Clear Sky Science · en
Rapid evolution of Klebsiella pneumoniae biofilms in vitro delineates adaptive changes selected during infection
Why sticky hospital germs matter
Many hospital infections start not in our tissues, but on the plastic and silicone surfaces of catheters, breathing tubes, and other medical devices. On these artificial surfaces, bacteria such as Klebsiella pneumoniae can build slimy, protective communities called biofilms that are hard to remove and difficult to treat with antibiotics. This study asks a simple but crucial question: how quickly, and in what ways, do these common hospital germs evolve to become better at forming biofilms on catheter‑like surfaces—and what does that mean for their ability to cause disease?
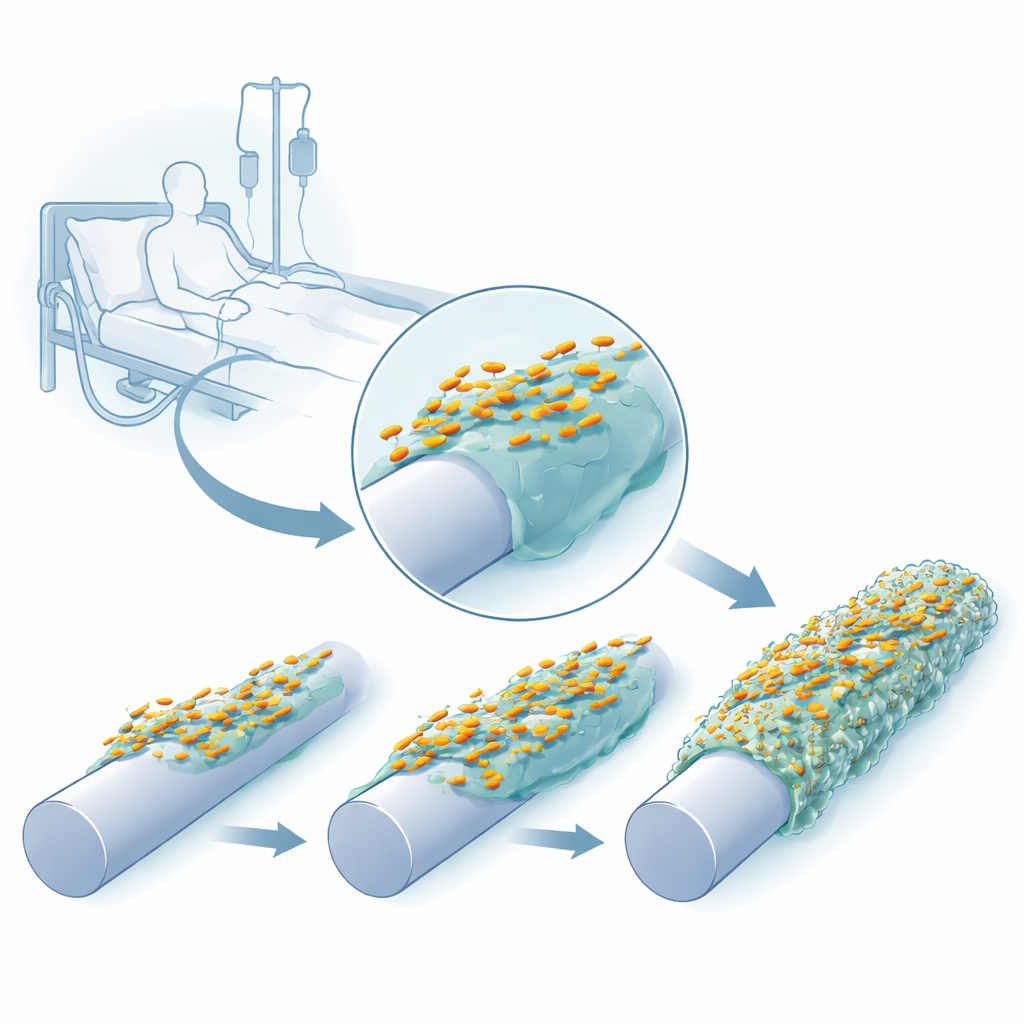
Following bacteria as they adapt to plastic
The researchers recreated a catheter‑like environment in the lab using a peg device whose surfaces could be left bare, coated with silicone, or covered with both silicone and a human blood protein, fibrinogen, which naturally collects on inserted catheters. They repeatedly grew three clinical strains of K. pneumoniae on these pegs, harvested the attached cells, and used them to start the next cycle. Within just a few growth cycles, the bacteria formed thicker, more robust biofilms, and their colonies on agar plates began to look different—some becoming unusually slimy and stringy, others turning more translucent or forming wrinkled surfaces.
Genetic shortcuts to becoming stickier
By sequencing the genomes of over a hundred evolved clones, the team showed that these new appearances reflected specific genetic changes that repeatedly arose in independent lineages. Many mutations hit the capsule system, a sugary coat that normally helps the bacteria evade immune attack. Subtle alterations in a key capsule protein (Wzc) produced hyper‑slimy, highly viscous cells that spread along silicone and fibrinogen‑coated surfaces, while other mutations shut capsule production off entirely, yielding non‑slimy but strongly adhesive cells. A second major target was the tip protein (MrkD) of a hair‑like surface structure (type 3 fimbriae) that acts as a grappling hook. Small structural tweaks in this protein, or changes in the signaling networks that regulate these fimbriae, boosted early attachment and altered how biofilms spread and clumped on different surfaces.
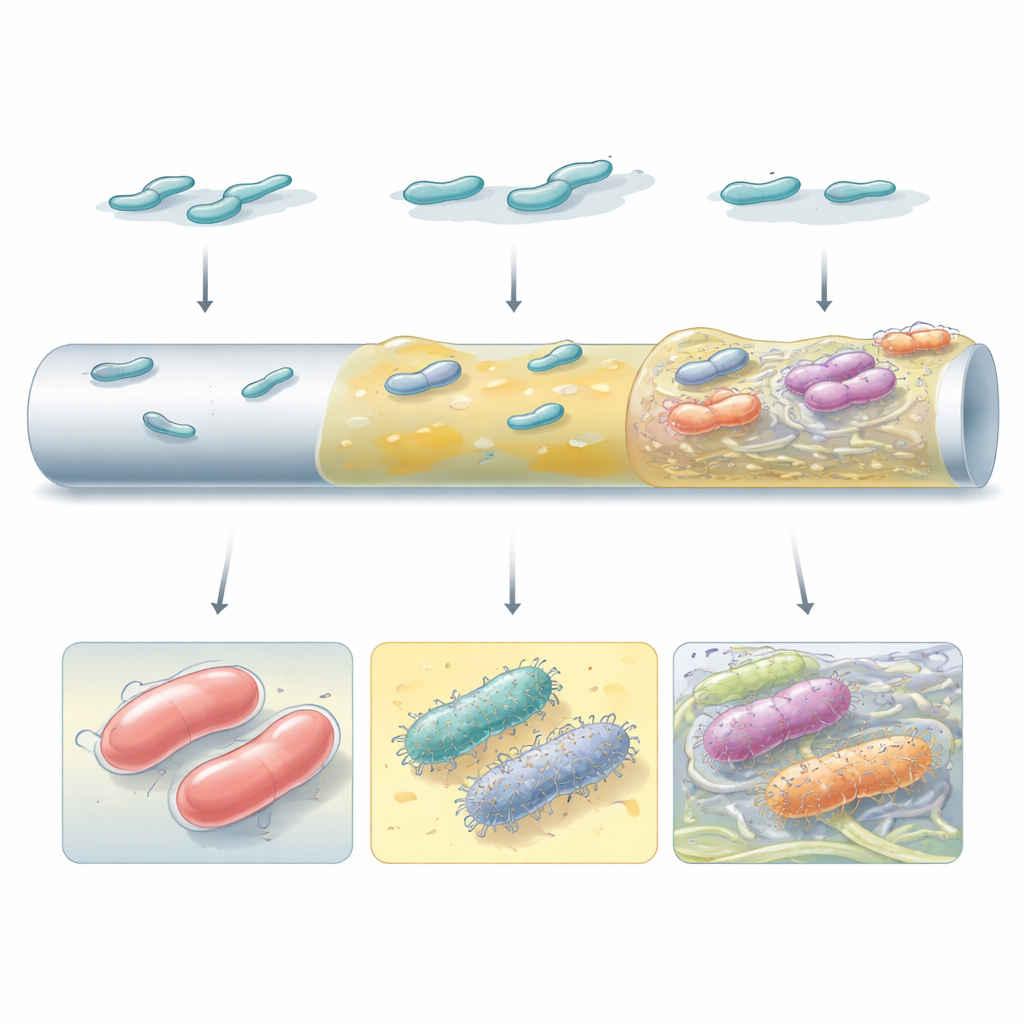
Biofilms built from different building blocks
Not all high‑performing biofilms looked or worked the same. Some mutants relied on thick capsule material to form smooth, continuous layers over the catheter‑like surface. Others, especially those that had lost their capsule or rewired internal signaling via the small molecule c‑di‑GMP, produced abundant surface hairs and cellulose fibers that tied cells together into discrete clusters. Electron microscopy revealed that these distinct strategies created very different architectures: from uniform slime coats to patchy, tower‑like aggregates embedded in a fibrous matrix. Which strategy won out depended on both the bacterial strain and the surface type, underscoring that genetic background and local environment jointly shape how biofilms evolve.
A trade‑off between sticking around and striking hard
Becoming a better biofilm former came with hidden costs. When grown as free‑swimming cells in human serum, both capsule‑deficient mutants and the hyper‑slimy Wzc variants were far more easily killed by complement, a key arm of our innate immune system. Yet once organized in a biofilm, these same mutants were protected from serum attack. In an infection model using wax moth larvae, some biofilm‑enhanced mutants became less deadly, while others did not change or even increased in virulence. This showed that “more biofilm” does not automatically mean “more severe disease”; the exact mutation matters.
Mirroring what happens inside patients
Strikingly, many of the same genes—and even identical single‑letter DNA changes—seen in the lab‑evolved biofilm specialists were also found in K. pneumoniae isolates collected from patients during a real hospital outbreak, especially from urinary tract and wound infections involving catheters. This close overlap suggests that catheter surfaces in the body exert similar selective pressures to those in the lab, steadily favoring variants that are better at clinging, clustering, and surviving on devices. At the same time, some of these adaptations can weaken the bacteria’s ability to cause fast, invasive disease, potentially shifting them toward persistence and chronic infection. Understanding these evolutionary trade‑offs may help in designing devices and treatments that disrupt biofilm‑friendly paths before they become entrenched.
Citation: Zaborskytė, G., Coelho, P., Wrande, M. et al. Rapid evolution of Klebsiella pneumoniae biofilms in vitro delineates adaptive changes selected during infection. Nat Commun 17, 3454 (2026). https://doi.org/10.1038/s41467-026-71505-w
Keywords: Klebsiella pneumoniae, biofilms, catheter-associated infections, bacterial evolution, antibiotic resistance