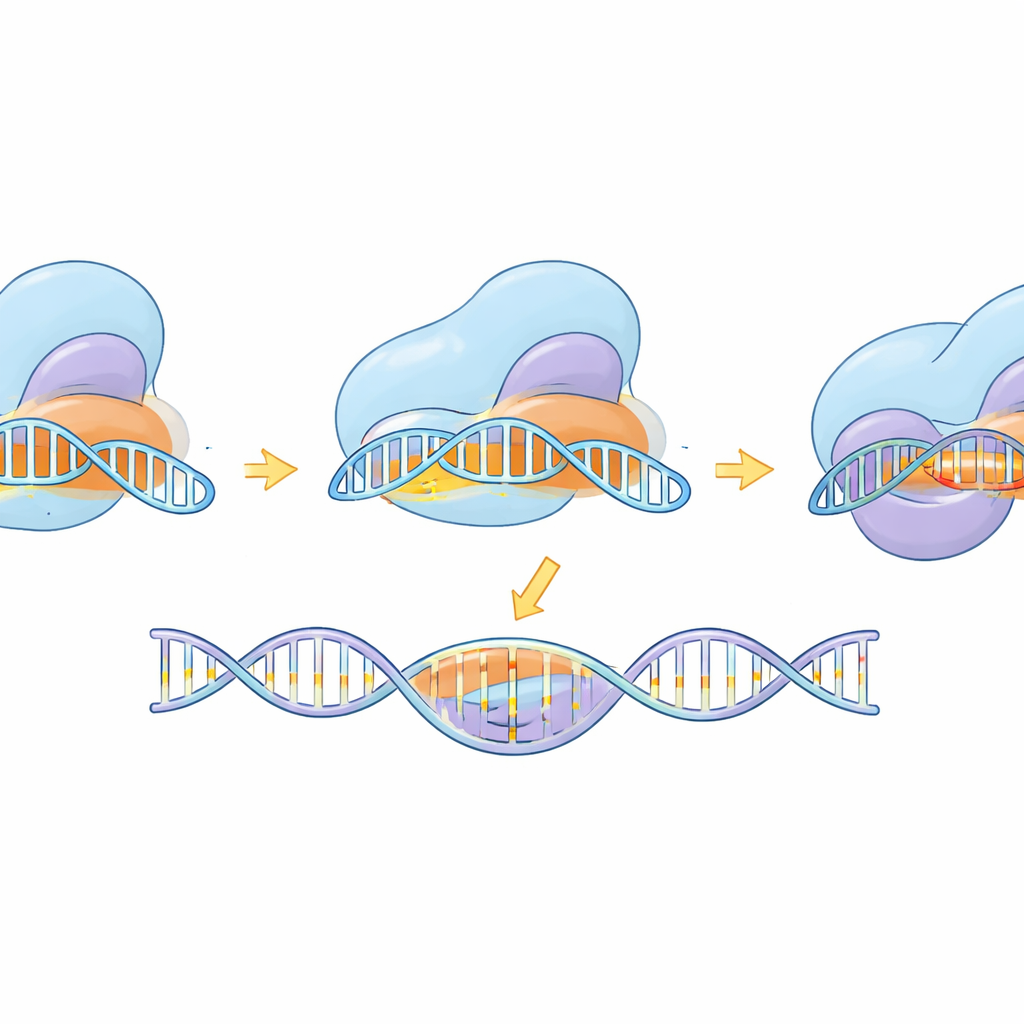
Watching the Molecular Machine in Action
Beyond tool-building, the study used cryo-electron microscopy to capture eSaCas9-NNG in multiple snapshots along its working cycle—before binding DNA, while testing potential sites, during partial pairing with the target, and in fully activated form ready to cut. These structures reveal how the enzyme first latches onto a compatible NNG tag, then gradually forms an RNA–DNA hybrid helix. Only when the helix is long and correctly matched do internal domains swing into place and align the cutting centers on both DNA strands. The engineered mutations that improved specificity were seen to destabilize binding to distorted, mismatched DNA at the far end of the helix, favoring release over erroneous cutting. Comparison to a related, more flexible SpCas9 variant shows that, despite different shapes, both follow a shared logic: full, accurate base pairing is required to unlock the final cutting motion.
What This Means for Future Gene Therapies
For non-specialists, the central message is that the authors have crafted a smaller, more versatile, and more careful gene-editing tool. eSaCas9-NNG can target a far larger fraction of the genome than the original SaCas9, yet maintains high precision, and is compact enough to be delivered in a single AAV package—a key requirement for many in vivo therapies. Its success in correcting clotting defects in hemophilia B mouse models, together with detailed structural insight into how it works and avoids mistakes, positions this enzyme as a promising backbone for next-generation treatments and for future refinements such as even more precise base and prime editors.
Citation: Omura, S.N., Nakagawa, R., Kajimoto, S. et al. Engineering a compact high-fidelity Staphylococcus aureus Cas9 variant with broader targeting range and mechanistic insights into its activation. Nat Commun 17, 3584 (2026). https://doi.org/10.1038/s41467-026-71626-2
Keywords: CRISPR, gene therapy, Cas9, base editing, hemophilia B