Clear Sky Science · en
Balancing the efficacy and safety of chimeric antigen receptor T-cell therapy by affinity combination
Why this matters for future cancer care
Engineered immune cells called CAR T cells can cure some blood cancers, but they can also cause dangerous side effects and sometimes fail in the long run. This study asks a deceptively simple question with big implications: instead of building a single “super-strong” cancer-hunting cell, what if we deliberately mix cells with stronger and weaker binding to the same cancer target to better balance power and safety?
How designer immune cells fight cancer
CAR T cells are regular T cells from a patient that are reprogrammed with an artificial receptor so they can latch onto markers on cancer cells and destroy them. Most approved therapies target a molecule called CD19 on B cells and use receptors that grip this marker far more tightly than natural T-cell receptors do. That extra-strong binding helps them recognize and kill cancer cells, but it also drives very intense activation. This can lead to severe immune reactions such as cytokine release syndrome (CRS), damage to healthy cells, early exhaustion of the CAR T cells, and cancer relapses when tumor cells lose or reduce CD19.
Finding the sweet spot in receptor strength
To explore how binding strength shapes CAR T behavior, the researchers compared a standard high-affinity CD19 CAR (JCAR017) with a lower-affinity version (JCAR021) and then engineered a large panel of further variants, some with binding more like a natural T-cell receptor. In lab dishes, most of these CAR T cells looked surprisingly similar: as long as the receptor was stably displayed on the cell surface, even quite weak binders could still activate, secrete key immune messengers, and kill CD19-positive target cells. Only the very weakest, TCR-like variants started to show reduced function under demanding conditions, such as when target cells carried very little CD19.
What happens inside living organisms
When the team moved into mouse models, differences between strong and weak binders became much more pronounced. High-affinity CAR T cells expanded briskly, homed efficiently into the bone marrow where CD19-positive lymphoma cells resided, and controlled tumors best. Lower-affinity CARs delayed tumor growth but rarely matched the top performer, and ultra-weak binders were largely ineffective. At the same time, the strong binders drove more intense activation, faster differentiation of T cells, and—importantly in a humanized mouse model with a reconstituted immune system—more severe CRS-like illness that worsened as the CAR T-cell dose increased.
Mixing strong and gentle hunters
Inspired by how our natural immune system recruits a mix of high- and low-affinity T cells, the authors tested a “blended” CAR T product that combined equal numbers of high- and low-affinity CD19 CAR T cells. In laboratory co-cultures with cancer cells and human monocytes, these mixed products killed targets as effectively as a full-dose high-affinity product, but released fewer inflammatory cytokines, resembling the profile of a half-dose of strong CAR T cells. Under repeated or chronic exposure to cancer cells, low-affinity CAR T cells gradually became the dominant population in the mixture, whereas high-affinity CAR T cells alone showed more markers of exhaustion and stress.
How blending changes long-term behavior
In tumor-bearing mice, the combined product again performed best overall. High-affinity CAR T cells were still essential: low-affinity CARs alone could not fully clear tumors, even at higher doses. But when high- and low-affinity cells were transferred together at fixed total doses, tumors shrank faster and more completely than with the same number of high-affinity cells alone. Single-cell RNA sequencing of recovered CAR T cells revealed that high-affinity cells in the mixed groups expressed fewer genes linked to exhaustion and terminal differentiation, and more genes associated with sustained cytotoxic function and tissue residency. This suggests that low-affinity partner cells help “share the load” of antigen recognition, limiting over-stimulation of the strongest binders.
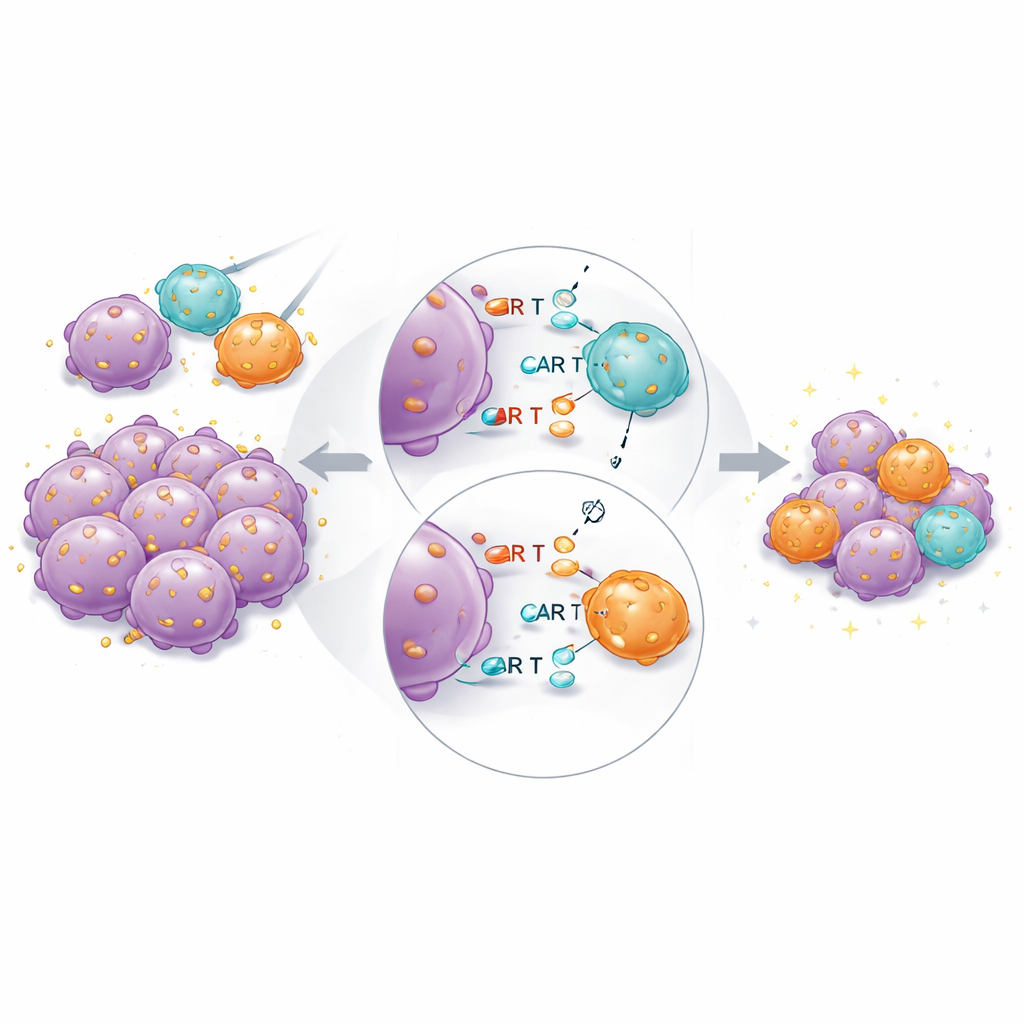
What this means for patients
The study shows that there is a practical lower limit to how weak a CAR can bind and still work well in current designs, and that binding strength which maximizes tumor killing also tends to magnify toxicity and burnout. Rather than searching for a single perfect affinity, mixing CAR T cells with different binding strengths to the same cancer marker emerges as a promising strategy. In this approach, high-affinity cells provide rapid, powerful attack, while lower-affinity cells help dampen runaway inflammation and sustain long-term control. If confirmed in clinical trials and expanded to other targets beyond CD19, such affinity-combined CAR T products could widen the treatment window—making cell therapies safer, more durable, and suitable for a broader range of cancers.
Citation: Warmuth, L., Dötsch, S., Trebo, M. et al. Balancing the efficacy and safety of chimeric antigen receptor T-cell therapy by affinity combination. Nat Commun 17, 3413 (2026). https://doi.org/10.1038/s41467-026-71354-7
Keywords: CAR T-cell therapy, CD19, receptor affinity, cytokine release syndrome, cancer immunotherapy